Abstract
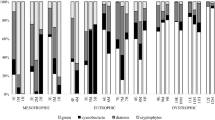
This study demonstrates that clear and humic freshwater pelagic communities respond differently to the same environmental stressors, i.e. nutrient and light availability. Thus, effects on humic communities cannot be generalized from existing knowledge about these environmental stressors on clear water communities. Small humic lakes are the most numerous type of lake in the boreal zone, but little is known about how these lakes will respond to increased inflows of nutrients and terrestrial dissolved organic C (t-DOC) due to climate change and increased human impacts. Therefore, we compared the effects of nutrient addition and light availability on pelagic humic and clear water lake communities in a mesocosm experiment. When nutrients were added, phytoplankton production (PPr) increased in both communities, but pelagic energy mobilization (PEM) and bacterial production (BP) only increased in the humic community. At low light conditions, the addition of nutrients led to increased PPr only in the humic community, suggesting that, in contrast to the clear water community, humic phytoplankton were already adapted to lower ambient light levels. Low light significantly reduced PPr and PEM in the clear water community, but without reducing total zooplankton production, which resulted in a doubling of food web efficiency (FWE = total zooplankton production/PEM). However, total zooplankton production was not correlated with PEM, PPr, BP, PPr:BP or C:nutrient stoichiometry for either community type. Therefore, other factors such as food chain length, food quality, ultra-violet radiation or duration of the experiment, must have determined total zooplankton production and ultimately FWE.




Similar content being viewed by others
References
Ahlgren G (1983) Comparison of methods for estimation of phytoplankton carbon. Arch Hydrobiol 98:489–508
Andersson A, Jurgensone I, Rowe OF, Simonelli P, Bignert A, Lundberg E, Karlsson J (2013) Can humic water discharge counteract eutrophication in coastal waters? PLoS One 8(4):e61293. doi:10.1371/journal.pone.0061293
Ask J, Karlsson J, Persson L, Ask P, Bystrom P, Jansson M (2009) Terrestrial organic matter and light penetration: effects on bacterial and primary production in lakes. Limnol Oceanogr 54:2034–2040. doi:10.4319/lo.2009.54.6.2034
Berglund J, Muren U, Bamstedt U, Andersson A (2007) Efficiency of a phytoplankton-based and a bacteria-based food web in a pelagic marine system. Limnol Oceanogr 52:121–131
Bergström AK, Jansson M (2000) Bacterioplankton production in humic Lake Ortrasket in relation to input of bacterial cells and input of allochthonous organic carbon. Microb Ecol 39:101–115
Bērzinš B, Bertilsson J (1990) Occurrence of limnic microcrustaceans in relation to pH and humic content in Swedish water bodies. Hydrobiologia 199:65–71. doi:10.1007/bf00007834
Blackburn N, Hagström Å, Wikner J, Cuadros-Hansson R, Bjørnsen PK (1998) Rapid determination of bacterial abundance, biovolume, morphology, and growth by neural network-based image analysis. Appl Environ Microbiol 64:3246–3255
Bottrell HH, Duncan A, Gliwicz ZM, Grygierek E, Herzig A, Hillbrichtilkowska A, Kurasawa H, Larsson P, Weglenska T (1976) Review of some problems in zooplankton production studies. Norw J Zool 24:419–456
Carpenter SR, Cole JJ, Kitchell JF, Pace ML (1998) Variable productivity in whole-lake experiments: roles of dissolved organic carbon, phosphorus and grazing. Limnol Oceanogr 43:73–80
