Abstract
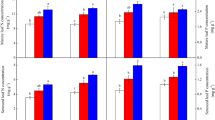
Plant resorption of multiple nutrients during leaf senescence has been established but stoichiometric changes among N, P and K during resorption and after fertilization are poorly understood. We anticipated that increased N supply would lead to further P limitation or co-limitation with N or K [i.e. P-(co)limitation], decrease N resorption and increase P and K resorption, while P and K addition would decrease P and K resorption and increase N resorption. Furthermore, Ca would accumulate while Mg would be resorbed during leaf senescence, irrespective of fertilization. We investigated the effect of N, P and K addition on resorption in two evergreen shrubs (Chamaedaphne calyculata and Rhododendron groenlandicum) in a long-term fertilization experiment at Mer Bleue bog, Ontario, Canada. In general, N addition caused further P-(co)limitation, increased P and K resorption efficiency but did not affect N resorption. P and K addition did not shift the system to N limitation and affect K resorption, but reduced P resorption proficiency. C. calyculata resorbed both Ca and Mg while R. groenlandicum resorbed neither. C. calyculata showed a higher resorption than R. groenlandicum, suggesting it is better adapted to nutrient deficiency than R. groenlandicum. Resorption during leaf senescence decreased N:P, N:K and K:P ratios. The limited response of N and K and the response of P resorption to fertilization reflect the stoichiometric coupling of nutrient cycling, which varies among the two shrub species; changes in species composition may affect nutrient cycling in bogs.



Similar content being viewed by others
References
Aerts R (1996) Nutrient resorption from senescing leaves of perennials: are there general patterns? J Ecol 84:597–608. doi:10.2307/2261481
Aerts R, Bobbink R (1999) The impact of atmospheric nitrogen deposition on vegetation processes in terrestrial, non-forest ecosystems. In: Langan SJ (ed) The impact of nitrogen deposition on natural and semi-natural ecosystems. Kluwer, the Netherlands, pp 85–122
Aerts R, Chapin FS (2000) The mineral nutrition of wild plants revisited: a re-evaluation of processes and patterns. Adv Ecol Res 30:1–67. doi:10.1016/S0065-2504(08)60016-1
Aerts R, Wallén B, Malmer N (1992) Growth-limiting nutrients in Sphagnum-dominated bogs subject to low and high atmospheric nitrogen supply. J Ecol 80:131–140. doi:10.2307/2261070
Anderson W, Eickmeier WG (2000) Nutrient resorption in Claytonia virginica L.: implications for deciduous forest nutrient cycling. Can J Bot 78:832–839. doi:10.1139/b00-056
Birk EM, Vitousek PM (1986) Nitrogen availability and nitrogen use efficiency in loblolly pine stands. Ecology 67:69–79. doi:10.2307/1938504
Bobbink R, Hornung M, Roelofs JGM (1998) The effects of air-borne nitrogen pollutants on species diversity in natural and semi-natural European vegetation. J Ecol 86:717–738. doi:10.1046/j.1365-2745.1998.8650717.x
Bragazza L, Tahvanainen T, Kutnar L, Rydin H, Limpens J, Hájek M, Grosvernier P, Hájek T, Hajkova P, Hansen I, Iacumin P, Gerdol R (2004) Nutritional constraints in ombrotrophic Sphagnum plants under increasing atmospheric nitrogen deposition in Europe. New Phytol 163:609–616. doi:10.1111/j.1469-8137.2004.01154.x
Bubier JL, Moore TR, Bledzki LA (2007) Effects of nutrient addition on vegetation and carbon cycling in an ombrotrophic bog. Global Change Biol 13:1168–1186. doi:10.1111/j.1365-2486.2007.01346.x
