Abstract
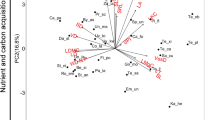
In Neotropical forests, lianas are increasing in abundance relative to trees. This increased species richness may reflect a positive response to global change factors including increased temperature, atmospheric CO2, habitat fragmentation, and drought severity; however, questions remain as to the specific mechanisms facilitating the response. Previous work suggests that lianas may gain an ecological advantage over trees through leaf functional traits that offer a quick return on investment of resources, although it is unknown whether this pattern extends to root traits and relationships with fungal or bacterial symbionts belowground. We sampled confamilial pairs of liana and tree species and quantified morphological and chemical traits of leaves and fine roots, as well as root symbiont abundance, to determine whether functional traits associated with resource acquisition differed between the two. Compared to trees, lianas possessed higher specific leaf area, specific root length, root branching intensity, and root nitrogen (N) and phosphorus (P) concentrations, and lower leaf and root tissue density, leaf and root carbon (C), root diameter, root C:P and N:P, and mycorrhizal colonization. Our study provides new evidence that liana leaf and root traits are characterized by a rapid resource acquisition strategy relative to trees. These liana functional traits may facilitate their response to global change, raising questions about how increased liana dominance might affect ecosystem processes of Neotropical forests.



Similar content being viewed by others
References
Ackerly DD (2004) Adaptation, niche conservatism, and convergence: comparative studies of leaf evolution in the California chaparral. Am Nat 163:654–671
Baillie I, Elsenbeer H, Barthold F et al (2007) Semi-detailed soil survey of Barro Colorado Island, Panama
Bardgett RD (2011) Plant-soil interactions in a changing world. F1000 Biol Rep 3:16. doi:10.3410/B3-16
Benítez-Malvido J, Martínez-Ramos M, Dynamics B et al (2003) Impact of forest fragmentation on understory plant species richness in Amazonia. Conserv Biol 17:389–400
Bethlenfalvay GJ, Brown MS, Stafford AE (1985) Glycine-Glomus-Rhizobium symbiosis: II. Antagonistic effects between mycorrhizal colonization and nodulation. Plant Physiol 79:1054–1058. doi:10.1104/pp.79.4.1054
Bethlenfalvay GJ, Andrade G, Azcón-Aguilar C (1997) Plant and soil responses to mycorrhizal fungi and rhizobacteria in nodulated or nitrate-fertilized peas (Pisum sativum L.). Biol Fertil Soils 24:164–168. doi:10.1007/s003740050225
Bloom AJ, Chapin FS, Mooney HA (1985) Resource limitation in plants—an economic analogy. Annu Rev Ecol Syst 16:363–392. doi:10.1146/annurev.ecolsys.16.1.363
Brundrett M (1991) Mycorrhizas in natural ecosystems. Adv Ecol Res 21:171–313. doi:10.1016/S0065-2504(08)60099-9
Brundrett M (2002) Coevolution of roots and mycorrhizas of land plants. New Phytol 154:275–304
Cai Z-Q, Poorter L, Cao K-F, Bongers F (2007) Seedling growth strategies in Bauhinia species: comparing lianas and trees. Ann Bot 100:831–838. doi:10.1093/aob/mcm179
Caplan JS, Wheaton CN, Mozdzer TJ (2014) Belowground advantages in construction cost facilitate a cryptic plant invasion. AoB Plants 6:1–10. doi:10.1093/aobpla/plu020
Comas LH, Eissenstat DM (2004) Linking fine root traits to maximum potential growth rate among 11 mature temperate tree species. Funct Ecol 18:388–397. doi:10.1111/j.0269-8463.2004.00835
Comas LH, Callahan HS, Midford PE (2014) Patterns in root traits of woody species hosting arbuscular and ectomycorrhizas: implications for the evolution of belowground strategies. Ecol Evol 4:2979–2990. doi:10.1002/ece3.1147
Cornelissen JHC, Lavorel S, Garnier E, Díaz S, Buchmann N, Gurvich DE, Reich PB, Ter Steege H, Morgan HD, Van Der Heijden MGA, Pausas JG, Poorter H (2003) A handbook of protocols for standardised and easy measurement of plant functional traits worldwide. Aust J Bot 51:335
Craine J, Bond W, Lee WG et al (2003) The resource economics of chemical and structural defenses across nitrogen supply gradients. Oecologia 137:547–556. doi:10.1007/s00442-003-1370-9
