Abstract
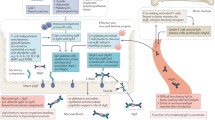
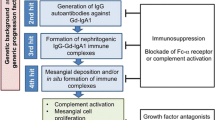
Recent data suggest that gut-associated lymphoid tissue (GALT) plays a major role in the development of immunoglobulin A (IgA) nephropathy (IgAN). A genome-wide association study showed that most loci associated with the risk of IgAN are also associated with immune-mediated inflammatory bowel diseases, maintenance of the intestinal barrier and regulation of response to gut pathogens. Studies involving experimental models have demonstrated a pivotal role of intestinal microbiota in the development of IgAN in mice producing high levels of IgA and in transgenic mice overexpressing BAFF, a B-cell factor crucial for IgA synthesis, indicating the role of genetic background, B-cell activity, GALT intestinal immunity and diet. The effect of diet was suggested by pilot studies carried out 30 years ago which showed that a gluten-rich diet induced IgAN in mice and that some patients benefited from a gluten-free diet. A recent experimental model in mice expressing human IgA1 and Fc alpha receptor CD89 reported clinical and histological improvement after a gluten-free diet. Clinical observations have elicited new interest in GALT hyper-reactivity in IgAN patients. In a pilot study, a reduction in proteinuria was attained using an enteric controlled-release formulation of the corticosteroid budesonide targeted to the Peyer’s patches at the ileocecal junction. This formulation was tested in the placebo-controlled NEFIGAN phase 2b trial, with a reduction in proteinuria after 9 months of treatment together with stabilization of renal function in patients with persistent proteinuria. In conclusion, the gut–kidney axis modulated by microbiota and diet is a promising target for focused treatment of IgAN in genetically predisposed patients at risk of progression.
Similar content being viewed by others
References
Wyatt RJ, Julian BA (2013) IgA nephropathy. N Engl J Med 368:2402–2414
Coppo R, Amore A, Peruzzi L, Vergano L, Camilla R (2010) Innate immunity and IgAnephropathy. J Nephrol 23:626–632
Coppo R (2015) The intestine–renal connection in IgA nephropathy. Nephrol Dial Transplant 30:360–366
Cesta MF (2006) Normal structure, function, and histology of mucosa-associatedlymphoid tissue. Toxicol Pathol 34:599–608
Kang W, Kudsk K (2007) Is there evidence that the gut contributes to mucosal immunity in human? J Parenter Enter Nutr 31:246–258
Kiryluk K, Novak J (2014) The genetics and immunobiology of IgA nephropathy. J Clin Invest 124:2325–2332
Woof JM, Kerr MA (2006) The function of immunoglobulin A in immunity. J Pathol 208:270–282
Coppo R (2008) Pediatric IgA nephropathy: clinical and therapeutic perspectives. Semin Nephrol 28:18–26
Oortwijn BD, Rastaldi MP, Roos A, Mattinzoli D, Daha MR, van Kooten C (2007) Demonstration of secretory IgA in kidneys of patients with IgA nephropathy. Nephrol Dial Transplant 22:3191–3195
Barratt J, Bailey EM, Buck KS, Mailley J, Moayyedi P, Feehally J, Turney JH, Crabtree JE, Allen AC (1999) Exaggerated systemic antibody response to mucosal Helicobacter pylori infection in IgA nephropathy. Am J Kidney Dis 33:1049–1057
Suzuki H, Kiryluk K, Novak J, Moldoveanu Z, Herr AB, Renfrow MB, Wyatt RJ, Scolari F, Mestecky J, Gharavi AG, Julian BA (2011) The pathophysiology of IgA nephropathy. J Am Soc Nephrol 22:1795–1803
Yanagawa H, Suzuki H, Suzuki Y, Kiryluk K, Gharavi AG, Matsuoka K, Makita Y, Julian BA, Novak J, Tomino Y (2014) A panel of serum biomarkers differentiates IgA nephropathy from other renal diseases. PLoS One 9:e98081
Coppo R (2016) Biomarkers and targeted new therapies for IgA nephropathy. Pediatr Nephrol. doi:10.1007/s00467-016-3390-9
