Abstract
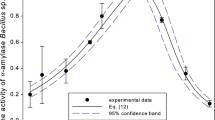
Starch hydrolysis was performed by the synergistic action of amylase and glucoamylase. For that purpose glucoamylase (Dextrozyme) and two amylases (Liquozyme and Termamyl) in different combinations were investigated. Experiments were carried out in the repetitive- and fed-batch modes at 65 °C and pH 5.5 with and without the addition of Ca2+ ions. 100 % conversion of starch to glucose was achieved in batch experiments. Calcium ions significantly enhanced stability of the amylase Termamyl. The intensity of synergism between amylase Termamyl and glucoamylase Dextrozyme was higher than in the experiments carried out with amylase Liquozyme and Dextrozyme. Mathematical model of the complete reaction system was developed. Using the model, a possible explanation of the synergism between the amylase and glucoamylase was provided.


Similar content being viewed by others
Abbreviations
- c :
-
Concentration, g dm−3
- k d :
-
Inactivation constant, min−1
- K i :
-
Inhibition constant, g dm−3
- K m :
-
Michaelis–Menten constant, g dm−3
- m :
-
Mass, kg
- q :
-
Flow rate, dm3 min−1
- r :
-
Reaction rate, g dm−3 min−1
- R 2 :
-
Coefficient of determination
- SD :
-
Standard deviation
- t :
-
Time, min, h
- T :
-
Temperature, °C
- V :
-
Volume, dm3
- V m :
-
Maximal activity, g dm−3 min−1
- Y :
-
Yield
- φ:
-
Volume ratio of enzyme, dm3dm−3
References
Lim LH, Macdonald DG, Hill GA (2003) Hydrolysis of starch particles using immobilized barley α-amylase. Biochem Eng J 13:53–62
Crabb WD, Shetty JK (1999) Commodity scale production of sugars from starches. Curr Opin Microbiol 2:252–256
Ji Y, Ao Z, Han J-A, Jane J-L, BeMiller JN (2004) Waxy maize starch subpopulations with different gelatinization temperatures. Carbohyd Res 57:177–190
Liu H, Yu L, Xie F, Chen L (2006) Gelatinization of corn starch with different amylase/amylopectin content. Carbohyd Res 65:357–363
Zobel HF, Young SN, Rocca LA (1988) Starch gelatinization: an X-ray diffraction study. Cereal Chem 65:443–446
Schwimmer S (1945) The role of maltase in the enzymolysis of raw starch. J Biol Chem 161:219–233
Ma Y, Cai C, Wang J, Sun D-W (2006) Enzymatic hydrolysis of corn starch for producing fat mimetics. J Food Eng 73:297–303
Balls AK, Schwimmer S (1944) Digestion of raw starch. J Biol Chem 156:203–210
Wong DWS, Robertson GH, Lee CC, Wagschal K (2007) Synergystic action of recombinant α-amylase and glucoamylase on the hydrolysis of starch granules. Prot J 26:159–164
Gorinstein S (1993) Kinetic studies during enzyme hydrolysis of potato and cassava starches. Starch 45:91–95
Bryjak J, Ciesielski K, Zbiciński I (2004) Modelling of glucoamylase thermal inactivation in the presence of starch by artificial neural network. J Biotechnol 114:177–185
Cepeda E, Hermosa M, Ballesteros A (2001) Optimization of maltodextrin hydrolysis by glucoamylase in a batch reactor. Biotechnol Bioeng 76:70–76
Shiraishi F, Kawakami K, Kusunoki K (1985) Kinetics of condensation of glucose into maltose and isomaltose in hydrolysis of starch by glucoamylase. Biotechnol Bioeng 27:498–502
Polakovič M, Bryjak J (2002) Modelling of the kinetics of thermal inactivation of glucoamylase from Aspergillus niger. J Mol Cat Enzym: B 19–20:443–450
Presečki VA, Blažević ZF, Vasić-Rački Đ (2013) Mathematical modeling of maize starch liquefaction catalyzed by alpha-amylases from Bacillus licheniformis—effect of calcium, pH and temperature. Bioprocess Biosyst Eng 36:117–126
Roy I, Gupta MN (2004) Hydrolysis of starch by a mixture of glucoamylase and pullulanase entrapped individually in calcium alginate beads. Enzym Microb Tech 34:26–32
Adachi S, Ueda Y, Hashimoto K (1984) Kinetics of formation of maltose and isomaltose through condensation of glucose by glucoamylase. Biotechnol Bioeng 26:121–127
