Abstract
Chloride channels are expressed along the entire mammalian nephron. They participate in transepithelial chloride transport, cell volume regulation and acidification of intracellular vesicles. Some chloride channels are constitutively active and others are regulated by either second messengers such as cAMP or Ca++ or secondary to changes in membrane potential. The molecular identities of a number of chloride channels within the kidney are still unknown. Abnormalities in chloride channel expression and function in the kidney can cause a range of disorders such as autosomal recessive Dent’s disease, Bartter’s syndrome, renal tubular acidosis and diabetes insipidus. The purpose of this review is to give an overview of the chloride channels in the kidney and to focus on the function of renal chloride channels as revealed by diseases associated with channel dysfunction.



Similar content being viewed by others
References
Jentsch TJ, Steinmeyer K, Schwarz G (1990) Primary structure of Torpedo marmorata chloride channel isolated by expression cloning in Xenopus oocytes. Nature 348:510–514
Fahlke C, Knittle T, Gurnett CA, Campbell KP, George AL Jr (1997) Subunit stoichiometry of human muscle chloride channels. J Gen Physiol 109:93–104
Schmitt R, Kahl T, Mutig K, Bachmann S (2004) Selectively reduced expression of thick ascending limb Tamm-Horsfall protein in hypothyroid kidneys. Histochem Cell Biol 121:319–327
Saviane C, Conti F, Pusch M (1999) The muscle chloride channel ClC-1 has a double-barreled appearance that is differentially affected in dominant and recessive myotonia. J Gen Physiol 113:457–468
Weinreich F, Jentsch TJ (2001) Pores formed by single subunits in mixed dimers of different CLC chloride channels. J Biol Chem 276:2347–2353
Jentsch TJ, Stein V, Weinreich F, Zdebik AA (2002) Molecular structure and physiological function of chloride channels. Physiol Rev 82:503–568
Ludewig U, Pusch M, Jentsch TJ (1996) Two physically distinct pores in the dimeric ClC-0 chloride channel. Nature 383:340–343
Ludewig U, Pusch M, Jentsch TJ (1997) Independent gating of single pores in CLC-0 chloride channels. Biophys J 73:789–797
Miller C (1982) Open-state substructure of single chloride channels from Torpedo electroplax. Philos Trans R Soc Lond B Biol Sci 299:401–411
Dutzler R, Campbell EB, Cadene M, Chait BT, MacKinnon R (2002) X-ray structure of a ClC chloride channel at 3.0 A reveals the molecular basis of anion selectivity. Nature 415:287–294
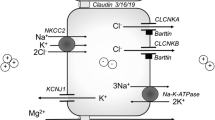
Estevez R, Boettger T, Stein V, Birkenhager R, Otto E, Hildebrandt F, Jentsch TJ (2001) Barttin is a Cl- channel beta-subunit crucial for renal Cl- reabsorption and inner ear K+ secretion. Nature 414:558–561
Liantonio A, Pusch M, Picollo A, Guida P, De Luca A, Pierno S, Fracchiolla G, Loiodice F, Tortorella P, Conte CD (2004) Investigations of pharmacologic properties of the renal CLC-K1 chloride channel co-expressed with barttin by the use of 2-(p-Chlorophenoxy)propionic acid derivatives and other structurally unrelated chloride channels blockers. J Am Soc Nephrol 15:13–20
Waldegger S, Jeck N, Barth P, Peters M, Vitzthum H, Wolf K, Kurtz A, Konrad M, Seyberth HW (2002) Barttin increases surface expression and changes current properties of ClC-K channels. Pflugers Arch 444:411–418
Dutzler R, Campbell EB, MacKinnon R (2003) Gating the selectivity filter in ClC chloride channels. Science 300:108–112