Tomino Y, Sakai H, Endoh M, Suga T, Miura M, Kaneshige H, Nomoto Y (1983) Cross-reactivity of IgA antibodies between renal mesangial areas and nuclei of tonsillar cells in patients with IgA nephropathy. Clin Exp Immunol 51:605–610
Tomino Y (2016) Diagnosis and treatment of patients with IgA nephropathy in Japan. Kidney Res Clin Pract 35:197–203
Xie Y, Nishi S, Ueno M, Imai N, Sakatsume M, Narita I, Suzuki Y, Akazawa K, Shimada H, Arakawa M, Gejyo F (2003) The efficacy of tonsillectomy on long-term renal survival in patients with IgA nephropathy. Kidney Int 63:1861–1867
Zand L, Fervenza FC (2014) Does tonsillectomy have a role in the treatment of patients with immunoglobulin A nephropathy? Nephrol Dial Transplant 29:1456–1459
Kawamura T, Yoshimura M, Miyazaki Y, Okamoto H, Kimura K, Hirano K, Matsushima M, Utsunomiya Y, Ogura M, Yokoo T, Okonogi H, Ishii T, Hamaguchi A, Ueda H, Furusu A, Horikoshi S, Suzuki Y, Shibata T, Yasuda T, Shirai S, Imasawa T, Kanozawa K, Wada A, Yamaji I, Miura N, Imai H, Kasai K, Soma J, Fujimoto S, Matsuo S, Tomino Y, Special IgA Nephropathy Study Group (2014) A multicenter randomized controlled trial of tonsillectomy combined with steroid pulse therapy in patients with immunoglobulin A nephropathy. Nephrol Dial Transplant 29:1546–1553
Feehally J, Coppo R, Troyanov S, Bellur SS, Cattran D, Cook T, Roberts IS, Verhave JC, Camilla R, Vergano L, Egido J, Wiecek A, Karkoszka H, Tesar V, Maixnerova D, Ots-Rosenberg M, Quaglia M, Rollino C, Magistroni R, Cusinato S, Cravero R, Peruzzi L, Lundberg S, Gesualdo L, Cancarini G, Feriozzi S, Ferrario F, VALIGA study of ERA-EDTA Immunonephrology Working Group (2016) Tonsillectomy in a European cohort of 1,147 patients with IgA nephropathy. Nephron 132:15–24
Vergano L, Loiacono E, Albera R, Coppo R, Camilla R, Peruzzi L, Amore A, Donadio ME, Chiale F, Boido A, Mariano F, Mazzucco G, Ravera S, Cancarini G, Magistroni R, Beltrame G, Rollino C, Stratta P, Quaglia M, Bergia R, Cravero R, Cusinato S, Benozzi L, Savoldi S, Licata C (2015) Can tonsillectomy modify the innate and adaptive immunity pathways involved in IgA nephropathy? J Nephrol 28:51–58
Rollino C, Vischini G, Coppo R (2016) IgA nephropathy and infections. J Nephrol 29:463–468
Watanabe H, Goto S, Mori H, Higashi K, Hosomichi K, Aizawa N, Takahashi N, Tsuchida M, Suzuki Y, Yamada T, Horii A, Inoue I, Kurokawa K, Narita I (2016) Comprehensive microbiome analysis of tonsillar crypts in IgA nephropathy. Nephrol Dial Transplant. doi:10.1093/ndt/gfw343
Muto M, Manfroi B, Suzuki H, Joh K, Nagai M, Wakai S, Righini C, Maiguma M, Izui S, Tomino Y, Huard B, Suzuki Y (2016) Toll-like receptor 9 stimulation induces aberrant expression of a proliferation-inducing ligand by tonsillar germinal center B cells in IgA nephropathy. J Am Soc Nephrol. doi:10.1681/ASN.2016050496
Amore A, Coppo R, Nedrud JG, Sigmund N, Lamm ME, Emancipator SN (2004) The role of nasal tolerance in a model of IgA nephropathy induced in mice by Sendai virus. Clin Immunol 113:101–108
Gesualdo L, Lamm ME, Emancipator SN (1990) Defective oral tolerance promotes nephritogenesis in experimental IgA nephropathy induced by oral immunization. J Immunol 145:3684–3691
Wang J, Anders RA, Wu Q, Peng D, Cho JH, Sun Y, Karaliukas R, Kang HS, Turner JR, Fu YX (2004) Dysregulated LIGHT expression on T cells mediates intestinal inflammation and contributes to IgA nephropathy. J Clin Invest 113:826–835