Li M, Kim j-W, Peeples TL (2002) Kinetic enhancement of starch bioconversion in thermoseparating aqueous two-phase reactor systems. Biochem Eng J 11:25–32
Fuji M, Kawamura Y (1985) Synergistic action of α-amylase and glucoamylase on hydrolysis of starch. Biotechnol Bioeng 27:260–265
Findrik Z, Vrsalović Presečki A, Vasić-Rački Đ (2010) Mathematical modeling of maltose hydrolysis in different types of reactor. Bioprocess Biosyst Eng 33:299–307
Xiao Z, Storms R, Tsang A (2006) A quantitative starch-iodine method for measuring alpha-amylase and glucoamylase activities. Anal Biochem 351:146–148
Apar DK, Özbek B (2007) Estimation of kinetic parameters for rice starch hydrolysis inhibited by added materials. Chem Eng Comm 194:334–344
Violet M, Meunier JC (1989) Kinetic study of the irreversible thermal denaturation of Bacillus licheniformis α-amylase. Biochem J 263:665–670
SCIENTIST handbook (1986–1995) Micromath®, Salt Lake City
Lopez C, Torrado A, Fucinos P, Guerra NP, Pastrana L (2004) Enzymatic hydrolysis of chestnut purée: process optimization using mixtures of α-amylase and glucoamylase. J Agric Food Chem 52:2907–2914
Sanroman A, Murado MA, Lema JM (1995) The influence of substrate structure on the kinetics of the hydrloysis of starch by glucoamylase. Appl Biochem Biotechnol 59:329–336
Lopez C, Torrado A, Fucinos P, Guerra NP, Pastrana L (2006) Enzymatic inhibition and thermal inactivation in the hydrolysis of chestnut purée with an amylases mixture. Enzyme Microb Tech 39:252–258
Miranda M, Murado MA, Sanroman A, Lema JM (1991) Mass transfer control of enzymatic hydrolysis of polisaccharides by glucoamylase. Enzyme Microb Techn 13:142–147
Soni KS, Kaur A, Gupta JK (2003) A solid state fermentation based bacterial α-amylase and fungal glucoamylase system and its suitability for the hydrolysis of wheat starch. Process Biochem 39:185–192
Liakopoulou-Kyriakides M, Karakatsanis A, Stamatoudis M, Psomas S (2001) Synergistic hydrolysis of crude corn starch by α-amylases and glucoamylases of various origins. Cereal Chem 78:603–607
Lopez C, Torrado A, Guerra NP, Pastrana L (2005) Optimization of solid-state enzymatic hydrolysis of chestnut purée using mixtures of α-amylase and glucoamylase. J Agric Food Chem 53:989–995
Akerberg C, Zacchi G, Torto N, Gorton L (2000) A kinetic model for enzymatic wheat starch saccharification. J Chem Technol Biotechnol 75:306–314
Gouda MD, Kumar MA, Thakur MS, Karanth NG (2002) Enhancement of operational stability of an enzyme biosensor for glucose and sucrose using protein based stabilizing agents. Biosens Bioelectron 17:503–507
Gouda MD, Singh SA, Rao AGA, Thakur MS, Karanth NG (2003) Thermal inactivation of glucose oxidase-mechanism and stabilization using additives. J Biol Chem 278:24324–24333
Iyer PV, Ananthanarayan L (2008) Enzyme stability and stabilization—Aqueous and non-aqueous environment. Process Biochem 43:1019–1032
Costa SA, Tzanov T, Carneiro AF, Paar A, Gübitz GM, Cavaco-Paulo A (2002) Studies of stabilization of native catalase using additives. Enzyme Microb Tech 30:387–391
Acknowledgments
This research was supported by the Croatian Ministry of science, education and sport by grant 125-1252086-2793. The authors gratefully acknowledge Novozymes (Denmark) for the gift of Termamyl, Liquozyme and Dextrozyme.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Presečki, A.V., Blažević, Z.F. & Vasić-Rački, Đ. Complete starch hydrolysis by the synergistic action of amylase and glucoamylase: impact of calcium ions. Bioprocess Biosyst Eng 36, 1555–1562 (2013). https://doi.org/10.1007/s00449-013-0926-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-013-0926-2