Abstract
Background
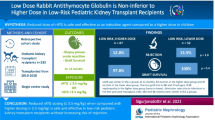
Chronic antibody-mediated rejection (cAMR) is the leading cause of late kidney graft loss, but current therapies are often ineffective. Rabbit anti-human thymocyte immunoglobulin (rATG) may be helpful, but its use is virtually undocumented.
Methods
Data were analyzed retrospectively from nine pediatric kidney transplant patients with cAMR were treated with rATG (1.5 mg/kg × 5 days) at our center after non-response to pulsed prednisolone, intravenous immunoglobulin, rituximab, and increased immunosuppressive intensity (including switching to belatacept in some cases), with or without bortezomib.
Results
The median time from diagnosis to cAMR was 179 days. rATG was started 5–741 days after diagnosis. Median estimated glomerular filtration rate (eGFR) increased from 40 mL/min/1.73 m2 when rATG was started to 62 mL/min/1.73 m2 9 months later (p = 0.039). Four patients showed substantially higher eGFR after 9 months and 2 patients showed a small improvement; eGFR continued to decline in 3 patients after starting rATG. No grafts were lost during follow-up. At last follow-up, donor-specific antibodies (DSAs) were no longer detectable in 4 out of 8 patients for whom data were available, median fluorescence intensity had decreased substantially in 1 out of 8 patients; anti-HLA DQ DSAs persisted in 2 out of 8 patients. No adverse events with a suspected relation to rATG, including allergic reactions, leukocytopenia or infections, were observed in any of the patients.
Conclusions
In this small series of patients, rATG appears a promising treatment for unresponsive cAMR. Further evaluation, including earlier introduction of rATG, is warranted.



Similar content being viewed by others
Change history
08 February 2018
The article “Rabbit anti-human thymocyte immunoglobulin for the rescue treatment of chronic antibody-mediated rejection after pediatric kidney transplantation”, written by Yasemen Cihan, Nele Kanzelmeyer, Jens Drube, Martin Kreuzer, Christian Lerch, Imke Hennies, Kerstin Froede, Murielle Verboom.
References
Clatworthy MR (2011) Targeting B cells and antibody in transplantation. Am J Transplant 11:1359–1367
Loupy A, Jordan SC (2013) Transplantation: donor-specific HLA antibodies and renal allograft failure. Nat Rev Nephrol 9:130–131
Fotheringham J, Angel C, Goodwin J, Harmer AW, McKane WS (2011) Natural history of proteinuria in renal transplant recipients developing de novo human leukocyte antigen antibodies. Transplantation 91:991–996
Sadaka B, Alloway RR, Woodle ES (2013) Management of antibody-mediated rejection in transplantation. Surg Clin North Am 93:1451–1466
Sellarés J, De Freitas DG, Mengel M, Reeve J, Einecke G, Sis B, Hidalgo LG, Famulski K, Matas A, Halloran PF (2012) Understanding the causes of kidney transplant failure: the dominant role of antibody-mediated rejection and nonadherence. Am J Transplant 12:388–399
El-Zoghby ZM, Stegall MD, Lager DJ, Kremers WK, Amer H, Gloor JM, Cosio FG (2009) Identifying specific causes of kidney allograft loss. Am J Transplant 9:527–535
NAPRTCS 2014 Annual Transplant Report. Available at https://web.emmes.com/study/ped/annlrept/annualrept2014.pdf Accessed 26 May 2017
