Abstract
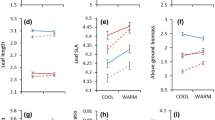
Variation in freezing severity is hypothesized to have influenced the distribution and evolution of the warm desert evergreen genus Larrea. If this hypothesis is correct, performance and survival of species and populations should vary predictably along gradients of freezing severity. If freezing environment changes in the future, the ability of Larrea to adapt will depend on the structure of variation for freezing resistance within populations. To test whether freezing responses vary among and within Larrea populations, we grew maternal families of seedlings from high and low latitude L. divaricata and high latitude L. tridentata populations in a common garden. We measured survival, projected plant area and dark-adapted chlorophyll fluorescence (F v /F m) before and after cold acclimation and for 2 weeks following a single freeze. We detected significant variation in freezing resistance among species and populations. Maternal family lines differed significantly in their responses to cold acclimation and/or freezing for two out of the three populations: among L. tridentata maternal families and among low latitude L. divaricata maternal families. There were no significant differences across maternal families of high latitude L. divaricata. Our results indicate that increased freezing resistance in high latitude populations likely facilitated historical population expansion of both species into colder climates, but this may have occurred to a greater extent for L. tridentata than for L. divaricata. Differences in the structure of variation for cold acclimation and freezing responses among populations suggest potential differences in their ability to evolve in response to future changes in freezing severity.




Similar content being viewed by others
References
Adams WW III, Demmig-Adams B (2004) Chlorophyll fluorescence as a tool for monitoring plant response to the environment. Springer, Massachusetts
Agrawal AA, Conner JK, Stinchcombe JR (2004) Evolution of plant resistance and tolerance to frost damage. Ecol Lett 7:1199–1208. doi:10.1111/j.1461-0248.2004.00680.x
Beatley JC (1974) Phenological events and their environmental triggers in Mojave Desert ecosystems. Ecology 55:856–863
Beaulieu JM, Leitch IJ, Patel S, Pendharkar A, Knight CA (2008) Genome size is a strong predictor of cell size and stomatal density in angiosperms. New Phytol 179:975–986. doi:10.1111/j.1469-8137.2008.02528.x
Benowicz A, Guy RD, El-Kassaby YA (2000) Geographic pattern of genetic variation in photosynthetic capacity and growth in two hardwood species from British Columbia. Oecologia 123:168–174
Boorse GC, Bosma TL, Meyer A-C, Ewers FW, Davis SD (1998) Comparative methods of estimating freezing temperatures and freezing injury in leaves of chaparral shrubs. Int J Plant Sci 159:513–521
Castro-Diez P, Puyravaud JP, Cornelissen JHC, Villar-Salvador P (1998) Stem anatomy and relative growth rate in seedlings of a wide range of woody plant species and types. Oecologia 116:57–66
Clausen J, Keck DD, Hiesey WM (1941) Regional differentiation in plant species. Am Nat 75:231–250
Coop JD, Givnish TJ (2008) Constraints on tree seedling establishment in montane grasslands of the Valles Caldera, New Mexico. Ecology 89:1101–1111. doi:10.1890/06-1333.1
Cortes MC, Hunziker JH (1997) Isozymes in Larrea divaricata and Larrea tridentata (Zygophyllaceae): a study of two amphitropical vicariants and autopolyploidy. Genetica 101:115–124
Davis SD, Sperry JS, Hacke UG (1999) The relationship between xylem conduit diameter and cavitation caused by freezing. Am J Bot 86:1367–1372
Demmig-Adams B, Adams WW III (2006) Photoprotection in an ecological context: the remarkable complexity of thermal energy dissipation. New Phytol 172:11–21. doi:10.1111/j.1469-8137.2006.01835.x
Ehlert B, Hincha DK (2008) Chlorophyll fluorescence imaging accurately quantifies freezing damage and cold acclimation responses in Arabidopsis leaves. Plant Methods 4:12. doi:10.1186/1746-4811-4-12
