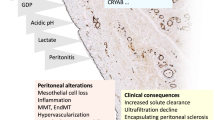
Abstract
Peritoneal dialysis (PD) is a viable treatment option for end stage renal disease (ESRD) patients worldwide. PD may provide a survival advantages over hemodialysis (HD) in the early years of treatment. However, the benefits of PD are short-lived, as peritoneal membrane failure ensues in many patients, owing mainly to structural and functional changes in the peritoneal membrane from the use of conventional bio-incompatible PD solutions, which are hyperosmolar, acidic, have lactate buffer and contain high concentrations of glucose and glucose degradation products (GDPs). Current data suggest that chronic exposure of the peritoneum to contemporary PD fluids provokes activation of various inflammatory, fibrogenic and angiogenic cytokines, interplay of which leads to progressive peritoneal fibrosis, vasculopathy and neoangiogenesis. There is emerging evidence that peritoneal vascular changes are mainly responsible for increased solute transport and ultrafiltration failure in long-term PD. However, the precise pathophysiologic mechanisms initiating and propagating peritoneal fibrosis and angiogenesis remain elusive. The protection of the peritoneal membrane from long-term toxic and metabolic effects of high GDP-containing, conventional, glucose-based solutions is a prime objective to improve PD outcome. Recent development of new, more biocompatible, PD solutions should help to preserve peritoneal membrane function, promote ultrafiltration, improve nutritional status and, hopefully, preserve peritoneal membrane and improve overall PD outcomes. Elucidation of molecular mechanisms involved in the cellular responses leading to peritoneal fibrosis and angiogenesis spurs new therapeutic strategies that might protect the peritoneal membrane against the consequences of longstanding PD.

Similar content being viewed by others
References
Alexander SR, Warady BA (2004) The demographics of dialysis in children. In: Warady BA, Schaefer F, Fine RN, Alexander S (eds) Pediatric dialysis. Kluwer, Dordrecht, The Netherlands, pp 35–45
Vonesh EF, Snyder JJ, Foley RN, Collins AJ (2006) Mortality studies comparing peritoneal dialysis and hemodialysis: what do they tell us? Kidney Int 70 [Suppl 103]:S3–S11
Kawaguchi Y, Hasegawa T, Nakayama M, Kubo H, Shigematu T (1997) Issues affecting the longevity of continuous ambulatory peritoneal dialysis. Kidney Int 52 [Suppl 62]:S105–S107
Schaefer F, Klaus G, Muller-Wiefel DE, Mehls O; Mid European Pediatric Peritoneal Dialysis Study Group (MEPPS) (1999) Current practice of peritoneal dialysis in children: results of a longitudinal survey. Perit Dial Int 19 [Suppl 2]:S445–S449
Andreoli SP, Langefeld CD, Stadler S, Smith P, Stars A, West K (1993) Risks of peritoneal membrane failure in children undergoing long-term peritoneal dialysis. Pediatr Nephrol 7:543–547
Davies S, Phillips L, Griffiths AM, Russell LH, Naish PF, Russell GI (1998) What really happens to people on long-term peritoneal dialysis? Kidney Int 54:2207–2217
Nagy JA (1996) Peritoneal morphology and function. Kidney Int 50 [Suppl 56]:S2–S11
Fischbach M, Haraldsson B, Helms P, Danner S, Laugel V, Terzic J (2003) The peritoneal membrane: a dynamic dialysis membrane in children. Adv Perit Dial 19:265–268
Dobbie JW (1990) New concepts in molecular biology and ultrastructural pathology of the peritoneum: their significance for peritoneal dialysis. Am J Kidney Dis 15:97–109