Bubier JL, Smith R, Juutinen S, Moore TR, Minocha R, Long S, Minocha S (2011) Effects of nutrient addition on leaf chemistry, morphology, and photosynthetic capacity of three bog shrubs. Oecologia 167:355–368. doi:10.1007/s00442-011-1998-9
Canadian Climate Normals (1971–2000) National climate data and information archive. http://climate.weatheroffice.ec.gc.ca/climate_normals. Accessed 01-April-2013
Chapin FS, Kedrowski RA (1983) Seasonal changes in nitrogen and phosphorus fractions and autumn re-translocation in evergreen and deciduous taiga trees. Ecology 64:376–391. doi:10.2307/1937083
Chapin FS, Moilanen L (1991) Nutritional controls over nitrogen and phosphorus resorption from Alaskan birch leaves. Ecology 72:709–715. doi:10.2307/2937210
Clarkson DT, Hanson JB (1980) The mineral nutrition of higher plants. Annu Rev Plant Physiol 31:239–298. doi:10.1146/annurev.pp.31.060180.001323
Diehl P, Mazzarino MJ, Funes F, Fontenla S, Gobbi M, Ferrari J (2003) Nutrient conservation strategies in native Andean-Patagonian forests. J Veg Sci 14:63–70. doi:10.1111/j.1654-1103.2003.tb02128.x
Eckstein RL, Karlsson PS, Weih M (1998) The significance of resorption of leaf resources for shoot growth in evergreen and deciduous woody plants from a subarctic environment. Oikos 81:567–575. doi:10.2307/3546777
Escudero A, Delarco JM, Sanz IC, Ayala J (1992) Effects of leaf longevity and re-translocation efficiency on the retention time of nutrients in the leaf biomass of different woody species. Oecologia 90:80–87. doi:10.1007/BF00317812
Feller IC, McKee KL, Whigham DF, O’Neill JP (2003) Nitrogen vs. phosphorus limitation across an ecotonal gradient in a mangrove forest. Biogeochemistry 62:145–175. doi:10.1023/A:1021166010892
Glaser PH (1992) Raised bogs in eastern North America—regional controls for species richness and floristic assemblages. J Ecol 80:535–554. doi:10.2307/2260697
Gorham E (1991) Northern peatlands: role in the carbon cycle and probable responses to climatic warming. Ecol Appl 1:182–195. doi:10.2307/1941811
Gunnarsson U, Rydin H (2000) Nitrogen fertilization reduces Sphagnum production in bog communities. New Phytol 147:527–537. doi:10.1046/j.1469-8137.2000.00717.x
Güsewell S (2005) Nutrient resorption of wetland graminoids is related to the type of nutrient limitation. Funct Ecol 19:344–354. doi:10.1111/j.1365-2435.2005.00967.x
Hoosbeek MR, Van Breemen N, Vasander H, Buttler A, Berendse F (2002) Potassium limits potential growth of bog vegetation under elevated atmospheric CO2 and N deposition. Global Change Biol 8:1130–1138. doi:10.1046/j.1365-2486.2002.00535.x
Karlsson PF (1994) The significance of internal nutrient cycling in branches for growth and reproduction of Rhododendron lapponicum. Oikos 70:191–200. doi:10.2307/3545630
Killingbeck KT (1986) The terminological jungle revisited: making a case for use of the term resorption. Oikos 46:263–264. doi:10.2307/3565477
Killingbeck KT (1996) Nutrients in senesced leaves: keys to the search for potential resorption and resorption proficiency. Ecology 77:1716–1727. doi:10.2307/2265777
Knops JMH, Koenig WD, Nash TH (1997) On the relationship between nutrient use efficiency and fertility in forest ecosystems. Oecologia 110:550–556. doi:10.2307/4221644
Kobe RK, Lepczyk CA, Iyer M (2005) Resorption efficiency decreases with increasing green leaf nutrients in a global data set. Ecology 86:2780–2792. doi:10.1890/04-1830
Lambers H, Chapin FS, Pons TL (1998) Plant physiological ecology. Springer, New York