Cramer W, Bondeau A, Woodward FI et al (2001) Global response of terrestrial ecosystem structure and function to CO2 and climate change: results from six dynamic global vegetation models. Glob Chang Biol 7:357–373. doi:10.1046/j.1365-2486.2001.00383.x
Croat TB (1978) Flora of Barro Colorado Island. Stanford University Press, Stanford, 943 pp
Darwin C (1867) On the movements and habits of climbing plants. J Linn Soc 9:1–118
De Deyn GB, Cornelissen JHC, Bardgett RD (2008) Plant functional traits and soil carbon sequestration in contrasting biomes. Ecol Lett 11:516–531. doi:10.1111/j.1461-0248.2008.01164.x
Dewalt SJ, Schnitzer SA, Denslow JS (2000) Density and diversity of lianas along a chronosequence in a central Panamanian lowland forest. J Trop Ecol 16:1–19. doi:10.1017/S0266467400001231
Díaz S, Cabido M (2001) Vive la différence: plant functional diversity matters to ecosystem processes. Trends Ecol Evol 16:646–655
Díaz S, Hodgson JG, Thompson K et al (2004) The plant traits that drive ecosystems: evidence from three continents. J Veg Sci 15:295. doi:10.1658/1100-9233(2004)015[0295:TPTTDE]2.0.CO;2
Eissenstat DM (1992) Costs and benefits of constructing roots of small diameter. J Plant Nutr 15:763–782. doi:10.1080/01904169209364361
Eissenstat DM, Yanai RD (1997) The ecology of root lifespan. Adv Ecol Res 27:1–60. doi:10.1016/S0065-2504(08)60005-7
Eissenstat DM, Wells CE, Yanai RD, Whitbeck JL (2000) Building roots in a changing environment: implications for root longevity. New Phytol 147:33–42. doi:10.1046/j.1469-8137.2000.00686.x
Enders RK (1935) Mammalian life histories from Barro Colorado Island, Panama. Bull Mus Comp Zool 78:385–502
Fitter A (1982) Morphometric analysis of root systems: application of the technique and influence of soil fertility on root system development in two herbaceous species. Plant Cell Environ 5:313–322. doi:10.1111/1365-3040.ep11573079
Fitter AH, Stickland TR, Harvey ML, Wilson GW (1991) Architectural analysis of plant root systems. 1. Architectural correlates of exploitation efficiency. New Phytol 118:375–382. doi:10.1111/j.1469-8137.1991.tb00018.x
Gill RA, Jackson RB (2000) Global patterns of root turnover for terrestrial ecosystems. New Phytol 147:13–31. doi:10.1046/j.1469-8137.2000.00681.x
Grime JP (1977) Evidence for the existence of three primary strategies in plants and its relevance to ecological and evolutionary theory. Am Nat 111:1169. doi:10.1086/283244
Grime JP, Thompson K, Hunt R et al (1997) Integrated screening validates primary axes of specialisation in plants. Oikos 79:259–281
Guo D, Xia M, Wei X et al (2008) Anatomical traits associated with absorption and mycorrhizal colonization are linked to root branch order in twenty-three Chinese temperate tree species. New Phytol 180:673–683. doi:10.1111/j.1469-8137.2008.02573.x
Hetrick BAD (1991) Mycorrhizas and root architecture. Experientia 47:355–362. doi:10.1007/BF01972077
Hetrick BAD, Wilson GWT, Leslie JF (1991) Root architecture of warm- and cool-season grasses: relationship to mycorrhizal dependence. Can J Bot 69:112–118. doi:10.1139/b91-016
Holdaway RJ, Richardson SJ, Dickie IA et al (2011) Species- and community-level patterns in fine root traits along a 120 000-year soil chronosequence in temperate rain forest. J Ecol 99:954–963. doi:10.1111/j.1365-2745.2011.01821.x
Ingwell LL, Joseph Wright S, Becklund KK et al (2010) The impact of lianas on 10 years of tree growth and mortality on Barro Colorado Island, Panama. J Ecol 98:879–887. doi:10.1111/j.1365-2745.2010.01676.x
Kitajima K, Poorter L (2010) Tissue-level leaf toughness, but not lamina thickness, predicts sapling leaf lifespan and shade tolerance of tropical tree species. New Phytol 186:708–721. doi:10.1111/j.1469-8137.2010.03212.x
Kitajima K, Llorens AM, Stefanescu C et al (2012) How cellulose-based leaf toughness and lamina density contribute to long leaf lifespans of shade-tolerant species. New Phytol 195:640–652. doi:10.1111/j.1469-8137.2012.04203.x
Kress WJ, Erickson DL, Jones FA et al (2009) Plant DNA barcodes and a community phylogeny of a tropical forest dynamics plot in Panama. Proc Natl Acad Sci USA 106:18621–18626. doi:10.1073/pnas.0909820106
Laurance WF, Andrade AS, Magrach A et al (2014) Long-term changes in liana abundance and forest dynamics in undisturbed Amazonian forests. Ecology 95:1604–1611