Williams JD, Craig KJ, Ruhland CV, Topley N, Williams GT (2003) The natural course of peritoneal membrane biology during peritoneal dialysis. Kidney Int 64 [Suppl 88]:S43–S49
Krediet R (1999) The peritoneal membrane in chronic peritoneal dialysis. Kidney Int 55:341–356
Lopez-Cabrera M, Aguilera A, Aroeira LS, Ramirez-Huesca M, Perez-Lozano ML, Jimemez-Heffernan JA, Bajo MA, del Peso G, Sanches-Tomero JA, Selgas R (2006) Ex vivo analysis of dialysis effluent-derived mesothelial cells as an approach to unveiling the mechanism of peritoneal membrane failure. Perit Dial Int 26:26–34
Yanez-Mo M, Lara-Pezzi E, Selgas R, Ramirez-Huesca M, Dominguez-Jimenez C, Jiménez-Heffernan AJ, Aguilera A, Sánchez-Tomero AJ, Bajo MA, Álvarez V, Castro AM, del Peso G, Cirujeda A, Gmallo C, Sanchez-Madrid F, Lopez-Cabrera M (2003) Peritoneal dialysis and epithelial-to-mesenchymal transition of mesothelial cells. N Eng J Med 348:403–413
Davies S, Phillips L, Griffiths AM, Russell LH, Naish PF, Russell GI (1998) What really happens to people on long-term peritoneal dialysis? Kidney Int 54:2207–2217
Wardy BA, Fiyush B, Andreoli S, Kohaut E, Salusky I, Schlichting L, Pu K, Watkins S (1999) Longitudinal evaluation of transport kinetics in children receiving peritoneal dialysis. Pediatr Nephrol 13:571–576
Davies S, Bryan J, Phillips L, Russell GI (1996) Longitudinal changes in peritoneal kinetics: the effect of peritoneal dialysis and peritonitis. Nephrol Dial Transplant 11:498–506
Flessner MF (2005) The transport barrier in intraperitoneal therapy. Am J Physiol Renal Physiol 288:F433–F442
Brimble KS, Walker M, Margetts PJ, Kundhal KK, Rabbat CG (2006) Meta-analysis: peritoneal membrane transport, mortality and technique failure in peritoneal dialysis. J Am Soc Nephrol 17:2591–2598
Wang T, Hrimburger O, Waniewski J, Bergstrom J, Lindholm B (1998) Increased peritoneal permeability is associated with decreased fluid and small solute removal and higher mortality in CAPD patients. Nephrol Dial Transplant 13:1242–1249
Churchill DN, Thorpe KE, Nolph KD, Keshaviah PR, Oreopoulos DG, Page D (1998) Increased peritoneal membrane transport is associated with decreased patient and technique survival for continuous peritoneal dialysis patients. J Am Soc Nephrol 9:1285–1292
Margetts PJ, Oh K-H, Kulb M (2004) Transforming growth factor β. Importance in long-term peritoneal membrane changes. Perit Dial Int 25 [Suppl 3]:S15–S17
Margetts PJ, Bonniaud P (2003) Basic mechanisms and clinical implications of peritoneal fibrosis. Perit Dial Int 23:530–541
Combet S, Miyata T, Moulin P, Pouthier D, Goffin R, Devuyst O (2000) Vascular proliferation and enhanced expression of endothelial nitric oxide synthase in human peritoneum exposed to long-term peritoneal dialysis. J Am Soc Nephrol 11:717–728
Zweers MM, de Waart DR, Smit W, Struijk DG, Krediet RT (1999) Growth factors VEGF and TGF-beta1 in peritoneal dialysis. J Lab Clin Med 134:124–132
Noh H, Ha H, Yu MR, Kim YO, Kim HJ, Lee HB (2005) Angiotensin II mediated high glucose induced TGF β1 and fibronectin upregulation in HPMC through reactive oxygen species. Perit Dial Int 25:38–47
Rizkalla B, Forbes JM, Cooper ME, Cao Z (2002) Increased renal vascular endothelial growth factor and angiopoetins by angiotensin II infusion is mediated by both AT1 and AT2 receptors. J Am Soc Nephrol 14:3061–3071
Sauter M, Cohen CD, Wornle M, Mussack T, Ladurner R, Sitter T (2007) ACE inhibitor and AT-1 receptor blocker attenuate the production of VEGF in mesothelial cells. Perit Dial Int 27:167–172