Li XF, Zheng XB, Han SJ, Zheng JQ, Li TG (2010) Effects of nitrogen additions on nitrogen resorption and use efficiencies and foliar litterfall of six tree species in a mixed birch and poplar forest, northeastern China. Can J For Res 40:2256–2261. doi:10.1139/X10-167
Loneragan JF, Snowball K, Robson AD (1976) Remobilization of nutrients and its significance in plant nutrition. In: Wardlaw IF, Passioura JB (eds) Transport and transfer processes in plants. Academic Press, New York, pp 463–469
Moore T, Blodau C, Turunen J, Roulet N, Richard PJH (2004) Patterns of nitrogen and sulfur accumulation and retention in ombrotrophic bogs, eastern Canada. Global Change Biol 11:356–367. doi:10.1111/j.1365-2486.2004.00882.x
Murphy J, Riley JP (1962) A modified single solution method for determination of phosphate in natural waters. Anal Chim Acta 26:31–36. doi:10.1016/S0003-2670(00)88444-5
Nambiar EKS, Fife DN (1991) Nutrient re-translocation in temperate conifers. Tree Physiol 9:185–207. doi:10.1093/treephys/9.1-2.185
Näsholm T, Ekblad A, Nordin A, Giesler R, Högberg M, Högberg P (1998) Boreal forest plants take up organic nitrogen. Nature 392:914–916. doi:10.1038/31921
Negi GCS, Singh SP (1993) Leaf nitrogen dynamics with particular reference to re-translocation in evergreen and deciduous tree species of Kumaun Himalaya. Can J For Res 23:349–357. doi:10.1139/x93-051
Nordell KO, Karlsson PS (1995) Resorption of nitrogen and dry matter prior to leaf abscission: variation among individuals, sites and years in the mountain birch. Funct Ecol 9:326–333. doi:10.2307/2390581
Olde Venterink H, Wassen MJ, Verkroost AWM, de Ruiter PC (2003) Species richness-productivity patterns differ between N-, P-, and K-limited wetlands. Ecology 84:2191–2199. doi:10.1890/01-0639
Parkinson JA, Allen SE (1975) Wet oxidation procedure suitable for determination of nitrogen and mineral nutrients in biological material. Commun Soil Sci Plan 6:1–11. doi:10.1080/00103627509366539
Peri PL, Lasagno RG (2010) Biomass, carbon and nutrient storage for dominant grasses of cold temperate steppe grasslands in southern Patagonia, Argentina. J Arid Environ 74:23–34. doi:10.1016/j.jaridenv.2009.06.015
Pugnaire FI, Chapin FS (1993) Controls over nutrient resorption from leaves of evergreen Mediterranean species. Ecology 74:124–129. doi:10.2307/1939507
Ralhan PK, Singh SP (1987) Dynamics of nutrients and leaf mass in central Himalayan forest trees and shrubs. Ecology 68:1974–1983. doi:10.2307/1939888
Reader RJ (1980) Effects of nitrogen fertilizer, shade, and the removal of new growth on longevity of overwintering bog ericad leaves. Can J Bot 58:1737–1743. doi:10.1139/b80-201
Reed SC, Townsend AR, Davidson EA, Cleveland CC (2012) Stoichiometric patterns in foliar nutrient resorption across multiple scales. New Phytol 196:173–180. doi:10.1111/j.1469-8137.2012.04249.x
Rejmánková E (2005) Nutrient resorption in wetland macroPhytes: comparison across several regions of different nutrient status. New Phytol 167:471–482. doi:10.1111/j.1469-8137.2005.01449.x
Rustad LE, Campbell JL, Marion GM, Norby RJ, Mitchell MJ, Hartley AW, Cornelissen JHC, Gurevitch J, Gcte-News (2001) A meta-analysis of the response of soil respiration, net nitrogen mineralization, and aboveground plant growth to experimental ecosystem warming. Oecologia 126:543–562 doi: 10.1007/s004420000544
Rydin H, Jeglum JK (2006) The biology of peatlands. Oxford University Press, New York
Shipley B, Vile D, Garnier E, Wright IJ, Poorter H (2005) Functional linkages between leaf traits and net photosynthetic rate: reconciling empirical and mechanistic models. Funct Ecol 19:602–615. doi:10.1111/j.1365-2435.2005.01008.x