Kiryluk K, Li Y, Scolari F, Sanna-Cherchi S, Choi M, Verbitsky M, Fasel D, Lata S, Prakash S, Shapiro S, Fischman C, Snyder HJ, Appel G, Izzi C, Viola BF, Dallera N, Del Vecchio L, Barlassina C, Salvi E, Bertinetto FE, Amoroso A, Savoldi S, Rocchietti M, Amore A, Peruzzi L, Coppo R, Salvadori M, Ravani P, Magistroni R, Ghiggeri GM, Caridi G, Bodria M, Lugani F, Allegri L, Delsante M, Maiorana M, Magnano A, Frasca G, Boer E, Boscutti G, Ponticelli C, Mignani R, Marcantoni C, Di Landro D, Santoro D, Pani A, Polci R, Feriozzi S, Chicca S, Galliani M, Gigante M, Gesualdo L, Zamboli P, Battaglia GG, Garozzo M, Maixnerová D, Tesar V, Eitner F, Rauen T, Floege J, Kovacs T, Nagy J, Mucha K, Pączek L, Zaniew M, Mizerska-Wasiak M, Roszkowska-Blaim M, Pawlaczyk K, Gale D, Barratt J, Thibaudin L, Berthoux F, Canaud G, Boland A, Metzger M, Panzer U, Suzuki H, Goto S, Narita I, Caliskan Y, Xie J, Hou P, Chen N, Zhang H, Wyatt RJ, Novak J, Julian BA, Feehally J, Stengel B, Cusi D, Lifton RP, Gharavi AG (2014) Discovery of new risk loci for IgA nephropathy implicates genes involved in immunity against intestinal pathogens. Nat Genet 46:1187–1196
Shreiner AB, Kao JY, Young VB (2015) The gut microbiome in health and in disease. Curr Opin Gastroenterol 31:69–75
Forbes JD, Van Domselaar G, Bernstein CN (2016) The gut microbiota in immune-mediated inflammatory diseases. Front Microbiol 7:1081
Cuervo A, Hevia A, López P, Suárez A, Sánchez B, Margolles A, González S (2015) Association of polyphenols from oranges and apples with specific intestinal microorganisms in systemic lupus erythematosus patients. Nutrients 7:1301–1317
Evenepoel P, Poesen R, Meijers B (2016) The gut–kidney axis. Pediatr Nephrol. doi:10.1007/s00467-016-3527-x
Kamata T, Nogaki F, Fagarasan S, Sakiyama T, Kobayashi I, Miyawaki S, Ikuta K, Muso E, Yoshida H, Sasayama S, Honjo T (2000) Increased frequency of surface IgA-positive plasma cells in the intestinal lamina propria and decreased IgA excretion in hyper IgA [HIGA] mice, a murine model of IgA nephropathy with hyperserum IgA. J Immunol 165:1387–1394
Fagarasan S, Kawamoto S, Kanagawa O, Suzuki K (2010) Adaptive immune regulation in the gut: T cell-dependent and T cell-independent IgA synthesis. Annu Rev Immunol 28:243–273
McCarthy DD, Kujawa J, Wilson C, Papandile A, Poreci U, Porfilio EA, Ward L, Lawson MA, Macpherson AJ, McCoy KD, Pei Y, Novak L, Lee JY, Julian BA, Novak J, Ranger A, Gommerman JL, Browning JL (2011) Mice overexpressing BAFF develop a commensal flora-dependent, IgA-associated nephropathy. J Clin Invest 121:3991–4002
Xin G, Shi W, Xu LX, Su Y, Yan LJ, Li KS (2013) Serum BAFF is elevated in patients with IgA nephropathy and associated with clinical and histopathological features. J Nephrol 26:683–690
De Angelis M, Montemurno E, Piccolo M, Vannini L, Lauriero G, Maranzano V, Gozzi G, Serrazanetti D, Dalfino G, Gobbetti M, Gesualdo L (2014) Microbiota and metabolome associated with immunoglobulin A nephropathy (IgAN). PLoS One 9:e99006
Yiu JH, Dorweiler B, Woo CW (2017) Interaction between gut microbiota and toll-like receptor: from immunity to metabolism. J Mol Med 95(1):13–20
Mazmanian SK, Liu CH, Tzianabos AO, Kasper DL (2005) An immunomodulatory molecule of symbiotic bacteria directs maturation of the host immune system. Cell 122:107–118
Anders HJ, Andersen K, Stecher B (2013) The intestinal microbiota, a leaky gut, and abnormal immunity in kidney disease. Kidney Int 83:1010–1019
Davin JC, Forget P, Mahieu PR (1988) Increased intestinal permeability to [51 Cr] EDTA is correlated with IgA immune complex-plasma levels in children with IgA-associated nephropathies. Acta Paediatr Scand 77:118–124
Rostoker G, Wirquin V, Terzidis H, Chaumette MT, Lang P, Weil B (1993) Mucosal immunity in primary glomerulonephritis. III Study of intestinal permeability. Nephron 63:286–290
Qin W, Zhong X, Fan JM, Zhang YJ, Liu XR, Ma XY (2008) External suppression causes the low expression of the Cosmc gene in IgA nephropathy. Nephrol Dial Transplant 23:1608–1614
