Abstract
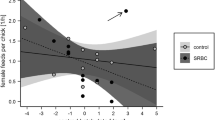
Maintenance of health and the production of offspring are competing processes that can result in trade-offs. As vertebrates invest substantial resources in their immune system, it is crucial to understand the interactions between immunity and reproductive strategies. In the lizard Zootoca vivipara, females have condition- and context-dependent mating strategies. We predicted that, if the risk of infection is higher for polyandrous females, then polyandrous females should invest more in immune system while monandrous females should invest more in reproduction. In order to test our prediction, we captured 62 gravid females of known age in a natural population; we kept them until parturition to access to their offspring. Then, using microsatellite marker-based paternity analyses within litters, we determine the mating strategy of females (monandrous or polyandrous). Females were also challenged with PHA to estimate their inflammatory response. Our results show that polyandrous females have a higher PHA response than the monandrous females, and that monandrous females produce more males and more juveniles of better body condition than polyandrous females. The relationship between mating behaviour and immune function may have consequences for females and may shape the evolution of mating systems.



Similar content being viewed by others
References
Ardia DR (2005) Tree swallows trade off immune function and reproductive effort differently across their range. Ecology 86:2040–2046
Blanckenhorn WU, Hosken DJ, Martin OY, Reim C, Teuschl Y, Ward PI (2002) The costs of copulating in the dung fly Sepsis cynipsea. Behav Ecol 13:353–358
Bleu J, Le Galliard JF, Meylan S, Massot M, Fitze PS (2011) Mating does not influence reproductive investment, in a viviparous lizard. J Exp Zool A Ecol Genet Physiol 315:458–464
Chamaille-Jammes S, Massot M, Aragon P, Clobert J (2006) Global warming and positive fitness response in mountain populations of common lizards Lacerta vivipara. Glob Change Biol 12:392–402
Cox RM, Calsbeek R (2011) An experimental test for alternative reproductive strategies underlying a female-limited polymorphism. J Evol Biol 24:343–353
Cox RM, Parker EU, Cheney DM, Liebl AL, Martin LB, Calsbeek R (2010) Experimental evidence for physiological costs underlying the trade-off between reproduction and survival. Funct Ecol 24:1262–1269
Dreiss AN, Cote J, Richard M, Federici P, Clobert J (2010) Age- and sex-specific response to population density and sex ratio. Behav Ecol 21:356–364
Evans JP, Magurran AE (2000) Multiple benefits of multiple mating in guppies. Proc Natl Acad Sci USA 97:10074–10076
Fitze PS, Le Galliard JF, Federici P, Richard M, Clobert J (2005) Conflict over multiple-partner mating between males and females of the polygynandrous common lizards. Evolution 59:2451–2459
Fitze PS, Cote J, Clobert J (2010) Mating order-dependent female mate choice in the polygynandrous common lizard Lacerta vivipara. Oecologia 162:331–341
Fortes P, Consoli FL (2011) Are there costs in the repeated mating activities of female Southern stink bugs Nezara viridula? Physiol Entomol 36:215–219
French SS, Johnston GIH, Moore MC (2007) Immune activity suppresses reproduction in food-limited female tree lizards Urosaurus ornatus. Funct Ecol 21:1115–1122
French SS, Moore MC, Demas GE (2009) Ecological immunology: the organism in context. Integr Comp Biol 49:246–253
Hamilton WD, Zuk M (1982) Heritable true fitness and bright birds—a role for parasites. Science 218:384–387
Heulin B (1988) Note on oviparous populations of Lacerta-vivipara. C R Acad Sci Ser III 306:63–68
Huey RB, Dunham AE, Overall KL, Newman RA (1990) Variation in locomotor performance in demographically known populations of the lizard Sceloporus-merriami. Physiol Zool 63:845–872
Ilmonen P, Taarna T, Hasselquist D (2000) Experimentally activated immune defence in female pied flycatchers results in reduced breeding success. Proc R Soc Lond B 267:665–670
Jennions MD, Petrie M (2000) Why do females mate multiply? A review of the genetic benefits. Biol Rev Camb Philos Soc 75:21–64
Kalinowski ST, Taper ML, Marshall TC (2007) Revising how the computer program CERVUS accommodates genotyping error increases success in paternity assignment. Mol Ecol 16:1099–1106
Kokko H, Rankin DJ (2006) Lonely hearts or sex in the city? Density-dependent effects in mating systems. Philos Trans R Soc Lond B 361:319–334
Kokko H, Ranta E, Ruxton G, Lundberg P (2002) Sexually transmitted disease and the evolution of mating systems. Evolution 56:1091–1100
Laloi D, Richard M, Lecomte J, Massot M, Clobert J (2004) Multiple paternity in clutches of common lizard Lacerta vivipara: data from microsatellite markers. Mol Ecol 13:719–723
Laloi D, Richard M, Federici P, Clobert J, Teillac-Deschamps P, Massot M (2009) Relationship between female mating strategy, litter success and offspring dispersal. Ecol Lett 12:823–829