Kreuzer M, Prüfe J, Oldhafer M, Bethe D, Dierks ML, Müther S, Thumfart J, Hoppe B, Büscher A, Rascher W, Hansen M, Pohl M, Kemper MJ, Drube J, Rieger S, John U, Taylan C, Dittrich K, Hollenbach S, Klaus G, Fehrenbach H, Kranz B, Montoya C, Lange-Sperandio B, Ruckenbrodt B, Billing H, Staude H, Heindl-Rusai K, Brunkhorst R, Pape L (2015) Transitional care and adherence of adolescents and young adults after kidney transplantation in Germany and Austria: a binational observatory census within the TRANSNephro trial. Medicine (Baltimore) 94:e2196
Loupy A, Lefaucheur C, Vernerey D, Prugger C, Duong van Huyen JP, Mooney N, Suberbielle C, Frémeaux-Bacchi V, Méjean A, Desgrandchamps F, Anglicheau D, Nochy D, Charron D, Empana JP, Delahousse M, Legendre C, Glotz D, Hill GS, Zeevi A, Jouven X (2013) Complement-binding anti-HLA antibodies and kidney-allograft survival. N Engl J Med 369:1215–1226
Hourmant M, Cesbron-Gautier A, Terasaki PI, Mizutani K, Moreau A, Meurette A, Dantal J, Giral M, Blancho G, Cantarovich D, Karam G, Follea G, Soulillou JP, Bignon JD (2005) Frequency and clinical implications of development of donor-specific and non-donor- specific HLA antibodies after kidney transplantation. J Am Soc Nephrol 16:2804–2812
Kim JJ, Balasubramanian R, Michaelides G, Wittenhagen P, Sebire NJ, Mamode N, Shaw O, Vaughan R, Marks SD (2014) The clinical spectrum of de novo donor-specific antibodies in pediatric renal transplant recipients. Am J Transplant 14:2350–2358
Djamali A, Kaufman DB, Ellis TM, Zhong W, Matas A, Samaniego M (2014) Diagnosis and management of antibody-mediated rejection: current status and novel approaches. Am J Transplant 14:255–271
Burton SA, Amir N, Asbury A, Lange A, Hardinger KL (2015) Treatment of antibody-mediated rejection in renal transplant patients: a clinical practice survey. Clin Transpl 29:118–123
Montgomery RA, Zachary AA, Racusen LC, Leffell MS, King KE, Burdick J, Maley WR, Ratner LE (2000) Plasmapheresis and intravenous immune globulin provides effective rescue therapy for refractory humoral rejection and allows kidneys to be successfully transplanted into cross-match-positive recipients. Transplantation 70:887–895
Rocha PN, Butterly DW, Greenberg A, Reddan DN, Tuttle-Newhall J, Collins BH, Kuo PC, Reinsmoen N, Fields T, Howell DN, Smith SR (2003) Beneficial effect of plasmapheresis and intravenous immunoglobulin on renal allograft survival of patients with acute humoral rejection. Transplantation 75:1490–1495
Montgomery RA, Orandi BJ, Racusen L, Jackson AM, Garonzik-Wang JM, Shah T, Woodle ES, Sommerer C, Fitts D, Rockich K, Zhang P, Uknis ME (2016) Plasma-derived C1 esterase inhibitor for acute antibody-mediated rejection following kidney transplantation: results of a randomized double-blind placebo-controlled pilot study. Am J Transplant 16:3468–3478
Choi J, Aubert O, Vo A, Loupy A, Haas M, Puliyanda D, Kim I, Louie S, Kang A, Peng A, Kahwaji J, Reinsmoen N, Toyoda M, Jordan SC (2017) Assessment of tocilizumab (anti-interleukin-6 receptor monoclonal) as a potential treatment for chronic antibody-mediated rejection and transplant glomerulopathy in HLA-sensitized renal allograft recipients. Am J Transplant. doi:10.1111/ajt.14228
Chih S, Tinckam KJ, Ross HJ (2013) A survey of current practice for antibody-mediated rejection in heart transplantation. Am J Transplant 13:1069–1074
Büchler M, Longuet H, Lemoine R, Herr F, Gatault P, Thibault G, Ternant D, Foulon C, Pilorge B, Lemay D, Sung C, Halimi JM, Baron C, Lebranchu Y (2013) Pharmacokinetic and pharmacodynamic studies of two different rabbit antithymocyte globulin dosing regimens: results of a randomized trial. Transpl Immunol 28:120–126