Fritsch S, Lindner V, Welsch S, Massfelder T, Grima M, Rothhut S, Barthelmebs M, Helwig J-J (2004) Intravenous delivery of PTH/PTHrP type 1 receptor cDNA to rats decreases heart rate, blood pressure, renal tone, renin angiotensin system, and stress-induced cardiovascular responses. J Am Soc Nephrol 13:2588–2600
Clemens TL, Cormier S, Eichinger A, Endlich K, Fiaschi-Taesch N, Fischer E, Friedman PA, Karaplis AC, Massfelder T, Rossert J, Schlitter K-D, Silve C, Stewart AF, Takane K, Helwig J-J (2001) Parathyroid hormone related protein and its receptors: nuclear functions and roles in the renal and cardiovascular systems, the placental trophoblasts and the pancreatic islets. Br J Pharmacol 134:1113–1136
Schreck R, Rieber P, Baeuerle PA (1991) Reactive oxygen intermediates as apparently widely used messengers in the activation of the NF-kB transcription factor and HIV-1. EMBO J 10:2247–2258
Alhamdani MS (2005) Impairment of glutathione biosynthetic pathway in uremia and dialysis. Nephrol Dial Transplant 20:124–128, 2005
Fabbrini P, Zareie M, ter Wee PM, Keuning ED, Beelen RHJ, van den Born J (2006) Peritoneal exposure model in the rat as a tool to unravel bio(in)compatibility of PDF. Nephrol Dial Transplant 21 [Suppl 2]:8–11
Cueto-Manzano AM, Rojas-Campos E, Martinez-Ramirez HR, Valera-Gonzalez I, Medina M, Montenon F, Ruiz N, Becerra M, Palomeque MA, Cortes-Sanabria L (2005) Can the inflammation markers of patients with high peritoneal permeability on continuous ambulatory peritoneal dialysis be reduced on nocturnal intermittent peritoneal dialysis? Perit Dial Int 26:341–348
Garosi G, Paolo ND (2000) Peritoneal sclerosis: one or two nosological entities? Semin Dial 13:297–308
Mactier RA, Sprosen TS, Gokal R, Williams PF, Lindbergh M, Naik RB, Wrege U, Gröntoft K-C, Larsson R, Berglund J, Tranæus AP, Faict D (1998) Bicarbonate and bicarbonate/lactate peritoneal dialysis solutions for the treatment of infusion pain. Kidney Int 53:1061–1067
Topley N (1997) In vitro biocompatibility of bicarbonate-based peritoneal dialysis solutions. Perit Dial Int 17:42–47
Wong TY, Philips AO, Witowski J, Topley N (2003) Glucose mediated induction of TGF β1 and MCP-1 in mesothelial cells in vitro is osmolality and polyol pathway dependent. Kidney Int 63:1404–1416
De Vriese AS, Mortier S, Lameire NH (2001) What happens to the peritoneal membrane in long-term peritoneal dialysis? Perit Dial Int 21 [Suppl 3]:S9–S18
Vardhan A, Zweers MM, Gokal R, Kreidet RT (2003) A solutions portfolio approach in peritoneal dialysis. Kidney Int 64 [Suppl 88]:S114–S123
Witowski J, Wisniewska J, Korybalska K, Bender TO, Breborowicz A, Gahl GM, Frei U, Passlick-Deetjen J, Jorres A (2001) Prolonged exposure to glucose degradation products impairs viability and function of human peritoneal mesothelial cells. J Am Soc Nephrol 12:2434–2441
Linden T, Cohen A, Deppisch R, Kjellstrand P, Wieslander A (2002) 3,4-Dideoxyglucosone-3-ene (3,4-DGE): a cytotoxic glucose degradation product in fluids for peritoneal dialysis. Kidney Int 62:697–703
Miyata T, Devuyst O, Kurokawa K, van Ypersele de Strihou C (2002) Towards better dialysis compatibility: advances in the biochemistry and pathophysiology of the peritoneal membranes. Kidney Int 61:375–386
Nakamura S, Tachikawa T, Tobita K, Miyazaki S, Sakai S, Morita T, Hirasawa Y, Weigle B, Pischetsrieder M, Niwa T (2003) Role of advanced glycation end products and growth factors in peritoneal dysfunction. Am J Kidney Dis 41 [Suppl 1]:S61–S67