Niemeyer MI, Cid LP, Zuniga L, Catalan M, Sepulveda FV (2003) A conserved pore-lining glutamate as a voltage- and chloride-dependent gate in the ClC-2 chloride channel. J Physiol 553:873–879
Obermuller N, Gretz N, Kriz W, Reilly RF, Witzgall R (1998) The swelling-activated chloride channel ClC-2, the chloride channel ClC-3, and ClC-5, a chloride channel mutated in kidney stone disease, are expressed in distinct subpopulations of renal epithelial cells. J Clin Invest 101:635–642
Furukawa T, Ogura T, Katayama Y, Hiraoka M (1998) Characteristics of rabbit ClC-2 current expressed in Xenopus oocytes and its contribution to volume regulation. Am J Physiol 274:C500–C512
Huber SM, Duranton C, Henke G, Van De SC, Heussler V, Shumilina E, Sandu CD, Tanneur V, Brand V, Kasinathan RS, Lang KS, Kremsner PG, Hubner CA, Rust MB, Dedek K, Jentsch TJ, Lang F (2004) Plasmodium induces swelling-activated ClC-2 anion channels in the host erythrocyte. J Biol Chem 279:41444–41452
Bosl MR, Stein V, Hubner C, Zdebik AA, Jordt SE, Mukhopadhyay AK, Davidoff MS, Holstein AF, Jentsch TJ (2001) Male germ cells and photoreceptors, both dependent on close cell-cell interactions, degenerate upon ClC-2 Cl(-) channel disruption. EMBO J 20:1289–1299
Yoshikawa M, Uchida S, Ezaki J, Rai T, Hayama A, Kobayashi K, Kida Y, Noda M, Koike M, Uchiyama Y, Marumo F, Kominami E, Sasaki S (2002) CLC-3 deficiency leads to phenotypes similar to human neuronal ceroid lipofuscinosis. Genes Cells 7:597–605
Kornak U, Kasper D, Bosl MR, Kaiser E, Schweizer M, Schulz A, Friedrich W, Delling G, Jentsch TJ (2001) Loss of the ClC-7 chloride channel leads to osteopetrosis in mice and man. Cell 104:205–215
Knohl SJ, Scheinman SJ (2004) Inherited hypercalciuric syndromes: Dent’s disease (CLC-5) and familial hypomagnesemia with hypercalciuria (paracellin-1). Semin Nephrol 24:55–60
Carballo-Trujillo I, Garcia-Nieto V, Moya-Angeler FJ, Anton-Gamero M, Loris C, Mendez-Alvarez S, Claverie-Martin F (2003) Novel truncating mutations in the ClC-5 chloride channel gene in patients with Dent’s disease. Nephrol Dial Transplant 18:717–723
Claverie-Martin F, Gonzalez-Acosta H, Flores C, Anton-Gamero M, Garcia-Nieto V (2003) De novo insertion of an Alu sequence in the coding region of the CLCN5 gene results in Dent’s disease. Hum Genet 113:480–485
Devuyst O, Christie PT, Courtoy PJ, Beauwens R, Thakker RV (1999) Intra-renal and subcellular distribution of the human chloride channel, CLC-5, reveals a pathophysiological basis for Dent’s disease. Hum Mol Genet 8:247–257
Piwon N, Gunther W, Schwake M, Bosl MR, Jentsch TJ (2000) ClC-5 Cl- -channel disruption impairs endocytosis in a mouse model for Dent’s disease. Nature 408:369–373
Wang SS, Devuyst O, Courtoy PJ, Wang XT, Wang H, Wang Y, Thakker RV, Guggino S, Guggino WB (2000) Mice lacking renal chloride channel, CLC-5, are a model for Dent’s disease, a nephrolithiasis disorder associated with defective receptor-mediated endocytosis. Hum Mol Genet 9:2937–2945
Christensen EI, Devuyst O, Dom G, Nielsen R, Van der SP, Verroust P, Leruth M, Guggino WB, Courtoy PJ (2003) Loss of chloride channel ClC-5 impairs endocytosis by defective trafficking of megalin and cubilin in kidney proximal tubules. Proc Natl Acad Sci USA 100:8472–8477
Steinmeyer K, Schwappach B, Bens M, Vandewalle A, Jentsch TJ (1995) Cloning and functional expression of rat CLC-5, a chloride channel related to kidney disease. J Biol Chem 270:31172–31177