Ewers FW, Lawson MC, Bowen TJ, Davis SD (2003) Freeze/thaw stress in Ceanothus of southern California chaparral. Oecologia 136:213–219. doi:10.1007/s00442-003-1273-9
Ezcurra E, Montaña C, Arizaga S (1991) Architecture, light interception and distribution of Larrea species in the Monte Desert, Argentina. Ecology 72:23–34
Falconer DS, Mackay TFC (1996) Introduction to quantitative genetics, 4th edn. Pearson, Essex
Feng S, Hu Q (2004) Changes in agro-meteorological indicators in the contiguous United States: 1951–2000. Theor Appl Climatol 78:247–264. doi:10.1007/s00704-004-0061-8
Geber MA, Griffen LR (2003) Inheritance and natural selection on functional traits. Int J Plant Sci 164:S21. doi:10.1086/368233
Gutschick VP, BassiriRad H (2003) Extreme events as shaping physiology, ecology, and evolution of plants: toward a unified definition and evaluation of their consequences. New Phytol 160:21–42. doi:10.1046/j.1469-8137.2003.00866.x
Hacke UG, Sperry JS (2001) Functional and ecological xylem anatomy. Perspect Plant Ecol Evol Syst 4:97
Hiesey WM, Clausen J, Keck DD (1942) Relations between climate and intra-specific variation in plants. Am Nat 76:5–22
Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A (2005) Very high resolution interpolated climate surfaces for global land areas. Int J Climatol 25:1965–1978. doi:10.1002/joc.1276
Hunter KL, Betancourt JL, Riddle BR, Van Devender TR, Cole KL, Spaulding WG (2001) Ploidy race distributions since the Last Glacial Maximum in the North American desert shrub, Larrea tridentata. Glob Ecol Biogeogr 10:521–533
Hunziker JH, Palacios RA, de Valesi AG, Poggio L (1972) Species disjunctions in Larrea: evidence from morphology, cytogenetics, phenolic compounds, and seed albumins. Ann Mo Bot Gard 59:224–233
Hunziker JH, Palacios RA, Poggio L, Naranjo CA, Yang TW (1977) Geographic distribution, morphology, hybridization, cytogenics, and evolution. In: Mabry TJ, Hunziker JH, Difeo D (eds) Creosote bush: biology and chemistry of Larrea in New World deserts. Dowden, Hutchinson and Ross, Stroudsburg, pp 10–46
Johnsen O, Fossdal CG, Nagy N, Mølmann J, Dæhlen OG, Skrøppa T (2005) Climatic adaptation in Picea abies progenies is affected by the temperature during zygotic embryogenesis and seed maturation. Plant Cell Environ 28:1090–1102. doi:10.1111/j.1365-3040.2005.01356.x
Johnson DM, Germino MJ, Smith WK (2004) Abiotic factors limiting photosynthesis in Abies lasiocarpa and Picea engelmannii seedlings below and above the alpine timberline. Tree Physiol 24:377–386
Jump AS, Peñuelas J (2005) Running to stand still: adaptation and the response of plants to rapid climate change. Ecol Lett 8:1010–1020. doi:10.1111/j.1461-0248.2005.00796.x
Karl TR, Knight RW, Easterling DR, Quayle RG (1996) Indices of climate change for the United States. Bull Am Meteorol Soc 77:279–292
Limin AE, Fowler DB (2001) Inheritance of cell size in wheat (Triticum aestivum L.) and its relationship to the vernalization loci. Theor Appl Genet 103:277–281
Littell RC, Milliken GA, Stroup WW, Wolfinger RD, Schabenberger O (2006) SAS for mixed models, 2nd edn. SAS Institute, Cary
Mabry TJ, Hunziker JH, DiFeo DR (eds) (1977) Creosote bush: biology and chemistry of Larrea in New World deserts. Dowden, Hutchinson, & Ross, Stroudsburg
Maggio A et al (2002) Does proline accumulation play an active role in stress-induced growth reduction? Plant J 31:699–712. doi:10.1046/j.1365-313X.2002.01389.x
Maherali H, Walden AE, Husband BC (2009) Genome duplication and the evolution of physiological responses to water stress. New Phytol 184:721–731. doi:10.1111/j.1469-8137.2009.02997.x
Martínez-Vilalta J, Pockman WT (2002) The vulnerability to freezing-induced xylem cavitation of Larrea tridentata (Zygophyllaceae) in the Chihuahuan desert. Am J Bot 89:1916–1924
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence—a practical guide. J Exp Bot 51:659–668
McGee KP, Marshall DL (1993) Effects of variable moisture availability on seed germination in three populations of Larrea tridentata. Am Midl Nat 130:75–82
Medeiros JS, Pockman WT (2011) Drought increases freezing tolerance of both leaves and xylem of Larrea tridentata. Plant Cell Environ 34:43–51
