Abstract
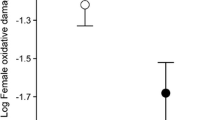
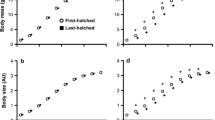
The early life period is characterized by fast growth and development, which can lead to high reactive oxygen species (ROS) production. Young animals thus have to balance their investment in growth versus ROS defence, and this balance is likely mediated by resource availability. Consequently resources transferred prenatally by the mother and nutritional conditions experienced shortly after birth may crucially determine the oxidative status of young animals. Here, we experimentally investigated the relative importance of pre- and early postnatal nutritional conditions on the oxidative status of great tit nestlings (Parus major). We show that resources transferred by the mother through the egg and nutritional conditions encountered after hatching affect the oxidative status of nestling in a sex-specific way. Daughters of non-supplemented mothers and daughters which did not receive extra food during the early postnatal period had higher oxidative damage than sons, while no differences between sons and daughters were found when extra food was provided pre- or postnatally. No effect of the food supplementations on growth, fledging mass or tarsus length was observed, indicating that female nestlings maintained their investment in growth at the expense of ROS defence mechanisms when resources were limited. The lower priority of the antioxidant defence system for female nestlings was also evidenced by lower levels of specific antioxidant components. These results highlight the important role of early parental effects in shaping oxidative stress in the offspring, and show that the sensitivity to these parental effects is sex-specific.


Similar content being viewed by others
References
Alan RR, McWilliams SR (2013) Oxidative stress, circulating antioxidants, and dietary preferences in songbirds. Comp Biochem Physiol B 164:185–193. doi:10.1016/j.cbpb.2012.12.005
Alberti A, Bolognini L, Macciantelli D, Caratelli M (2000) The radical cation of N,N-diethyl-para-paraphenylendiamine: a possible indicator of oxidative stress in biological samples. Res Chem Interm 26:253–267. doi:10.1163/156856700x00769
Alonso-Alvarez C, Bertrand S, Faivre B, Sorci G (2007) Increased susceptibility to oxidative damage as a cost of accelerated somatic growth in zebra finches. Funct Ecol 21:873–879
Arthur JR (2000) The glutathione peroxidases. Cell Mol Life Sci 57:1825–1835
Beaulieu M, Ropert-Coudert Y, Le Maho Y, Ancel A, Criscuolo F (2010) Foraging in an oxidative environment: relationship between delta-13C values and oxidative status in Adelie penguins. Proc R Soc B 277:1087–1092. doi:10.1098/rspb.2009.1881
Bindoli A, Fukuto JM, Forman HJ (2008) Thiol chemistry in peroxidase catalysis and redox signaling. Antiox Redox Signal 10:1549–1564. doi:10.1089/ars.2008.2063
Blouet C, Mariotti F, Azzout-Marniche D, Mathe V, Mikogami T, Tome D, Huneau JF (2007) Dietary cysteine alleviates sucrose-induced oxidative stress and insulin resistance. Free Radic Biol Med 42:1089–1097. doi:10.1016/j.freeradbiomed.2007.01.006
Bonatto F, Polydoro M, Andrades ME, da Frota Conte, Junior ML, Dal-Pizzol F, Rotta LN, Souza DO, Perry ML, Fonseca Moreira JC (2006) Effects of maternal protein malnutrition on oxidative markers in the young rat cortex and cerebellum. Neurosci Lett 406:281–284. doi:10.1016/j.neulet.2006.07.052
Buonocore G, Perrone S, Longini M, Terzuoli L, Bracci R (2000) Total hydroperoxide and advanced oxidation protein products in preterm hypoxic babies. Ped Res 47:221–224. doi:10.1203/00006450-200002000-00012
Carratelli M, Porcaro L, Ruscica M, De Simone E, Bertelli AAE, Corsi MM (2001) Reactive oxygen metabolites and prooxidant status in children with Down’s syndrome. Int J Clin Pharmacol 21:79–84
Catoni C, Peters A, Schaefer HM (2008a) Life history trade-offs are influenced by the diversity, availability and interactions of dietary antioxidants. Anim Behav 76:1107–1119. doi:10.1016/j.anbehav.2008.05.027
Catoni C, Schaefer HM, Peters A (2008b) Fruit for health: the effect of flavonoids on humoral immune response and food selection in a frugivorous bird. Funct Ecol 22:649–654. doi:10.1111/j.1365-2435.2008.01400.x
Christians JK (2002) Avian egg size: variation within species and inflexibility within individuals. Biol Rev 77:1–26. doi:10.1017/s1464793101005784