Moulin P, Igarashi T, Van der SP, Cosyns JP, Verroust P, Thakker RV, Scheinman SJ, Courtoy PJ, Devuyst O (2003) Altered polarity and expression of H+-ATPase without ultrastructural changes in kidneys of Dent’s disease patients. Kidney Int 63:1285–1295
Carr G, Simmons NL, Sayer JA (2006) Disruption of clc-5 leads to a redistribution of annexin A2 and promotes calcium crystal agglomeration in collecting duct epithelial cells. Cell Mol Life Sci 63:367–377
Hryciw DH, Ekberg J, Lee A, Lensink IL, Kumar S, Guggino WB, Cook DI, Pollock CA, Poronnik P (2004) Nedd4-2 functionally interacts with ClC-5: involvement in constitutive albumin endocytosis in proximal tubule cells. J Biol Chem 279:54996–55007
Adachi S, Uchida S, Ito H, Hata M, Hiroe M, Marumo F, Sasaki S (1994) Two isoforms of a chloride channel predominantly expressed in thick ascending limb of Henle’s loop and collecting ducts of rat kidney. J Biol Chem 269:17677–17683
Uchida S, Sasaki S, Nitta K, Uchida K, Horita S, Nihei H, Marumo F (1995) Localization and functional characterization of rat kidney-specific chloride channel, ClC-K1. J Clin Invest 95:104–113
Kieferle S, Fong P, Bens M, Vandewalle A, Jentsch TJ (1994) Two highly homologous members of the ClC chloride channel family in both rat and human kidney. Proc Natl Acad Sci USA 91:6943–6947
Wolf K, Meier-Meitinger M, Bergler T, Castrop H, Vitzthum H, Riegger GA, Kurtz A, Kramer BK (2003) Parallel down-regulation of chloride channel CLC-K1 and barttin mRNA in the thin ascending limb of the rat nephron by furosemide. Pflugers Arch 446:665–671
Kobayashi K, Uchida S, Okamura HO, Marumo F, Sasaki S (2002) Human CLC-KB gene promoter drives the EGFP expression in the specific distal nephron segments and inner ear. J Am Soc Nephrol 13:1992–1998
Castrop H, Kramer BK, Riegger GA, Kurtz A, Wolf K (2000) Overexpression of chloride channel CLC-K2 mRNA in the renal medulla of Dahl salt-sensitive rats. J Hypertens 18:1289–1295
Vitzthum H, Castrop H, Meier-Meitinger M, Riegger GA, Kurtz A, Kramer BK, Wolf K (2002) Nephron specific regulation of chloride channel CLC-K2 mRNA in the rat. Kidney Int 61:547–554
Vandewalle A, Cluzeaud F, Bens M, Kieferle S, Steinmeyer K, Jentsch TJ (1997) Localization and induction by dehydration of ClC-K chloride channels in the rat kidney. Am J Physiol 272:F678–F688
Akizuki N, Uchida S, Sasaki S, Marumo F (2001) Impaired solute accumulation in inner medulla of Clcnk1-/- mice kidney. Am J Physiol Renal Physiol 280:F79–F87
Uchida S, Marumo F (2000) Severely impaired urine-concentrating ability in mice lacking the CLC-K1 chloride channel. Exp Nephrol 8:361–365
Hayama A, Rai T, Sasaki S, Uchida S (2003) Molecular mechanisms of Bartter syndrome caused by mutations in the BSND gene. Histochem Cell Biol 119:485–493
Konrad M, Vollmer M, Lemmink HH, van den Heuvel LP, Jeck N, Vargas-Poussou R, Lakings A, Ruf R, Deschenes G, Antignac C, Guay-Woodford L, Knoers NV, Seyberth HW, Feldmann D, Hildebrandt F (2000) Mutations in the chloride channel gene CLCNKB as a cause of classic Bartter syndrome. J Am Soc Nephrol 11:1449–1459
Jeck N, Waldegger S, Lampert A, Boehmer C, Waldegger P, Lang PA, Wissinger B, Friedrich B, Risler T, Moehle R, Lang UE, Zill P, Bondy B, Schaeffeler E, Asante-Poku S, Seyberth H, Schwab M, Lang F (2004) Activating mutation of the renal epithelial chloride channel ClC-Kb predisposing to hypertension. Hypertension 43:1175–1181
