Abstract
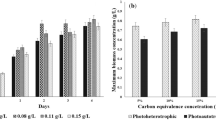
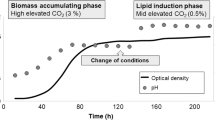
A mixed trophic state production process for algal lipids for use as feedstock for renewable biofuel production was developed and deployed at subpilot scale using a green microalga, Auxenochlorella (Chlorella) protothecoides. The process is composed of two separate stages: (1) the photoautotrophic stage, focused on biomass production in open ponds, and (2) the heterotrophic stage focused on lipid production and accumulation in aerobic bioreactors using fixed carbon substrates (e.g., sugar). The process achieved biomass and lipid productivities of 0.5 and 0.27 g/L/h that were, respectively, over 250 and 670 times higher than those obtained from the photoautotrophic cultivation stage. The biomass oil content (over 60 % w/DCW) following the two-stage process was predominantly monounsaturated fatty acids (~82 %) and largely free of contaminating pigments that is more suitable for biodiesel production than photosynthetically generated lipid. Similar process performances were obtained using cassava hydrolysate as an alternative feedstock to glucose.






Similar content being viewed by others
References
Heinrich JM, Niizawa I, Botta FA, Trombert AR, Irazoqui HA (2012) Analysis and design of photobioreactors for microalgae production II: experimental validation of a radiation field simulator based on a Monte Carlo algorithm. Photochem Photobiol 88(4):952–960. doi:10.1111/j.1751-1097.2012.01149.x
Xu L, Guo C, Wang F, Zheng S, Liu C-Z (2011) A simple and rapid harvesting method for microalgae by in situ magnetic separation. Bioresour Technol 102(21):10047–10051. doi:10.1016/j.biortech.2011.08.021
Halim R, Danquah MK, Webley PA (2012) Extraction of oil from microalgae for biodiesel production: a review. Biotechnol Advances 30(3):709–732. doi:10.1016/j.biotechadv.2012.01.001
Mercer P, Armenta R (2011) Developments in oil extraction from microalgae. Eur J Lipid Sci Technol. doi:10.1002/ejlt.201000455
Faeth J, Valdez P, Savage P (2013) Fast hydrothermal liquefaction of Nannochloropis sp. to produce biocrude. Energy Fuels 27:1391–1398
Onwudili JA, Lea-Langton AR, Ross AB, Williams PT (2013) Catalytic hydrothermal gasification of algae for hydrogen production: composition of reaction products and potential for nutrient recycling. Bioresour Technol 127:72–80. doi:10.1016/j.biortech.2012.10.020
Rampton R, Zabarenko D (2012) Algae biofuel not sustainable now, review says. msnbccom
Leite GB, Abdelaziz AEM, Hallenbeck PC (2013) Algal biofuels: challenges and opportunities. Bioresour Technol 145:134–141. doi:10.1016/j.biortech.2013.02.007
Samejima H, Myers J (1958) On the heterotrophic growth of Chlorella pyrenoidosa. J Gen Microbiol 18(1):107–117
Miao X, Wu Q (2006) Biodiesel production from heterotrophic microalgal oil. Bioresour Technol 97(6):841–846. doi:10.1016/j.biortech.2005.04.008
Azam F, Hemmingsen BB, Volcani BE (1974) Role of silicon in diatom metabolism. V. Silicic acid transport and metabolism in the heterotrophic diatom Nitzschia alba. Arch Microbiol 97(2):103–114
Lewin J, Hellebust JA (1975) Heterotrophic nutrition of the marine pennate diatom Navicula pavillardi Hustedt. Can J Microbiol 21(9):1335–1342
Chojnacka K, Noworyta A (2004) Evaluation of Spirulina sp. growth in photoautotrophic, heterotrophic and mixotrophic cultures. Enzyme Microb Technol 34(5):461–465. doi:10.1016/j.enzmictec.2003.12.002
Fabregas J, Garcia D, Lamela T, Morales ED, Otero A (1999) Mixotrophic production of phycoerythrin and exopolysaccharide by the microalga Porphyridium cruentum. Cryptogamie Algologie 20(2):89–94
Kamjunke N, Tittel J (2009) Mixotrophic algae constrain the loss of organic carbon by exudation. J Phycol 45(4):807–811. doi:10.1111/j.1529-8817.2009.00707.x
Sforza E, Cipriani R, Morosinotto T, Bertucco A, Giacometti GM (2012) Excess CO2 supply inhibits mixotrophic growth of Chlorella protothecoides and Nannochloropsis salina. Bioresour Technol 104:523–529. doi:10.1016/j.biortech.2011.10.025
Hu B, Min M, Zhou W, Du Z, Mohr M, Chen P, Zhu J, Cheng Y, Liu Y, Ruan R (2012) Enhanced mixotrophic growth of microalga Chlorella sp. on pretreated swine manure for simultaneous biofuel feedstock production and nutrient removal. Bioresour Technol 126C:71–79. doi:10.1016/j.biortech.2012.09.031