Clutton-Brock TH, Albon SD, Guinness FE (1985) Parental investment and sex-differences in juvenile mortality in birds and mammals. Nature 313:131–133
Cohen AA, McGraw KJ, Robinson WD (2009) Serum antioxidant levels in wild birds vary in relation to diet, season, life history strategy, and species. Oecologia 161:673–683. doi:10.1007/s00442-009-1423-9
Costantini D (2008) Oxidative stress in ecology and evolution: lessons from avian studies. Ecol Lett 11:1238–1251. doi:10.1111/j.1461-0248.2008.01246.x
Costantini D, Casagrande S, De Filippis S, Brambilla G, Fanfani A, Tagliavini J, Dell’Omo G (2006) Correlates of oxidative stress in wild kestrel nestlings (Falco tinnunculus). J Comp Physiol B 176:329–337. doi:10.1007/s00360-005-0055-6
Costantini D, Coluzza C, Fanfani A, Dell’Omo G (2007) Effects of carotenoid supplementation on colour expression, oxidative stress and body mass in rehabilitated captive adult kestrels (Falco tinnunculus). J Comp Physiol B 177:723–731. doi:10.1007/s00360-007-0169-0
Costantini D, Monaghan P, Metcalfe NB (2011) Biochemical integration of blood redox state in captive zebra finches (Taeniopygia guttata). J Exp Biol 214:1148–1152. doi:10.1242/jeb.053496
Cremers CM, Jakob U (2013) Oxidant sensing by reversible disulfide bond formation. J Biol Chem 288:26489–26496. doi:10.1074/jbc.R113.462929
De Coster G, De Neve L, Verhulst S, Lens L (2012) Maternal effects reduce oxidative stress in female nestlings under high parasite load. J Avian Biol 43:177–185. doi:10.1111/j.1600-048X.2012.05551.x
Dickinson DA, Forman HJ (2002) Cellular glutathione and thiols metabolism. Biochem Pharmacol 64:1019–1026. doi:10.1016/s0006-2952(02)01172-3
Dringen R, Pfeiffer B, Hamprecht B (1999) Synthesis of the antioxidant glutathione in neurons: supply by astrocytes of CysGly as precursor for neuronal glutathione. J Neurosci 19:562–569
Feoli AM, Siqueira IR, Almeida L, Tramontina AC, Vanzella C, Sbaraini S, Schweigert ID, Netto CA, Perry MLS, Goncalves CA (2006) Effects of protein malnutrition on oxidative status in rat brain. Nutrition 22:160–165. doi:10.1016/j.nut.2005.06.007
Freeman-Gallant CR, Amidon J, Berdy B, Wein S, Taff CC, Haussmann MF (2011) Oxidative damage to DNA related to survivorship and carotenoid-based sexual ornamentation in the common yellowthroat. Biol Lett 7:429–432. doi:10.1098/rsbl.2010.1186
Hall ME, Blount JD, Forbes S, Royle NJ (2010) Does oxidative stress mediate the trade-off between growth and self-maintenance in structured families? Funct Ecol 24:365–373. doi:10.1111/j.1365-2435.2009.01635.x
Halliwell B, Gutteridge JMC (2007) Free radicals in biology and medicine, 4th edn. Oxford University Press, Oxford
Handy DE, Loscalzo J (2012) Redox regulation of mitochondrial function. Antiox Redox Signal 16:1323–1367. doi:10.1089/ars.2011.4123
Hulbert AJ, Pamplona R, Buffenstein R, Buttemer WA (2007) Life and death: metabolic rate, membrane composition, and life span of animals. Physiol Rev 87:1175–1213. doi:10.1152/physrev.00047.2006
Hwangbo J, Hong EC, Jang A, Kang HK, Oh JS, Kim BW, Park BS (2009) Utilization of house fly-maggots, a feed supplement in the production of broiler chickens. J Environ Biol 30:609–614
Institute Inc. SAS (1989–2007) JMP statistical software version 10.0. SAS Institute, Cary, NC
Jensen K, Mayntz D, Toft S, Raubenheimer D, Simpson SJ (2011) Nutrient regulation in a predator, the wolf spider Pardosa prativaga. Anim Behav 81:993–999. doi:10.1016/j.anbehav.2011.01.035
Jones KS, Nakagawa S, Sheldon BC (2009) Environmental sensitivity in relation to size and sex in birds: meta-regression analysis. Am Nat 174:122–133. doi:10.1086/599299
Karadas F, Pappas AC, Surai PF, Speake BK (2005) Embryonic development within carotenoid-enriched eggs influences the post-hatch carotenoid status of the chicken. Comp Biochem Physiol B 141:244–251. doi:10.1016/j.cbpc.2005.04.001
Keller LF, Van Noordwijk AJ (1994) Effects of local environmental-conditions on nestling growth in the great tit Parus major L. Ardea 82:349–362
Lee S, Han KH, Nakamura Y, Kawakami S, Shimada K, Hayakawa T, Onoue H, Fukushima M (2013) Dietary l-Cysteine improves the antioxidative potential and lipid metabolism in rats fed a normal diet. Biosci Biotechnol Biochem 77:1430–1434. doi:10.1271/bbb.130083
Leeuwenburgh C, Ji LL (1995) Glutathione depletion in rested and exercised mice—biochemical consequences and adaptation. Arch Biochem Biophys 316:941–949. doi:10.1006/abbi.1995.1125