Kho MM, Bouvy AP, Cadogan M, Kraaijeveld R, Baan CC, Weimar W (2012) The effect of low and ultra-low dosages Thymoglobulin on peripheral T, B and NK cells in kidney transplant recipients. Transpl Immunol 26:186–190
Ramos EJ, Pollinger HS, Stegall MD, Gloor JM, Dogan A, Grande JP (2007) The effect of desensitization protocols on human splenic B-cell populations in vivo. Am J Transplant 7:402–407
Pankewycz O, Leca N, Kohli R, Wallace PK, Said M, Feng L, Alnimri M, Patel S, Laftavi MR (2011) Low-dose rabbit antithymocyte globulin induction therapy results in prolonged selective lymphocyte depletion irrespective of maintenance immunosuppression. Transplant Proc 43:462–465
Gurkan S, Luan Y, Dhillon N, Allam SR, Montague T, Bromberg JS, Ames S, Lerner S, Ebcioglu Z, Nair V, Dinavahi R, Sehgal V, Heeger P, Schroppel B, Murphy B (2010) Immune reconstitution following rabbit antithymocyte globulin. Am J Transplant 10:2132–2141
Brokhof MM, Sollinger HW, Hager DR, Muth BL, Pirsch JD, Fernandez LA, Bellingham JM, Mezrich JD, Foley DP, D’Alessandro AM, Odorico JS, Mohamed MA, Vidyasagar V, Ellis TM, Kaufman DB, Djamali A (2014) Antithymocyte globulin is associated with a lower incidence of de novo donor-specific antibodies in moderately sensitized renal transplant recipients. Transplantation 97:612–617
Pascual J, Zuckermann A, Djamali A, Hertig A, Naesens M (2016) Rabbit antithymocyte globulin and donor-specific antibodies in kidney transplantation--a review. Transplant Rev (Orlando) 30:85–91
Pape L, Lehner F, Blume C, Ahlenstiel T (2011) Pediatric kidney transplantation followed by de novo therapy with everolimus, low-dose cyclosporine a, and steroid elimination: 3-year data. Transplantation 92:658–662
Pape L, Offner G, Kreuzer M, Froede K, Drube J, Kanzelmeyer N, Ehrich JH, Ahlenstiel T (2010) De novo therapy with everolimus, low-dose ciclosporine a, basiliximab and steroid elimination in pediatric kidney transplantation. Am J Transplant 10:2349–2354
Gupta A, Murillo D, Yarlagadda SG, Wang CJ, Nawabi A, Schmitt T, Brimacombe M, Bryan CF (2016) Donor-specific antibodies present at the time of kidney transplantation in immunologically unmodified patients increase the risk of acute rejection. Transpl Immunol 37:18–22
Malheiro J, Tafulo S, Dias L, Martins LS, Fonseca I, Beirão I, Castro-Henriques A, Cabrita A (2015) Analysis of preformed donor-specific anti-HLA antibodies characteristics for prediction of antibody-mediated rejection in kidney transplantation. Transpl Immunol 32:66–71
Eskandary F, Bond G, Regele H, Kozakowski N, Kikić Z, Wahrmann M, Haslacher H, Oberbauer R, Ramassar V, Halloran P, Böhmig GA (2014) Late antibody-mediated rejection in a large prospective cross-sectional study of kidney allograft recipients--preliminary results of the screening phase of the BORTEJECT trial. Clin Transpl 2014:189–195
Haas M, Sis B, Racusen LC, Solez K, Glotz D, Colvin RB, Castro MC, David DS, David-Neto E, Bagnasco SM, Cendales LC, Cornell LD, Demetris AJ, Drachenberg CB, Farver CF, Farris AB 3rd, Gibson IW, Kraus E, Liapis H, Loupy A, Nickeleit V, Randhawa P, Rodriguez ER, Rush D, Smith RN, Tan CD, Wallace WD, Mengel M, Banff meeting report writing committee (2014) Banff 2013 meeting report: inclusion of c4d-negative antibody-mediated rejection and antibody-associated arterial lesions. Am J Transplant 14:272–283