Ogbonna JC, Masui H, Tanaka H (1997) Sequential heterotrophic/autotrophic cultivation—an efficient method of producing Chlorella biomass for health food and animal feed. J Appl Phycol 9:359–366
Oyler JR (2008) Two-stage process for producing oil from microalgae. USA Patent, 8,475,543
Sayre R, Pereira S (2010) Molecuar approaches for the optimization of biofuel productions. USA Patent, 20100317073
Wu Q, Xiong W (2009) Method for producing biodiesel from an alga. USA Patent, 20090298159
Krohn BJ, McNeff CV, Yan B, Nowlan D (2011) Production of algae-based biodiesel using the continuous catalytic Mcgyan process. Bioresour Technol 102(1):94–100. doi:10.1016/j.biortech.2010.05.035
Heredia-Arroyo T, Wei W, Hu B (2010) Oil accumulation via heterotrophic/mixotrophic Chlorella protothecoides. Appl Biochem Biotechnol 162(7):1978–1995. doi:10.1007/s12010-010-8974-4
Xu H, Miao X, Wu Q (2006) High quality biodiesel production from a microalga Chlorella protothecoides by heterotrophic growth in fermenters. J Biotechnol 126(4):499–507. doi:10.1016/j.jbiotec.2006.05.002
Huss V, Frank C, Hartmann E, Hirmer M, Kloboucek A, Seidel B, Wenzeler P, Kessler E (1999) Biochemical taxonomy and molecular phylogeny of the genus Chlorella sensu lato (Chlorophyta). J Phycol 35:587–598
Miao X, Wu Q (2004) High yield bio-oil production from fast pyrolysis by metabolic controlling of Chlorella protothecoides. J Biotechnol 110(1):85–93. doi:10.1016/j.jbiotec.2004.01.013
Konieczny A, Ausubel FM (1993) A procedure for mapping Arabidopsis mutations using co-dominant ecotype-specific PCR-based markers. Plant J 4(2):403–410
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37(8):911–917
Greenspan P, Mayer EP, Fowler SD (1985) Nile red: a selective fluorescent stain for intracellular lipid droplets. J Cell Biol 100(3):965–973
Sayre RT (2009) Optimization of biofuel production. USA Patent US20090181438
Santos CA, Nobre B, Lopes da Silva T, Pinheiro HM, Reis A (2014) Dual-mode cultivation of Chlorella protothecoides applying inter-reactors gas transfer improves microalgae biodiesel production. J Biotechnol 184:74–83. doi:10.1016/j.jbiotec.2014.05.012
Ramos MJ, Fernandez CM, Casas A, Rodriguez L, Perez A (2009) Influence of fatty acid composition of raw materials on biodiesel properties. Bioresour Technol 100(1):261–268. doi:10.1016/j.biortech.2008.06.039
Hu Q, Sommerfeld M, Jarvis E, Ghirardi M, Posewitz M, Seibert M, Darzins A (2008) Microalgal triacylglycerols as feedstocks for biofuel production: perspectives and advances. Plant J 54(4):621–639. doi:10.1111/j.1365-313X.2008.03492.x
Xiong W, Li X, Xiang J, Wu Q (2008) High-density fermentation of microalga Chlorella protothecoides in bioreactor for microbio-diesel production. Appl Microbiol Biotechnol 78(1):29–36. doi:10.1007/s00253-007-1285-1
Hortensteiner S, Chinner J, Matile P, Thomas H, Donnison IS (2000) Chlorophyll breakdown in Chlorella protothecoides: characterization of degreening and cloning of degreening-related genes. Plant Mol Biol 42(3):439–450
Ramos Tercero EA, Sforza E, Morandini M, Bertucco A (2014) Cultivation of Chlorella protothecoides with urban wastewater in continuous photobioreactor: biomass productivity and nutrient removal. Appl Biochem Biotechnol 172(3):1470–1485. doi:10.1007/s12010-013-0629-9
Lu Y, Zhai Y, Liu M, Wu Q (2010) Biodiesel production from algal oil using cassava (Manihot esculenta Crantz) as feedstock. J Appl Phycol 22(5):573–578. doi:10.1007/s10811-009-9496-8
Shetty J, Chotani G, Gand D, Bates D (2007) Cassava as an alternative feedstock in the production of renewable transportation fuels. Intl Sugar J 109(1307):663–677
Acknowledgments
This research project was supported under the Department of Energy grant DE-FE-0000888 awarded by National Energy Technology Laboratory. Additional support was supplied by a large group of excellent support staff at Phycal in the R&D group who worked tirelessly to help enable this process.
Conflict of interest
All authors were former employees of Phycal Inc. and paid in part from a grant supplied by the US Department of Energy.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rismani-Yazdi, H., Hampel, K.H., Lane, C.D. et al. High-productivity lipid production using mixed trophic state cultivation of Auxenochlorella (Chlorella) protothecoides . Bioprocess Biosyst Eng 38, 639–650 (2015). https://doi.org/10.1007/s00449-014-1303-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-014-1303-5