Lessells CM, Boag PT (1987) Unrepeatable repeatabilities: a common mistake. Auk 104:116–121
Lindström J (1999) Early development and fitness in birds and mammals. Trends Ecol Evol 14:343–348
Losdat S, Helfenstein F, Blount JD, Marri V, Maronde L, Richner H (2013) Nestling erythrocyte resistance to oxidative stress predicts fledging success but not local recruitment in a wild bird. Biol Lett 9:20120888
Metcalfe NB, Alonso-Alvarez C (2010) Oxidative stress as a life-history constraint: the role of reactive oxygen species in shaping phenotypes from conception to death. Funct Ecol 24:984–996. doi:10.1111/j.1365-2435.2010.01750.x
Monaghan P (2008) Early growth conditions, phenotypic development and environmental change. Philos Trans R Soc B 363:1635–1645. doi:10.1098/rstb.2007.0011
Monaghan P, Metcalfe NB, Torres R (2009) Oxidative stress as a mediator of life history trade-offs: mechanisms, measurements and interpretation. Ecol Lett 12:75–92. doi:10.1111/j.1461-0248.2008.01258.x
Mousseau TA, Fox CW (1998) Maternal effects as adaptations. Oxford University Press, New York
Naef-Daenzer B, Keller LF (1999) The foraging performance of great and blue tits (Parus major and P. caerulens) in relation to caterpillar development, and its consequences for nestling growth and fledging weight. J Anim Ecol 68:708–718. doi:10.1046/j.1365-2656.1999.00318.x
Noguera JC, Morales J, Perez C, Velando A (2010) On the oxidative cost of begging: antioxidants enhance vocalizations in gull chicks. Behav Ecol 21:479–484. doi:10.1093/beheco/arq005
Noguera JC, Kim SY, Velando A (2012) Pre-fledgling oxidative damage predicts recruitment in a long-lived bird. Biol Lett 8:61–63. doi:10.1098/rsbl.2011.0756
Norris K, Evans MR (2000) Ecological immunology: life history trade-offs and immune defense in birds. Behav Ecol 11:19–26
Oddie KR (2000) Size matters: competition between male and female great tit offspring. J Anim Ecol 69:903–912
Paglia DE, Valentine WN (1967) Studies on quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70:158–169
Pamplona R, Costantini D (2011) Molecular and structural antioxidant defenses against oxidative stress in animals. Am J Physiol Regul Integr Comp Physiol 301:R843–R863. doi:10.1152/ajpregu.00034.2011
Rollo CD, Carlson J, Sawada M (1996) Accelerated aging of giant transgenic mice is associated with elevated free radical processes. Can J Zool 74:606–620. doi:10.1139/z96-070
Saino N, Caprioli M, Romano M, Boncoraglio G, Rubolini D, Ambrosini R, Bonisoli-Alquati A, Romano A (2011) Antioxidant defenses predict long-term survival in a passerine bird. PLoS One 6:e19593. doi:10.1371/journal.pone.0019593
Sies H (1991) The role of reactive oxygen species in biological processes. Klin Wochenschr 69:965–968. doi:10.1007/bf01645140
Surai P, Fisinin VI (2013) Natural antioxidants in hens’ embryogenesis and antistress defences in postnatal development. Agric Biol 2:3–18
Thomas DW, Blondel J, Perret P, Lambrechts MM, Speakman JR (2001) Energetic and fitness costs of mismatching resource supply and demand in seasonally breeding birds. Science 291:2598–2600. doi:10.1126/science.1057487
Tschirren B, Fitze PS, Richner H (2003) Sexual dimorphism in susceptibility to parasites and cell-mediated immunity in great tit nestlings. J Anim Ecol 72:839–845
Visser ME, Adriaensen F, van Balen JH, Blondel J, Dhondt AA, van Dongen S, du Feu C, Ivankina EV, Kerimov AB, de Laet J, Matthysen E, McCleery R, Orell M, Thomson DL (2003) Variable responses to large-scale climate change in European Parus populations. Proc R Soc B 270:367–372. doi:10.1098/rspb.2002.2244
Acknowledgments
We thank Martina Keller, Kathrin Näpflin and Jamal Hanafi for field assistance and help in the lab and two anonymous reviewers for comments on the manuscript. The study was financially supported by the Swiss National Science Foundation (grant PP00P3_128386 B. T.). All procedures were conducted under licence from the Veterinary Office of the Canton of Zurich (195/2010) and the Federal Office for the Environment (324).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Ola Olsson.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Giordano, M., Costantini, D. & Tschirren, B. Sex-specific effects of prenatal and postnatal nutritional conditions on the oxidative status of great tit nestlings. Oecologia 177, 123–131 (2015). https://doi.org/10.1007/s00442-014-3100-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-014-3100-x