Billing H, Rieger S, Süsal C, Waldherr R, Opelz G, Wühl E, Tönshoff B (2012) IVIG and rituximab for treatment of chronic antibody-mediated rejection: a prospective study in paediatric renal transplantation with a 2-year follow-up. Transplant Int 5:1165–1173
Lerch C, Kanzelmeyer NK, Ahlenstiel-Grunow T, Froede K, Kreuzer M, Drube J, Verboom M, Pape L (2017) Belatacept after kidney transplantation in adolescents: a retrospective study. Transpl Int 30:494–501
Walsh RC, Alloway RR, Girnita AL, Woodle ES (2012) Proteasome inhibitor-based therapy for antibody-mediated rejection. Kidney Int 81:1067–1074
Schwartz GJ, Muñoz A, Schneider MF, Mak RH, Kaskel F, Warady BA, Furth SL (2009) New equations to estimate GFR in children with CKD. J Am Soc Nephrol 20:629–637
Reed RP, Zhang Z, Gebel H, Bray RA, Guleria I, Lunz J, Mohanakumar T, Nickerson P, Tambur AR, Zeevi A, Heeger PS, Gjertson D (2013) Comprehensive assessment and standardization of solid phase multiplex-bead arrays for the detection of antibodies to HLA. Am J Transplant 13:1859–1870
Vo AA, Lukovsky M, Toyoda M, Wang J, Reinsmoen NL, Lai CH, Peng A, Villicana R, Jordan SC (2008) Rituximab and intravenous immune globulin for desensitization during renal transplantation. N Engl J Med 359:242–251
Sberro-Soussan R, Zuber J, Suberbielle-Boissel C, Candon S, Martinez F, Snanoudj R, Rabant M, Pallet N, Nochy D, Anglicheau D, Leruez M, Loupy A, Thervet E, Hermine O, Legendre C (2010) Bortezomib as the sole post-renal transplantation desensitization agent does not decrease donor-specific anti-HLA antibodies. Am J Transplant 10:681–686
Haririan A (2015) Current status of the evaluation and management of antibody-mediated rejection in kidney transplantation. Curr Opin Nephrol Hypertens 24:576–581
Frémeaux-Bacchi V, Legendre CM (2015) The emerging role of complement inhibitors in transplantation. Kidney Int 88:967–973
Tagliamacco A, Cioni M, Comoli P, Ramondetta M, Brambilla C, Trivelli A, Magnasco A, Biticchi R, Fontana I, Dulbecco P, Palombo D, Klersy C, Ghiggeri GM, Ginevri F, Cardillo M, Nocera A (2014) DQ molecules are the principal stimulators of de novo donor-specific antibodies in nonsensitized pediatric recipients receiving a first kidney transplant. Transpl Int 27:667–673
Mohty M, Bacigalupo A, Saliba F, Zuckermann A, Morelon E, Lebranchu Y (2014) New directions for rabbit antithymocyte globulin (Thymoglobulin®) in solid organ transplants, stem cell transplants and autoimmunity. Drugs 74:1605–1634
Hertig A, Zuckermann A (2015) Rabbit antithymocyte globulin induction and risk of post-transplant lymphoproliferative disease in adult and pediatric solid organ transplantation: an update. Transpl Immunol 32:179–1787
Popow I, Leitner J, Grabmeier-Pfistershammer K, Majdic O, Zlabinger GJ, Kundi M, Steinberger P (2013) A comprehensive and quantitative analysis of the major specificities in rabbit antithymocyte globulin preparations. Am J Transplant 13:3103–3113
Rebellato LM, Gross U, Verbanac KM, Thomas JM (1994) A comprehensive definition of the major antibody specificities in polyclonal rabbit antithymocyte globulin. Transplantation 57:685–694
Zand MS, Vo T, Huggins J, Felgar R, Liesveld J, Pellegrin T, Bozorgzadeh A, Sanz I, Briggs BJ (2005) Polyclonal rabbit antithymocyte globulin triggers B-cell and plasma cell apoptosis by multiple pathways. Transplantation 79:1507–1515