Yoon HJ, Shin JH, Yang SH, Chae DW, Kim H, Lee DS, Kim HL, Kim S, Lee JS, Kim YS (2003) Association of the CD14 gene -159C polymorphism with progression of IgA nephropathy. J Med Genet 40:104–108
Coppo R, Camilla R, Amore A, Peruzzi L, Daprà V, Loiacono E, Vatrano S, Rollino C, Sepe V, Rampino T, Dal Canton A (2010) Toll-like receptor 4 expression is increased in circulating mononuclear cells of patients with immunoglobulin A nephropathy. Clin Exp Immunol 159:73–81
Donadio ME, Loiacono E, Peruzzi L, Amore A, Camilla R, Chiale F, Vergano L, Boido A, Conrieri M, Bianciotto M, Bosetti FM, Coppo R (2014) Toll-like receptors, immunoproteasome and regulatory T cells in children with Henoch–Schönlein purpura and primary IgA nephropathy. Pediatr Nephrol 29:1545–1551
Emancipator SN, Gallo GR, Lamm ME (1983) Experimental IgA nephropathy induced by oral immunization. J Exp Med 157:572–582
Nagy J, Scott H, Brandtzaeg P (1988) Antibodies to dietary antigens in IgA nephropathy. Clin Nephrol 29:275–279
Coppo R, Amore A, Roccatello D, Gianoglio B, Molino A, Piccoli G, Clarkson AR, Woodroffe AJ, Sakai H, Tomino Y (1991) IgA antibodies to dietary antigens and lectin-binding IgA in sera from Italian, Australian, and Japanese IgA nephropathy patients. Am J Kidney Dis 17:480–487
Helin H, Mustonen J, Reunala T, Pasternack A (1983) IgA nephropathy associated with celiac disease and dermatitis herpetiformis. Arch Pathol Lab Med 107:324–327
Coppo R, Mazzucco G, Martina G, Roccatello D, Amore A, Novara R, Bargoni A, Piccoli G, Sena LM (1989) Gluten-induced experimental IgA glomerulopathy. Lab Investig 60:499–506
Amore A, Emancipator SN, Roccatello D, Gianoglio B, Peruzzi L, Porcellini MG, Piccoli G, Coppo R (1994) Functional consequences of the binding of gliadin to cultured rat mesangial cells: bridging immunoglobulin A to cells and modulation of eicosanoid synthesis and altered cytokine production. Am J Kidney Dis 23:290–301
Papista C, Lechner S, Ben Mkaddem S, LeStang MB, Abbad L, Bex-Coudrat J, Pillebout E, Chemouny JM, Jablonski M, Flamant M, Daugas E, Vrtovsnik F, Yiangou M, Berthelot L, Monteiro RC (2015) Gluten exacerbates IgA nephropathy in humanized mice through gliadin-CD89 interaction. Kidney Int 88:276–285
Berthelot L, Papista C, Maciel TT, Biarnes-Pelicot M, Tissandie E, Wang PH, Tamouza H, Jamin A, Bex-Coudrat J, Gestin A, Boumediene A, Arcos-Fajardo M, England P, Pillebout E, Walker F, Daugas E, Vrtosvnik F, Flamant M, Benhamou M, Cogné M, Moura IC, Monteiro RC (2012) Transglutaminase is essential for IgA nephropathy development acting through IgA receptors. J Exp Med 209:793–806
Coppo R, Basolo B, Rollino C, Roccatello D, Martina G, Amore A, Piccoli G (1986) Dietary gluten and primary IgA nephropathy. N Engl J Med 315:1167–1168
Coppo R, Basolo B, Rollino C, Roccatello D, Martina G, Amore A, Bongiorno G, Piccoli G (1986) Mediterranean diet and primary IgA nephropathy. Clin Nephrol 26:72–82
Coppo R, Roccatello D, Amore A, Quattrocchio G, Molino A, Gianoglio B, Amoroso A, Bajardi P, Piccoli G (1990) Effects of a gluten-free diet in primary IgA nephropathy. Clin Nephrol 33:72–86
Smerud HK, Fellström B, Hällgren R, Osagie S, Venge P, Kristjánsson G (2009) Gluten sensitivity in patients with IgA nephropathy. Nephrol Dial Transplant 24:2476–2481
Smerud HK, Fellström B, Hällgren R, Hällgren R, Osagie S, Venge P, Kristjánsson G (2010) Gastrointestinal sensitivity to soy and milk proteins in patients with IgA nephropathy. Clin Nephrol 74:364–371
Kovács T, Mette H, Per B, Kun L, Schmelczer M, Barta J, Jean-Claude D, Nagy J (1996) Relationship between intestinal permeability and antibodies against food antigens in IgA nephropathy. Orv Hetil 137:65–69