Small E (1972) Photosynthetic rates in relation to nitrogen recycling as an adaptation to nutrient deficiency in peat bog plants. Can J Bot 50:2227–2233. doi:10.1139/b72-289
Sterner RW, Elser J (2002) Ecological stoichiometry: the biology of elements from molecules to the biosphere. Princeton University Press, New Jersey
Straker CJ (1996) Ericoid mycorrhiza: ecological and host specificity. Mycorrhiza 6:215–225. doi:10.1007/s005720050129
Turunen J, Roulet NT, Moore TR, Richard PJH (2004) Nitrogen deposition and increased carbon accumulation in ombrotrophic peatlands in eastern Canada. Global Biogeochem Cycles 18:GB3002 doi: 10.1029/2003gb002154
van den Driessche R, Rieche K (1974) Prediction of mineral nutrient status of trees by foliar analysis. Bot Rev 40:347–394. doi:10.1007/BF02860066
van Heerwaarden LM, Toet S, Aerts R (2003a) Current measures of nutrient resorption efficiency lead to a substantial underestimation of real resorption efficiency: facts and solutions. Oikos 101:664–669. doi:10.1034/j.1600-0706.2003.12351.x
van Heerwaarden LM, Toet S, Aerts R (2003b) Nitrogen and phosphorus resorption efficiency and proficiency in six sub-arctic bog species after 4 years of nitrogen fertilization. J Ecol 91:1060–1070. doi:10.1046/j.1365-2745.2003.00828.x
Vergutz L, Manzoni S, Porporato A, Novais RF, Jackson RB (2012) Global resorption efficiencies and concentrations of carbon and nutrients in leaves of terrestrial plants. Ecol Monogr 82:205–220. doi:10.1890/11-0416.1
Verhoeven JTA, Keuter A, Logtestijn RV, Kerkhoven MB, Wassen M (1996) Control of local nutrient dynamics in mires by regional and climatic factors: a comparison of Dutch and Polish sites. J Ecol 84:647–656. doi:10.2307/2261328
Vitousek PM (1998) Foliar and litter nutrients, nutrient resorption, and decomposition in Hawaiian Metrosideros polymorpha. Ecosystems 1:401–407. doi:10.1007/s100219900033
Wright IJ, Westoby M (2003) Nutrient concentration, resorption and lifespan: leaf traits of Australian sclerophyll species. Funct Ecol 17:10–19. doi:10.1046/j.1365-2435.2003.00694.x
Yuan ZY, Chen HYH (2009a) Global-scale patterns of nutrient resorption associated with latitude, temperature and precipitation. Global Ecol Biogeogr 18:11–18. doi:10.1111/j.1466-8238.2008.00425.x
Yuan ZY, Chen HYH (2009b) Global trends in senesced-leaf nitrogen and phosphorus. Global Ecol Biogeogr 18:532–542. doi:10.1111/j.1466-8238.2009.00474.x
Yuan ZY, Li LH, Han XG, Huang JH, Jiang GM, Wan SQ, Zhang WH, Chen QS (2005) Nitrogen resorption from senescing leaves in 28 plant species in a semi-arid region of northern China. J Arid Environ 63:191–202. doi:10.1016/j.jaridenv.2005.01.023
Acknowledgments
We gratefully acknowledge the field and/or laboratory assistance of Angela Grant, Agnieszka Adamowicz-Walczak, Hélène Lalande and Mike Dalva and the comments of Jill Bubier. M. W. was financially supported by the Chinese Scholarship Council for his Ph.D. study and the research was funded by a Natural Sciences and Engineering Research Council Discovery Grant to T. R. M.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jason P. Kaye.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, M., Murphy, M.T. & Moore, T.R. Nutrient resorption of two evergreen shrubs in response to long-term fertilization in a bog. Oecologia 174, 365–377 (2014). https://doi.org/10.1007/s00442-013-2784-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-013-2784-7