Cashman MJ, Wehr JD, Truhn K (2013) Elevated light and nutrients alter the nutritional quality of stream periphyton. Freshwater Biol 58:1447–1457. doi:10.1111/fwb.12142
Cole JJ, Carpenter SR, Kitchell JF, Pace ML (2002) Pathways of organic carbon utilization in small lakes: results from a whole-lake C-13 addition and coupled model. Limnol Oceanogr 47:1664–1675
Cole JJ, Carpenter SR, Kitchell J, Pace ML, Solomon CT, Weidel B (2011) Strong evidence for terrestrial support of zooplankton in small lakes based on stable isotopes of carbon, nitrogen, and hydrogen. Proc Natl Acad Sci USA 108:1975–1980. doi:10.1073/pnas.1012807108
Dahlgren K, Andersson A, Larsson U, Hajdu S, Bamstedt U (2010) Planktonic production and carbon transfer efficiency along a north-south gradient in the Baltic Sea. Mar Ecol Prog Ser 409:77–94. doi:10.3354/meps08615
del Giorgio PA, Cole JJ (1998) Bacterial growth efficiency in natural aquatic systems. Annu Rev Ecol Syst 29:503–541
Dickman EM, Newell JM, Gonzalez MJ, Vanni MJ (2008) Light, nutrients, and food-chain length constrain planktonic energy transfer efficiency across multiple trophic levels. Proc Natl Acad Sci USA 105:18408–18412. doi:10.1073/pnas.0805566105
Drakare S, Blomqvist P, Bergstrom AK, Jansson M (2002) Primary production and phytoplankton composition in relation to DOC input and bacterioplankton production in humic Lake Ortrasket. Freshwater Biol 47:41–52. doi:10.1046/j.1365-2427.2002.00779.x
Drakare S, Blomqvist P, Bergstrom AK, Jansson M (2003) Relationships between picophytoplankton and environmental variables in lakes along a gradient of water colour and nutrient content. Freshwater Biol 48:729–740
Dubinsky Z, Stambler N (2009) Photoacclimation processes in phytoplankton: mechanisms, consequences, and applications. Aquat Microb Ecol 56:163–176. doi:10.3354/ame01345
Evans CD, Jones TG, Burden A, Ostle N, Zielinski P, Cooper MDA, Peacock M, Clark JM, Oulehle F, Cooper D, Freeman C (2012) Acidity controls on dissolved organic carbon mobility in organic soils. Glob Change Biol 18:3317–3331. doi:10.1111/j.1365-2486.2012.02794.x
Faithfull CL, Bergstrom AK, Vrede T (2011a) Effects of nutrients and physical lake characteristics on bacterial and phytoplankton production: a meta-analysis. Limnol Oceanogr 56:1703–1713. doi:10.4319/lo.2011.56.5.1703
Faithfull CL, Wenzel A, Vrede T, Bergstrom AK (2011b) Testing the light: nutrient hypothesis in an oligotrophic boreal lake. Ecosphere 2 doi:10.1890/es11-00223.1
Forsström L, Roiha T, Rautio M (2013) Responses of microbial food web to increased allochthonous DOM in an oligotrophic subarctic lake. Aquat Microb Ecol 68:171–184. doi:10.3354/ame01614
Geddes P (2009) Decoupling carbon effects and UV protection from terrestrial subsidies on pond zooplankton. Hydrobiologia 628:47–66. doi:10.1007/s10750-009-9745-5
Ha SY, Joo HM, Kang SH, Ahn IY, Shin KH (2014) Effect of ultraviolet irradiation on the production and composition of fatty acids in plankton in a sub-Antarctic environment. J Oceanogr 70:1–10. doi:10.1007/s10872-013-0207-3
Hansson L-A, Nicolle A, Graneli W, Hallgren P, Kritzberg E, Persson A, Bjork J, Nilsson PA, Bronmark C (2013) Food-chain length alters community responses to global change in aquatic systems. Nat Clim Change 3:228–233. doi:10.1038/nclimate1689
Hessen DO, Blomqvist P, Dahl-Hansen G, Drakare S, Lindstrom ES (2004) Production and food web interactions of Arctic freshwater plankton and responses to increased DOC. Arch Hydrobiol 159:289–307