Lavorel S, Garnier E (2002) Predicting changes in community composition and ecosystem functioning from plant traits: revisiting the Holy Grail. Funct Ecol 16:545–556
Lavorel S, Grigulis K (2012) How fundamental plant functional trait relationships scale-up to trade-offs and synergies in ecosystem services. J Ecol 100:128–140. doi:10.1111/j.1365-2745.2011.01914.x
Lavorel S, Díaz S, Cornelissen J et al (2007) Plant functional types: Are we getting any closer to the Holy Grail? In: Terrestrial ecosystems in a changing world, pp 149–160. doi:10.1007/978-3-540-32730-1_13
Leigh EGJ (1999) Tropical forest ecology: a view from Barro Colorado Island. Oxford University Press, Oxford, 264 pp
McCormack ML, Guo D (2014) Impacts of environmental factors on fine root lifespan. Front Plant Sci 5:205. doi:10.3389/fpls.2014.00205
McCormack ML, Adams TS, Smithwick EH, Eissenstat DM (2012) Predicting fine root lifespan from plant functional traits in temperate trees. New Phytol 195:823–831. doi:10.1111/j.1469-8137.2012.04198.x
McDougall BM, Rovira AD (1970) Sites of exudation of 14C-labelled compounds from wheat roots. New Phytol 69:999–1003. doi:10.1111/j.1469-8137.1970.tb02479.x
McGill BJ, Enquist BJ, Weiher E, Westoby M (2006) Rebuilding community ecology from functional traits. Trends Ecol Evol 21:178–185. doi:10.1016/j.tree.2006.02.002
McGonigle TP, Miller MH, Evans DG et al (1990) A new method which gives an objective measure of colonization of roots by vesicular-arbuscular mycorrhizal fungi. New Phytol 115:495–501. doi:10.1111/j.1469-8137.1990.tb00476.x
Menge DNL, Levin SA, Hedin LO (2008) Evolutionary tradeoffs can select against nitrogen fixation and thereby maintain nitrogen limitation. Proc Natl Acad Sci USA 105:1573–1578. doi:10.1073/pnas.0711411105
Osnas JLD, Lichstein JW, Reich PB, Pacala SW (2013) Global leaf trait relationships: mass, area, and the leaf economics spectrum. Science 340:741–744. doi:10.1126/science.1231574
Ostonen I, Püttsepp Ü, Biel C et al (2007) Specific root length as an indicator of environmental change. Plant Biosyst 141:426–442. doi:10.1080/11263500701626069
Phillips OL, Vásquez Martínez R, Arroyo L et al (2002) Increasing dominance of large lianas in Amazonian forests. Nature 418:770–774. doi:10.1038/nature00926
Pregitzer KS, Deforest JL, Burton AJ et al (2002) Fine root architecture of nine North American trees. Ecol Monogr 72:293–309
Putz FE (1983) Liana biomass and leaf area of a “tierra firme” forest in the Rio Negro basin, Venezuela. Biotropica 15:185–189
Reich B, Hall G (2013) Comparisons of structure and life span in roots and leaves among temperate trees. Author(s): Jennifer M. Withington, Peter B. Reich, Jacek Oleksyn and David M. Eissenstat. Reviewed work(s): Published by Ecological Society of America. Ecol Monogr 76:381–397
Reich PB, Walters MB, Ellsworth DS (1997) From tropics to tundra: global convergence in plant functioning. Proc Natl Acad Sci USA 94:13730–13734
Reich PB, Ellsworth DS, Walters MB et al (1999) Generality of leaf trait relationships: a test across six biomes. Ecology 80:1955–1969. doi:10.1890/0012-9658(1999)080[1955:GOLTRA]2.0.CO;2
Reich PB, Wright IJ, Cavender-Bares J et al (2003) The evolution of plant functional variation: traits, spectra, and strategies. Int J Plant Sci 164:S143–S164. doi:10.1086/374368
Reichstein M, Bahn M, Mahecha MD et al (2014) Linking plant and ecosystem functional biogeography. Proc Natl Acad Sci USA. doi:10.1073/pnas.1216065111
Roumet C, Urcelay C, Díaz S (2006) Suites of root traits differ between annual and perennial species growing in the field. New Phytol 170:357–368. doi:10.1111/j.1469-8137.2006.01667.x
Ryser P (1996) The importance of tissue density for growth and life span of leaves and roots: a comparison of five ecologically contrasting grasses. Funct Ecol 10:717. doi:10.2307/2390506
Sayer EJ, Heard MS, Grant HK, Marthews TR, Tanner EVJ (2011) Soil carbon release enhanced by increased tropical forest litterfall. Nat Clim Change 1:304–307
Schnitzer SA (2005) A mechanistic explanation for global patterns of liana abundance and distribution. Am Nat 166:262–276. doi:10.1086/431250