Jeck N, Waldegger P, Doroszewicz J, Seyberth H, Waldegger S (2004) A common sequence variation of the CLCNKB gene strongly activates ClC-Kb chloride channel activity. Kidney Int 65:190–197
Konig J, Schreiber R, Voelcker T, Mall M, Kunzelmann K (2001) The cystic fibrosis transmembrane conductance regulator (CFTR) inhibits ENaC through an increase in the intracellular Cl- concentration. EMBO Rep 2:1047–1051
Kunzelmann K (2003) ENaC is inhibited by an increase in the intracellular Cl(-) concentration mediated through activation of Cl(-) channels. Pflugers Arch 445:504–512
Konstas AA, Koch JP, Tucker SJ, Korbmacher C (2002) Cystic fibrosis transmembrane conductance regulator-dependent up-regulation of Kir1.1 (ROMK) renal K+ channels by the epithelial sodium channel. J Biol Chem 277:25377–25384
Belibi FA, Reif G, Wallace DP, Yamaguchi T, Olsen L, Li H, Helmkamp GM Jr, Grantham JJ (2004) Cyclic AMP promotes growth and secretion in human polycystic kidney epithelial cells. Kidney Int 66:964–973
Cotton CU, Avner ED (1998) PKD and CF: an interesting family provides insight into the molecular pathophysiology of polycystic kidney disease. Am J Kidney Dis 32:1081–1083
Nakanishi K, Sweeney WE, Jr., Macrae DK, Cotton CU, Avner ED (2001) Role of CFTR in autosomal recessive polycystic kidney disease. J Am Soc Nephrol 12:719–725
Suzuki M, Morita T, Iwamoto T (2006) Diversity of Cl(-) channels. Cell Mol Life Sci 63:12–24
Huang P, Liu J, Di A, Robinson NC, Musch MW, Kaetzel MA, Nelson DJ (2001) Regulation of human CLC-3 channels by multifunctional Ca2+/calmodulin-dependent protein kinase. J Biol Chem 276:20093–20100
Marquardt A, Stohr H, Passmore LA, Kramer F, Rivera A, Weber BH (1998) Mutations in a novel gene, VMD2, encoding a protein of unknown properties cause juvenile-onset vitelliform macular dystrophy (Best’s disease). Hum Mol Genet 7:1517–1525
Qu Z, Wei RW, Mann W, Hartzell HC (2003) Two bestrophins cloned from Xenopus laevis oocytes express Ca(2+)-activated Cl(-) currents. J Biol Chem 278:49563–49572
Sun H, Tsunenari T, Yau KW, Nathans J (2002) The vitelliform macular dystrophy protein defines a new family of chloride channels. Proc Natl Acad Sci USA 99:4008–4013
Hartzell HC, Qu Z (2003) Chloride currents in acutely isolated Xenopus retinal pigment epithelial cells. J Physiol 549:453–469
Tsunenari T, Sun H, Williams J, Cahill H, Smallwood P, Yau KW, Nathans J (2003) Structure-function analysis of the bestrophin family of anion channels. J Biol Chem 278:41114–41125
Tsunenari T, Nathans J, Yau KW (2006) Ca2+-activated Cl- current from human bestrophin-4 in excised membrane patches. J Gen Physiol 127:749–754
Duta V, Szkotak AJ, Nahirney D, Duszyk M (2004) The role of bestrophin in airway epithelial ion transport. FEBS Lett 577:551–554
Suzuki M, Mizuno A (2004) A novel Cl channel related to Drosophila flightless locus. J Biol Chem 279:22461–22468
Edwards JC (1999) A novel p64-related Cl- channel: subcellular distribution and nephron segment-specific expression. Am J Physiol 276:F398–F408
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Veizis, I.E., Cotton, C.U. Role of kidney chloride channels in health and disease. Pediatr Nephrol 22, 770–777 (2007). https://doi.org/10.1007/s00467-006-0355-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-006-0355-4