Holtgrieve GW, Schindler DE, Hobbs WO, Leavitt PR, Ward EJ, Bunting L, Chen G, Finney BP, Gregory-Eaves I, Holmgren S, Lisac MJ, Lisi PJ, Nydick K, Rogers LA, Saros JE, Selbie DT, Shapley MD, Walsh PB, Wolfe AP (2011) A coherent signature of anthropogenic nitrogen deposition to remote watersheds of the Northern Hemisphere. Science 334:1545–1548. doi:10.1126/science.1212267
IPCC (2007) Climate change 2007: synthesis report. Intergovernmental panel on climate change, fourth assessment report. Cambridge University Press, Cambridge, UK, pp 1–23
Jäger CG, Diehl S, Schmidt GM (2008) Influence of water-column depth and mixing on phytoplankton biomass, community composition, and nutrients. Limnol Oceanogr 53:2361–2373
Jansson M, Bergstrom AK, Blomqvist P, Drakare S (2000) Allochthonous organic carbon and phytoplankton/bacterioplankton production relationships in lakes. Ecology 81:3250–3255
Jansson M, Karlsson J, Blomqvist P (2003) Allochthonous organic carbon decreases pelagic energy mobilization in lakes. Limnol Oceanogr 48:1711–1716
Kalff J (2003) Limnology: inland water ecosystems. Prentice-Hall, New Jersey
Karlsson J, Jansson M, Jonsson A (2002) Similar relationships between pelagic primary and bacterial production in clearwater and humic lakes. Ecology 83:2902–2910
Karlsson J, Bystrom P, Ask J, Ask P, Persson L, Jansson M (2009) Light limitation of nutrient-poor lake ecosystems. Nature 460:506–580. doi:10.1038/nature08179
Kissman CEH, Williamson CE, Rose KC, Saros JE (2013) Response of phytoplankton in an alpine lake to inputs of dissolved organic matter through nutrient enrichment and trophic forcing. Limnol Oceanogr 58:867–880. doi:10.4319/lo.2013.58.3.0867
Klug JL (2005) Bacterial response to dissolved organic matter affects resource availability for algae. Can J Fish Aquat Sci 62:472–481. doi:10.1139/f04-229
Kreutzweiser DP, Hazlett PW, Gunn JM (2008) Logging impacts on the biogeochemistry of boreal forest soils and nutrient export to aquatic systems: a review. Environ Rev 16:157–179. doi:10.1139/a08-006
Lefebure R, Degerman R, Andersson A, Larsson S, Eriksson LO, Bamstedt U, Bystrom P (2013) Impacts of elevated terrestrial nutrient loads and temperature on pelagic food-web efficiency and fish production. Glob Change Biol 19:1358–1372. doi:10.1111/gcb.12134
Ma Z, Li W, Shen A, Gao K (2013) Behavioral responses of zooplankton to solar radiation changes: in situ evidence. Hydrobiologia 711:155–163. doi:10.1007/s10750-013-1475-z
Martinez-Garcia S, Fernandez E, Alvarez-Salgado XA, Gonzalez J, Lonborg C, Maranon E, Moran XAG, Teira E (2010) Differential responses of phytoplankton and heterotrophic bacteria to organic and inorganic nutrient additions in coastal waters off the NW Iberian Peninsula. Mar Ecol Prog Ser 416:17–33. doi:10.3354/meps08776
Menzel DW, Corwin N (1965) The measurement of total phosphorus in seawater based on the liberation of organically bound fractions by persulfate oxidation. Limnol Oceanogr 10:280–282
Monteith DT, Stoddard JL, Evans CD, de Wit HA, Forsius M, Hogasen T, Wilander A, Skjelkvale BL, Jeffries DS, Vuorenmaa J, Keller B, Kopacek J, Vesely J (2007) Dissolved organic carbon trends resulting from changes in atmospheric deposition chemistry. Nature 450:537–539. doi:10.1038/nature06316
Nagata T (1986) Carbon and nitrogen content of natural planktonic bacteria. Appl Environ Microbiol 52:28–32
Persson J, Brett MT, Vrede T, Ravet JL (2007) Food quantity and quality regulation of trophic transfer between primary producers and a keystone grazer (Daphnia) in pelagic freshwater food webs. Oikos 116:1152–1163. doi:10.1111/j.2007.0030-1299.15639.x