Nishida Y, Shao JC, Kiribayashi K, Nakamura C, Yorioka N (1998) Advanced glycation end products reduce the viability of human peritoneal mesothelial cells. Nephron 80:477–478
Boulanger E, Wautier MP, Wutier JL, Boval B, Panis Y, Wernert N, Danze P-M, Dequiedt P (2002) AGEs bind to mesothelial cells via RAGE and stimulate VCAM-1 expression. Kidney Int 61:148–156
de Boer AW, Schroder CH, van Vliet R, Willems JL, Monnens LA (2000) Clinical experience with icodextrin in children: ultrafiltration profiles and metabolism. Pediatr Nephrol 15:21–24
Michallat AC, Dheu C, Loichot C, Danner S, Fischbach M (2005) Long daytime exchange in children on continuous cycling peritoneal dialysis: preservation of drained volume because of icodextrin use. Adv Perit Dial 21:195–199
Dart A, Feber J, Wong H, Filler G (2005) Icodextrin re-absorption varies with age in children on automated peritoneal dialysis. Pediatr Nephrol 20:683–685
Wolfson M, Ogrinc F, Mujais S (2002) Review of clinical trial experience with icodextrin. Kidney Int 62 [Suppl 81]:S46–S52
Ha H, Cha MK, Choi HN, Lee HB (2002) Effects of peritoneal dialysis solutions on the secretion of growth factors and extracellular matrix proteins by human peritoneal mesothelial cells. Perit Dial Int 22:171–177
Davies SJ, Brown EA, Frandsen NE, Rodrigues AS, Rodriguez-Carmona A, Vychytil A, Macnamara E, Ekstrand A, Tranaeus A, Divino JC (2005) Longitudinal membrane function in functionally anuric patients treated with APD: data from EAPOS on the effects of glucose and icodextrin prescription. Kidney Int 67:1609–1615
Canepa A, Perfumo F, Carrea A, Menoni S, Verrina E, Trivelli A, Delucchi P, Gusmano R (1996) Protein and calorie intake, nitrogen losses and nitrogen balance in children undergoing chronic peritoneal dialysis. Adv Perit Dial 12:326–329
Young GA, Kopple JD, Lindholm B, Vonesh EF, De Vecchi A, Scalamogna A, Castelnova C, Oreopoulos DG, Anderson GH, Bergstrom J (1991) Nutritional assessment of continuous ambulatory peritoneal dialysis. An international study. Am J Kidney Dis 17:462–471
Canepa A, Verrina E, Perfumo F, Carrea A, Menoni S, Delucchi P, Gusmano R (1999) Value of intraperitoneal amino acids in children treated with chronic peritoneal dialysis. Perit Dial Int 19 [Suppl 2]:S435–S440
Park MS, Choi SR, Song YS, Yoon SY, Lee SY, Han DS (2006) New insight of amino-acid based dialysis solutions. Kidney Int 70 [Suppl 103]:S110–S114
Zareie M, van Lambalgen AA, ter Wee PM, Hekking LH, Kauning ED, Schadee-Eesrermans IL, Faict D, Degreve B, Tangelder GJ, Beelen RHJ, van den Born J (2005) Better preservation of the peritoneum in rats exposed to amino acid-based peritoneal dialysis fluid. Perit Dial Int 25:58–67
Van Biesen W, Boer W, De Greeve B, Dequidt C, Vijt D, Faict D, Lameire N (2004) A randomized clinical trial with a 0.6% amino acid/1.4% glycerol peritoneal dialysis solution. Perit Dial Int 24:222–230
Erixon M, Wieslander A, Linden T, Carlsson O, Forshback G, Svensson E, Jonsson JA, Kjellstrand P (2006) How to avoid glucose degradation products in peritoneal dialysis fluids. Perit Dial Int 26:490–497
Hoff CM (2003) In vitro biocompatibility performance of Physioneal. Kidney Int 64 [Suppl 88]:S57–S74
Pecoits-Filho R, Tranaeus A, Lindholm B (2003) Clinical trial experiences with Physioneal. Kidney Int 64 [Suppl 88]:S100–S104