Rantala I, Collin P, Holm K, Kainulainen H, Mustonen J, Mäki M (1999) Small bowel T cells, HLA class II antigen DR, and GroEL stress protein in IgA nephropathy. Kidney Int 55:2274–2280
Honkanen T, Mustonen J, Kainulainen H, Myllymäki J, Collin P, Hurme M, Rantala I (2005) Small bowel cyclooxygenase 2 (COX-2) expression in patients with IgA nephropathy. Kidney Int 67:2187–2195
Almroth G, Axelsson T, Müssener E, Grodzinsky E, Midhagen G, Olcén P (2006) Increased prevalence of antigliadin IgA-antibodies with aberrant duodenal histopathological findings in patients with IgA-nephropathy and related disorders. Ups J Med Sci 111:339–352
Bazzi C, Sinico RA, Petrini C, Rizza V, Torpia R, Arrigo G, Ragni A, D’Amico G (1992) Low doses of drugs able to alter intestinal mucosal permeability to food antigens [5-aminosalicylic acid and sodium cromoglycate] do not reduce proteinuria in patients with IgA nephropathy: a preliminary noncontrolled trial. Nephron 61:192–195
Sato M, Takayama K, Kojima H, Koshikawa S (1990) Sodium cromoglycate therapy in IgA nephropathy: a preliminary short-term trial. Am J Kidney Dis 15:141–146
Szigeti N, Kovács T, Degrell P, Fábián G, Wittmann I, Nagy J (2007) Secondary IgA nephropathy in gastroenterological diseases. Orv Hetil 148:313–318
Filiopoulos V, Trompouki S, Hadjiyannakos D, Paraskevakou H, Kamperoglou D, Vlassopoulos D (2010) IgA nephropathy in association with Crohn’s disease: a case report and brief review of the literature. Ren Fail 32:523–527
Ambruzs JM, Walker PD, Larsen CP (2014) The histopathologic spectrum of kidney biopsies in patients with inflammatory bowel disease. Clin J Am Soc Nephrol 9:265–270
Koning F (2014) Pathophysiology of celiac disease. J Pediatr Gastroenterol Nutr 59[Suppl 1]:S1–S4
Tjon JM, van Bergen J, Koning F (2010) Celiac disease: how complicated can it get? Immunogenetics 62:641–651
Welander A, Sundelin B, Fored M, Ludvigsson JF (2013) Increased risk of IgA nephropathy among individuals with celiac disease. J Clin Gastroenterol 47:678–683
Collin P, Syrjanen J, Partanen J, Pasternack A, Kaukinen K, Mustonen J (2002) Celiac disease and HLA DQ in patients with IgA nephropathy. Am J Gastroenterol 97:2572–2576
Moeller S, Canetta PA, Taylor AK, Arguelles-Grande C, Snyder H, Green PH, Kiryluk K, Alaedini A (2014) Lack of serologic evidence to link IgA nephropathy with celiac disease or immune reactivity to gluten. PLoS One 9:e94677
Smerud HK, Bárány P, Lindström K, Fernström A, Sandell A, Påhlsson P, Fellström B (2011) New treatment for IgA nephropathy: enteric budesonide targeted to the ileocecal region ameliorates proteinuria. Nephrol Dial Transplant 26:3237–3242
Fellstrom B, Barratt J, Cook H, Coppo R, Feehally J, W de Fijter J, Floege J, Hetzel G, Jardine A, Locatelli F, Maes MD, Mercer A, Ortiz F, Praga M, Sørensen S, Tesar V, Del Vecchio L, for the NEFIGAN Trial Investigators (2017) Targeted-release budesonide versus placebo in patients with IgA nephropathy (NEFIGAN): a double-blind, randomised, placebo-controlled phase 2b trial. Lancet. doi:10.1016/S0140-6736(17)30550-0
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that there is no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by the author.
Statement
The manuscript is not under consideration for publication elsewhere in a similar form in any language.
Additional information
Answers to questions
1) b
2) c
3) c
4) a
5) a
Rights and permissions
About this article
Cite this article
Coppo, R. The gut–kidney axis in IgA nephropathy: role of microbiota and diet on genetic predisposition. Pediatr Nephrol 33, 53–61 (2018). https://doi.org/10.1007/s00467-017-3652-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-017-3652-1