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2014) Linear and non-linear mixed effects models. Package ‘nlme’ 3.1-177. http://CRAN.R-project.org/package=nlme
Qualls RG, Haines BL (1991) Fluxes of dissolved organic nutrients and humic substances in a deciduous forest. Ecology 72:254–266. doi:10.2307/1938919
R Core Team (2014) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. URL http://www.R-project.org/
Salonen K, Sarvala J, Hakala I, Viljanen M-L (1976) The relation of energy and organic carbon in aquatic invertebrates. Limnol Oceanogr 21:724–730
Sandberg J, Andersson A, Johansson S, Wikner J (2004) Pelagic food web structure and carbon budget in the northern Baltic Sea: potential importance of terrigenous carbon. Mar Ecol Prog Ser 268:13–29. doi:10.3354/meps268013
Smith DC, Azam F (1992) A simple, economical method for measuring bacterial protein sythesis rates in seawater using 3H-leucine. Marine Microbial Food Webs 6:107–114
Sobek S, Tranvik LJ, Prairie YT, Kortelainen P, Cole JJ (2007) Patterns and regulation of dissolved organic carbon: an analysis of 7,500 widely distributed lakes. Limnol Oceanogr 52:1208–1219
Steinberg CEW (2003) Ecology of humic substances in freshwaters. Springer, Berlin
Sterner RW, Elser JJ, Fee EJ, Guildford SJ, Chrzanowski TH (1997) The light:nutrient ratio in lakes: the balance of energy and materials affects ecosystem structure and process. Am Nat 150:663–684
Vadstein O (2000) Heterotrophic, planktonic bacteria and cycling of phosphorus—phosphorus requirements, competitive ability, and food web interactions. Adv Microb Ecol 16(16):115–167
Wenzel A, Bergstrom A-K, Jansson M, Vrede T (2012) Survival, growth and reproduction of Daphnia galeata feeding on single and mixed Pseudomonas and Rhodomonas diets. Freshwater Biol 57:835–846. doi:10.1111/j.1365-2427.2012.02751.x
Weyhenmeyer GA, Karlsson J (2009) Nonlinear response of dissolved organic carbon concentrations in boreal lakes to increasing temperatures. Limnol Oceanogr 54:2513–2519. doi:10.4319/lo.2009.54.6_part_2.2513
Wilkinson GM, Carpenter SR, Cole JJ, Pace ML, Yang C (2013) Terrestrial support of pelagic consumers: patterns and variability revealed by a multilake study. Freshwater Biol 58:2037–2049. doi:10.1111/fwb.12189
Wissel B, Boeing WJ, Ramcharan CW (2003) Effects of water color on predation regimes and zooplankton assemblages in freshwater lakes. Limnol Oceanogr 48:1965–1976
Wu CH, Dahms HU, Cheng SH, Hwang JS (2011) Effects of food and light on naupliar swimming behavior of Apocyclops royi and Pseudodiaptomus annandalei (Crustacea, Copepoda). Hydrobiologia 666:167–178. doi:10.1007/s10750-011-0631-6
Zellmer ID, Arts MT, Abele D, Humbeck K (2004) Evidence of sublethal damage in Daphnia (Cladocera) during exposure to solar UV radiation in subarctic ponds. Arct Antarct Alp Res 36:370–377
Acknowledgments
We thank Anneli Lagesson and Elsa Nilsson for help in the field and two anonymous reviewers for their comments on the manuscript. This study was part of the Lake Ecosystem Response to Environmental Change (LEREC) and was supported with grants from the Wallenberg foundation and the Swedish Research Council for Environment, Agricultural Sciences and Spatial Planning (Formas).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Ulrich Sommer.
Rights and permissions
About this article
Cite this article
Faithfull, C.L., Mathisen, P., Wenzel, A. et al. Food web efficiency differs between humic and clear water lake communities in response to nutrients and light. Oecologia 177, 823–835 (2015). https://doi.org/10.1007/s00442-014-3132-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-014-3132-2