Abstract
The Wave Bioreactor® is widely used in cell culture due to the benefits of disposable technology and ease of use. A novel cellbag was developed featuring a frit sparger to increase the system's oxygen transfer. The purpose of this work was to evaluate the sparged cellbag for yeast cultivation. Oxygen mass transfer studies were conducted in simulated culture medium and the sparged system's maximum oxygen mass transfer coefficient (k L a) was 38 h-1. These measurements revealed that the sparger was ineffective in increasing the oxygen transfer capacity. Cultures of Saccharomyces cerevisiae were successfully grown in oxygen-blended sparged and oxygen-blended standard cellbags. Under steady state conditions for both cellbag designs, k L a values as high as 60 h-1 were obtained with no difference in growth characteristics. This is the first report of a successful cultivation of a microbe in a Wave Bioreactor® comparing conventional seed expansion in shake flasks and stirred tank bioreactors.









Similar content being viewed by others
Abbreviations
- C*:
-
saturated dissolved oxygen concentration in the liquid phase (mmol L−1)
- C 1 :
-
dissolved oxygen concentration at time t 1 (mmol L−1)
- C 2 :
-
dissolved oxygen concentration at time t 2 (mmol L−1)
- CER:
-
carbon dioxide evolution rate (mmol L−1 h−1)
- DO:
-
dissolved oxygen (% air sat)
- k L a :
-
volumetric oxygen transfer coefficient (h−1)
- OD:
-
optical density
- OTR:
-
oxygen transfer rate (mmol L−1 h−1)
- OUR:
-
oxygen uptake rate (mmol L−1 h−1)
- PBS:
-
phosphate buffered saline
- RPM:
-
rocks per minute (min−1)
- RQ:
-
respiration quotient (CER/OUR)
- s :
-
substrate (g L−1)
- vvm:
-
volume of gas sparge per volume liquid per minute (L L−1 min−1)
- Y x/s :
-
yield coefficient, g biomass/g glucose
- x :
-
biomass (g)
References
Namdev PK, Lio P (2000) Assessing a disposable bioreactor for attachment-dependant cell cultures. BioPharm 13:44–50
Weber W, Weber E, Geisse S, Memmert K (2002) Optimisation of protein expression and establishment for the Wave Bioreactor® for Baculovirus/insect cell culture. Cytotechnology 38:77–85
Palazón J, Mallol A, Eibl R, Lettenbauer C, Cusidó R, Pinol MT (2003) Growth and ginsenoside production in hairy root cultures of using a novel bioreactor. Planta Med 69:344–349
Jablonski-Lorin C, Melillo V, Hungerbuhler E (2003) Stereoselective bioreductions to a chiral building block on a kilogram scale. Chimia 57:574–576
Singh V (1999) Disposable bioreactor for cell culture using wave-induced agitation. Cytotechnology 30:149–158
Knevelman C, Hearle DC, Osman JJ, Khan M, Dean M, Smith M, Aiyedebinu A, Cheung K (2002) Characterisation and operation of a disposable bioreactor as a replacement for conventional stem in place inoculum bioreactors for mammalian cell culture processes. In: 224th Meeting of American Chemical Society, August 18–22, Boston
Tolosa L, Kostov Y, Harms P, Rao G (2002) Noninvasive measurements of dissolved oxygen in shake flasks. Biotech Bioeng 80:594–597
Otterstedt K, Larsson C, Bill RM, Ståhlberg A, Boles E, Hohmann S, Gustafsson L (2004) Switching the mode of metabolism in the yeast Saccharomyces cerevisiae. Eur Mol Biol Organ Rep 5:532–537
Verduyn C, Zomerdijk TPL, Van Dijken JP, Scheffers WA (1984) Continuous measurement of ethanol production by aerobic yeast suspension with an enzyme electrode. Appl Microbiol Biotechnol 19:181–185
Elbing K, Larsson C, Bill RM, Albers E, Snoep J, Boles E, Hohmann S, Gustafasson L (2004) Role of hexose transport in control of glycolytic flux in Saccharomyces cerevisiae. Appl Environ Microbiol 70:5323–5330
Diderich JA, Schepper M, van Hoek P, Luttis AH, van Dijken JP, Pronk JY, Klaassen P, Boelens HFM, Teixeira de Mattos MJ, van Dam K, Kruckeberg AL (1999) Glucose uptake kinetics and transcription of HXT genes in chemostat cultures of Saccharomyces cerevisiae. J Biol Chem 274(22):15350–15359
Rehm H-J, Reed G, Puhler A, Stadler P (1993). Biotechnology. VCH Verlagsgesellschaft mgH. Weinheim Federal Republic of Germany, pp 190–192. ISBN # 1-56081-153-6
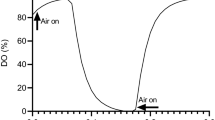
Merchuk JC, Yona S, Siegel MH, Ben Zvi A (1989). On the first-order approximation to the response of dissolved oxygen electrodes for dynamic k L a estimation. Biotechnol Bioeng 35:1161–1163
Tunac JB (1989) High-aeration capacity shake-flask system. J Ferm Bioeng 68:157–159
Verduyn C, Postma E, Scheffers WA, Van-Dijken JP (1992) Effect of benzoic acid in metabolic fluxes in yeasts a continuous-culture study on the regulation of respiration and alcoholic fermentation. Yeast 8:501–517
Zlokarnik M (1978) Advances in biochemical engineering. Springer, Berlin, vol 8, p 134
Gupta A, Rao G (2003) A study of oxygen transfer in shake flask using a non-invasive oxygen sensor. Biotechnol Bioeng 84:351–358
Anderlei T, Zang W, Papaspyrou M, Buchs J (2004) Online respiration activity measurement *OTR, CTR, RQ) in shake flasks. Biochem Eng J 17:187–194
Junker B, Seeley A, Lester M, Kovatch M, Schmitt J, Boryscwicz S, Lynch J, Zhang J, Greasham R (2002) Use of frozen bagged seed inoculum for secondary metabolite and bioconversion processes at the Pilot scale. Biotechnol Bioeng 79:628–640
Tan T, Zhang M, Gao H (2003) Esgosterol production by fed-batch fermentation of Saccharomyces cerevisiae. Enzyme Microbial Tech 33:366–370
Pierce LN, Shabram PW (2004) Scalability of a disposable bioreactor from 25L–500L Run in the perfusion mode with a CHO-based cell line: a tech review. BioProcess J 3:1–6
Acknowledgments
The authors would like to acknowledge and thank Dr. Arthur L. Kruckeberg for supplying the AlcoFree® strain and for useful discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mikola, M., Seto, J. & Amanullah, A. Evaluation of a novel Wave Bioreactor® cellbag for aerobic yeast cultivation. Bioprocess Biosyst Eng 30, 231–241 (2007). https://doi.org/10.1007/s00449-007-0119-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-007-0119-y