Schnitzer SA, Bongers F (2011) Increasing liana abundance and biomass in tropical forests: emerging patterns and putative mechanisms. Ecol Lett 14:397–406. doi:10.1111/j.1461-0248.2011.01590.x
Schnitzer SA, Mangan SA, Dalling JW et al (2012) Liana abundance, diversity, and distribution on Barro Colorado Island, Panama. PLoS One 7:e52114. doi:10.1371/journal.pone.0052114
Steeves TA, Sussex IM (1989) Patterns in plant development. Cambridge University Press, Cambridge, 388 pp. doi:10.1017/CBO9780511626227
Sterck F, Markesteijn L, Schieving F, Poorter L (2011) Functional traits determine trade-offs and niches in a tropical forest community. Proc Natl Acad Sci USA 108:20627–20632. doi:10.1073/pnas.1106950108
Sterner RW, Elser JJ (2002) Ecological stoichiometry: the biology of elements from molecules to the biosphere. Princeton University Press, Princeton, 439 pp
Tang Y, Kitching RL, Cao M (2011) Lianas as structural parasites: a re-evaluation. Chin Sci Bull 57:307–312. doi:10.1007/s11434-011-4690-x
Trumbore S, Da Costa ES, Nepstad DC et al (2006) Dynamics of fine root carbon in Amazonian tropical ecosystems and the contribution of roots to soil respiration. Glob Chang Biol 12:217–229. doi:10.1111/j.1365-2486.2005.001063.x
Wahl S, Ryser P (2000) Root tissue structure is linked to ecological strategies of grasses. New Phytol 148:459–471
Westoby M, Wright IJ (2006) Land-plant ecology on the basis of functional traits. Trends Ecol Evol 21:261–268. doi:10.1016/j.tree.2006.02.004
Witkowski ETF, Lamont BB (1991) Leaf specific mass confounds leaf density and thickness. Oecologia 88:486–493. doi:10.1007/BF00317710
Wright IJ, Reich PB, Westoby M et al (2004a) The worldwide leaf economics spectrum. Science 12:821–827
Wright SJ, Calderón O, Hernandéz A, Paton S (2004b) Are lianas increasing in importance in tropical forests? A 17-year record from Panama. Ecology 85:484–489. doi:10.1890/02-0757
Wright SJ, Kitajima K, Kraft NJB et al (2010) Functional traits and the growth-mortality trade-off in tropical trees. Ecology 91:3664–3674
Wurzburger N, Wright SJ (2015) Fine root responses to fertilization reveal multiple nutrient limitation in a lowland tropical forest. Ecology. doi:10.1890/14-1362.1
Yavitt JB, Wieder RK (1988) Nitrogen, phosphorus, and sulfur properties of some forest soils on Barro Colorado Island, Panama. Biotropica 20:2–10
Yavitt JB, Harms KE, Garcia MN, Mirabello MJ, Wright SJ (2011) Soil fertility and fine root dynamics in response to 4 years of nutrient (N, P, K) fertilization in a lowland tropical moist forest, Panama. Austral Ecol 36:433–445
Yorke SR, Schnitzer SA, Mascaro J et al (2013) Increasing liana abundance and basal area in a tropical forest: the contribution of long-distance clonal colonization. Biotropica 45:317–324. doi:10.1111/btp.12015
Zhu S-D, Cao K-F (2010) Contrasting cost-benefit strategy between lianas and trees in a tropical seasonal rain forest in southwestern China. Oecologia 163:591–599. doi:10.1007/s00442-010-1579-3
Acknowledgments
We thank Rufino Gonzalez and Oldemar Valdez for field assistance and plant identification, Rick Lankau for statistical advice, Stefan Schnitzer for liana species information, Tom Maddox and Ben Turner for chemical analyses, Shialoh Wilson, Tierney O’Sullivan and Dina Abdulhadi for assistance in the lab, Kelly Andersen for comments and logistical support, and Teresa Bohner and Jake Allgeier for assistance with graphics. Funding was provided by the University of Georgia and the Odum School of Ecology, and research was conducted under a short term fellowship at the Smithsonian Tropical Research Institute.
Author contribution statement
CGC and NW developed research ideas and designed the project. CGC and SJW conducted fieldwork and collected the data. CGC and NW analyzed the data. CGC and NW wrote the manuscript, and SJW provided editorial advice.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Bettina Engelbrecht.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Collins, C.G., Wright, S.J. & Wurzburger, N. Root and leaf traits reflect distinct resource acquisition strategies in tropical lianas and trees. Oecologia 180, 1037–1047 (2016). https://doi.org/10.1007/s00442-015-3410-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-015-3410-7