Le Galliard JF, Massot M, Landys MM, Meylan S, Clobert J (2006) Ontogenic sources of variation in sexual size dimorphism in a viviparous lizard. J Evol Biol 19:690–704
Lecomte J, Clobert J, Massot M (1992) Sex identification in juveniles of Lacerta vivipara. Amphibia-Reptilia 13:21–25
Lochmiller RL, Deerenberg C (2000) Trade-offs in evolutionary immunology: just what is the cost of immunity? Oikos 88:87–98
Magnhagen C (1991) Predation risk as a cost of reproduction. Trends Ecol Evol 6:183–185
Maklakov AA, Bilde T, Lubin Y (2005) Sexual conflict in the wild: elevated mating rate reduces female lifetime reproductive success. Am Nat 165:S38–S45
Martin LB, Weil ZM, Nelson RJ (2007) Immune defense and reproductive pace of life in Peromyscus mice. Ecology 88:2516–2528
Massot M, Clobert J (2000) Processes at the origin of similarities in dispersal behaviour among siblings. J Evol Biol 13:707–719
Massot M, Clobert J, Pilorge T, Lecomte J, Barbault R (1992) Density dependence in the common lizard—demographic consequences of a density manipulation. Ecology 73:1742–1756
Massot M, Clobert J, Lecomte J, Barbault R (1994) Incumbent advantage in common lizards and their colonizing ability. J Anim Ecol 63:431–440
Massot M, Clobert J, Ferriere R (2008) Climate warming, dispersal inhibition and extinction risk. Glob Change Biol 14:461–469
Massot M, Clobert J, Montes-Poloni L, Haussy C, Cubo J, Meylan S (2011) An integrative study of ageing in a wild population of common lizards. Funct Ecol 25:848–858
Meylan S, Haussy C, Voituron Y (2010) Physiological actions of corticosterone and its modulation by an immune challenge in reptiles. Gen Comp Endocrinol 169:158–166
Mills SC et al (2009) Testosterone-mediated effects on fitness-related phenotypic traits and fitness. Am Nat 173:475–487
Munoz FA, Estrada-Parra S, Romero-Rojas A, Work TM, Gonzalez-Ballesteros E, Estrada-Garcia I (2009) Identification of CD3+ T lymphocytes in the green turtle Chelonia mydas. Vet Immunol Immunopathol 131:211–217
Nordling D, Andersson M, Zohari S, Gustafsson L (1998) Reproductive effort reduces specific immune response and parasite resistance. Proc R Soc Lond B 265:1291–1298
Norris K, Evans MR (2000) Ecological immunology: life history trade-offs and immune defense in birds. Behav Ecol 11:19–26
Promislow DEL (1991) Senescence in natural-populations of mammals—a comparative-study. Evolution 45:1869–1887
Raberg L, Nilsson JA, Ilmonen P, Stjernman M, Hasselquist D (2000) The cost of an immune response: vaccination reduces parental effort. Ecol Lett 3:382–386
Richard M, Lecomte J, de Fraipont M, Clobert J (2005) Age-specific mating strategies and reproductive senescence. Mol Ecol 14:3147–3155
Richard M, Losdat S, Lecomte J, de Fraipont M, Clobert J (2009) Optimal level of inbreeding in the common lizard. Proc R Soc Lond B 276:2779–2786
Smits JE, Bortolotti GR, Tella JL (1999) Simplifying the phytohaemagglutinin skin-testing technique in studies of avian immunocompetence. Funct Ecol 13:567–572
Tella JL, Lemus JA, Carrete M, Blanco G (2008) The PHA test reflects acquired T-cell mediated immunocompetence in birds. PLoS One 3:e3295
Thrall PH, Antonovics J, Bever JD (1997) Sexual transmission of disease and host mating systems: within-season reproductive success. Am Nat 149:485–506
Trivers RL, Willard DE (1973) Natural-selection of parental ability to vary sex-ratio of offspring. Science 179:90–92
Uller T, Olsson M (2008) Multiple paternity in reptiles: patterns and processes. Mol Ecol 17:2566–2580
Uller T, Isaksson C, Olsson M (2006) Immune challenge reduces reproductive output and growth in a lizard. Funct Ecol 20:873–879
Vinkler M, Bainova H, Albrecht T (2010) Functional analysis of the skin-swelling response to phytohaemagglutinin. Funct Ecol 24:1081–1086
White J, Richard M, Massot M, Meylan S (2011) Cloacal bacterial diversity increases with multiple mates: evidence of sexual transmission in female common lizards. PLoS One 6:e22339
Yasui Y (1998) The ‘genetic benefits’ of female multiple mating reconsidered. Trends Ecol Evol 13:246–250
Acknowledgments
The authors are grateful to all students and colleagues who helped in collecting data over the years, the Office National des Forêts and the Parc National des Cévennes for field work facilities. This work was supported by the CNRS, the Ministère de la Recherche et des Nouvelles Technologies (Programmes ORE and ANR), and the Institut d’Ecologie IFR 101. The study was carried out in accordance with institutional guidelines, under licence from the French Ministry of the Environment and under permit from the Seine-et-Marne Veterinary department (permits number 77-02).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Raoul Van Damme.
Rights and permissions
About this article
Cite this article
Richard, M., Massot, M., Clobert, J. et al. Litter quality and inflammatory response are dependent on mating strategy in a reptile. Oecologia 170, 39–46 (2012). https://doi.org/10.1007/s00442-012-2282-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-012-2282-3