Williams J, Topley N, Craig K, Mackenzie R, Pischertsrieder M, Lage C, Passilick-Deetjen J (2004) The Euro-Balance Trial. The effect of a new biocompatible peritoneal dialysis fluid (balance) on the peritoneal membrane. Kidney Int 66:408–418
Lee H, Park H, Seo B, Do J, Yun S, Song H, Kim Y, Kim Y, Kim D, Kim Y, Ahn C, Kim M, Shin S (2005) Superior patient survival for continuous ambulatory peritoneal dialysis patients treated with a peritoneal dialysis fluid with neutral pH and low glucose degradation products concentration (balance). Perit Dial Int 25:248–255
Lee H, Choi H, Park H, Seo B, Do J, Yun S, Song H, Kim Y, Kim Y, Kim D, Kim Y, Kim M, Shin S (2006) Changing prescribing practice in CAPD patients in Korea. Increased utilization of low GDP solutions improves patient outcome. Nephrol Dial Transplant 21:2893–2899
Montenegro J, Saracho R, Gallardo I, Martinez I, Munoz R, Quintanilla N (2007) Use of pure bicarbonate-buffered peritoneal dialysis fluid reduces the incidence of CAPD peritonitis. Nephrol Dial Transplant 22:1703–1708
Schmitt CP, von Heyl D, Rieger S, Arbeiter K, Bonzel KE, Fischbach M, Misselwitz J, Pieper A-K, Schaefer F; Mid European Pediatric Peritoneal Dialysis Study Group (MEPPS) (2007) Reduced systemic advanced glycation endproducts in children receiving peritoneal dialysis with low glucose degradation product content. Nephrol Dial Transplant 22:2038–2044
Fischbach M, Terzic J, Chauve S, Laugel V, Muller A, Haraldsson B (2004) Effect of peritoneal dialysis fluid composition on peritoneal area available for exchange in children. Nephrol Dial Transplant 19:925–932
Haas S, Schmitt CP, Arbeiter K, Bonzel K-E, Fischbach M, John U, Pieper A-K, Schaub TP, Passlick-Deetjen J, Mehls O, Schaefer F; Mid European Pediatric Peritoneal Dialysis Study Group (2003) Improved acidosis correction and recovery of mesothelial cell mass with neutral pH bicarbonate dialysis solution among children undergoing automated peritoneal dialysis. J Am Soc Nephrol 14:2632–2638
le Poole CY, van Ittersum FJ, Weijmer MC, Valenttijn RM, ter Wee PM (2004) Clinical effects of a peritoneal dialysis regimen low in glucose in new peritoneal dialysis patients: a randomized crossover study. Adv Perit Dial 20:170–176
le Poole CY, Welten AG, Weijmer MC, Valenttijn RM, van Ittersum FJ, ter Wee PM (2005) Initiating CAPD with a regimen low in glucose and glucose degradation products, with icodextrin and amino acids (NEPP) is safe and efficacious. Perit Dial Int 25 [Suppl 3]:S64–S68
Miyata T, Kurokawa K, van Ypersele De Strihou C (2000) Advanced glycation and lipidoxidation end products: role of reactive carbonyl compounds generated during carbohydrate and lipid metabolism. J Am Soc Nephrol 11:1744–1752
Hobbs AJ, Higgs A, Noncada S (1999) Inhibition of nitric oxide synthase as a potential therapeutic target. Ann Rev Pharmacol Toxicol 39:191–220
De Vriese AS, Tilton RG, Seephan CC, Lameire N (2001) Diabetes-induced microvascular proliferation and hyperpermeability in the peritoneum: role of vascular endothelial growth factor. J Am Soc Nephrol 12:1734–1741
Miyazaki M, Obata Y, Abe K, Furusu A, Koji T, Tabata Y, Kohno S (2006) Technological advances in peritoneal dialysis. Gene transfer using nonviral delivery system. Perit Dial Int 26:633–640
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saxena, R. Pathogenesis and treatment of peritoneal membrane failure. Pediatr Nephrol 23, 695–703 (2008). https://doi.org/10.1007/s00467-007-0580-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-007-0580-5