Kohei N, Tanaka T, Tanabe K, Masumori N, Dvorina N, Valujskikh A, Baldwin WM 3rd, Fairchild RL (2016) Natural killer cells play a critical role in mediating inflammation and graft failure during antibody-mediated rejection of kidney allografts. Kidney Int 89:1293–1306
Kamburova EG, Koenen HJ, Borgman KJ, Ten Berge IJ, Joosten I, Hilbrands LB (2013) A single dose of rituximab does not deplete B cells in secondary lymphoid organs but alters phenotype and unction. Am J Transplant 13:1503–1511
Jackson AM, Kraus ES, Orandi BJ, Segev DL, Montgomery RA, Zachary AA (2015) A closer look at rituximab induction on HLA antibody rebound following HLA-incompatible kidney transplantation. Kidney Int 87:409–416
Becker YT, Becker BN, Pirsch JD, Sollinger HW (2004) Rituximab a treatment for refractory kidney transplant rejection. Am J Transplant 4:996–1001
Zheng J, Xue W, Qing X, Jing X, Hou J, Tian X, Guo Q, He X, Cai J (2014) Treatment of biopsy-proven acute antibody-mediated rejection using Thymoglobulin (ATG) monotherapy and a combination of rituximab, intravenous immunoglobulin, and plasmapheresis: lesson learned from primary experience. Clin Transpl 2014:223–230
Chehade H, Rotman S, Matter M, Girardin E, Aubert V, Pascual M (2015) Eculizumab to treat antibody-mediated rejection in a 7-year-old kidney transplant recipient. Pediatrics 132:e551–e555
Kutzler HL, Ye X, Rochon C, Martin ST (2016) Administration of antithymocyte globulin (rabbit) to treat a severe, mixed rejection episode in a pregnant renal transplant recipient. Pharmacotherapy 36:e18–e22
Kizilbash S, Claes D, Ashoor I, Chen A, Jandeska S, Matar RB, Misurac J, Sherbotie J, Twombley K, Verghese P (2017) Bortezomib in the treatment of antibody-mediated rejection in pediatric kidney transplant recipients: a multicenter Midwest Pediatric Nephrology Consortium study. Pediatr Transplant 21(3):e12873. doi:10.1111/petr.12873
Pearl MH, Nayak AB, Ettenger RB, Puliyanda D, Palma Diaz MF, Zhang Q, Reed EF, Tsai TW (2016) Bortezomib may stabilize pediatric renal transplant recipients with antibody-mediated rejection. Pediatr Nephrol 31:1341–1348
O’Leary JG, Samaniego M, Barrio MC, Potena L, Zeevi A, Djamali A, Cozzi E (2016) The influence of immunosuppressive agents on the risk of de novo donor-specific HLA antibody production in solid organ transplant recipients. Transplantation 100:39–53
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The study received no external funding. Medical writing support by a freelance medical writer was funded by Sanofi.
Ethics
This retrospective study did not require ethical approval.
Conflicts of interest
Lars Pape has received speaker’s honoraria and travel grants from Novartis Pharmaceuticals. None of the other authors has any conflicts of interest to declare.
Additional information
A correction to this article is available online at https://doi.org/10.1007/s00467-017-3858-2.
Rights and permissions
About this article
Cite this article
Cihan, Y., Kanzelmeyer, N., Drube, J. et al. Rabbit anti-human thymocyte immunoglobulin for the rescue treatment of chronic antibody-mediated rejection after pediatric kidney transplantation. Pediatr Nephrol 32, 2133–2142 (2017). https://doi.org/10.1007/s00467-017-3725-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-017-3725-1