Moore D (1989–2009) Sevilleta LTER meteorological data. In. http://sev.lternet.edu
Moore D (1999–2009) Sevilleta Larrea seedling monitoring study. In: http://sev.lternet.edu. http://sev.lternet.edu
Niering WA, Whittaker RH, Lowe CH (1963) The Saguaro: a population in relation to the environment. Science 142:15–23
Osmond CB (1994) What is photoinhibition? Some insights from comparisons of sun and shade plants. In: Baker NR, Bowyer JR (eds) Photoinhibition of photosynthesis: from molecular mechanisms to the field. BIOS, Oxford, pp 1–24
Pockman WT, Sperry JS (1997) Freezing-induced xylem cavitation and the northern limit of Larrea tridentata. Oecologia 109:19–27
Pockman WT, Sperry JS (2000) Vulnerability to xylem cavitation and the distribution of Sonoran desert vegetation. Am J Bot 87:1287–1299
Potvin C (2001) ANOVA: experimental layout and analysis. In: Scheiner SM, Gurevitch J (eds) Design and analysis of ecological experiments, 2nd edn. Oxford University Press, New York, pp 63–76
Rapp K, Junttila O (2000) Heritability estimates of winter hardiness in white clover based on field and laboratory experiments. Acta Agric Scand B Soil Plant Sci 50:143–148
Rossi BE, Debandi GO, Peralta IE, Palle EM (1999) Comparative phenology and floral patterns in Larrea species (Zygophyllaceae) in the Monte desert (Mendoza, Argentina). J Arid Environ 43:213
Ruosteenoja K, Carter TR, Jylha K, Tuomenvirta H (2003) Future climate in world regions: an intercomparison of model-based projections for the new IPCC emissions scenarios. In: vol 2010. Finnish Environment Institute, Helsinki http://www.environment.fi/download.asp?contentid=25835
Sakai A, Larcher W (1987) Frost survival of plants. Springer, Berlin
Stebbins GL (1971) Chromosomal evolution in higher plants. Edward Arnold Ltd, London
Tyler AP (2004) Regional variation in xylem anatomy of a wide-ranging desert shrub, Larrea tridentata. Master’s Thesis, University of New Mexico
Van Auken OW (2000) Shrub invasions of North America semiarid grasslands. Annu Rev Ecol Syst 31:197–215
Walker DJ, Romero P, Correal E (2010) Cold tolerance, water relations and accumulation of osmolytes in Bituminaria bituminosa. Biol Plant 54:293–298. doi:10.1007/s10535-010-0051-x
Wells PV, Hunziker JH (1976) Origin of Creosote bush (Larrea) deserts of Southwestern North America. Ann Mo Bot Gard 63:843–861
Wells PV, Shields LM (1964) Distribution of Larrea divaricata in relation to a temperature inversion at Yucca Flat, southern Nevada. Southwest Nat 9:51–55
Yang TW (1967) Ecotypic variation in Larrea divaricata. Am J Bot 54:1041–1044
Yang TW, Hunziker JH, Poggio L, Naranjo CA (1977) Hybridization between South American Jarilla and North American diploid Creosotebush (Larrea, Zygophyllaceae). Plant Syst Evol 126:331–346
Acknowledgments
The authors would like to thank R. Fernandez, F. Birrún, E. Fernandez and D. Ravetta for logistical support and G. Correo-Todesco, M. Barr-Lamas and G. Jaramillo for field assistance in Argentina. Thanks also to D. Hanson for the loan of equipment, J. Avritt for help with plant propagation and to two anonymous reviewers for helpful comments on the manuscript. Funding provided by the National Center for Research Resources (NCRR) (#P20RR18754), Graduate Summer Research Stipend from the Sevilleta Long Term Ecological Research site (#DEB-0080529 and #DEB-0217774), the E-MRGE NSF Graduate Teaching Fellows in K12 Education Program (#DGE-0538396) and the University of New Mexico Graduate Student Association’s Graduate Research Development fund. L. divaricata seeds imported into the USA under USDA import permit #3788757.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Frederick Meinzer.
Rights and permissions
About this article
Cite this article
Medeiros, J.S., Marshall, D.L., Maherali, H. et al. Variation in seedling freezing response is associated with climate in Larrea . Oecologia 169, 73–84 (2012). https://doi.org/10.1007/s00442-011-2181-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-011-2181-z