Abstract
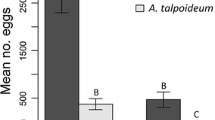
To predict the impacts of climate change on animal populations, we need long-term data sets on the effects of annual climatic variation on the demographic traits (growth, survival, reproductive output) that determine population viability. One frequent complication is that fecundity also depends upon maternal body size, a trait that often spans a wide range within a single population. During an eight-year field study, we measured annual variation in weather conditions, frog abundance and snake reproduction on a floodplain in the Australian wet-dry tropics. Frog numbers varied considerably from year to year, and were highest in years with hotter wetter conditions during the monsoonal season (“wet season”). Mean maternal body sizes, egg sizes and post-partum maternal body conditions of frog-eating snakes (keelback, Tropidonophis mairii, Colubridae) showed no significant annual variation over this period, but mean clutch sizes were higher in years with higher prey abundance. Larger females were more sensitive to frog abundance in this respect than were smaller conspecifics, so that the rate at which fecundity increased with body size varied among years, and was highest when prey availability was greatest. Thus, the link between female body size and reproductive output varied among years, with climatic factors modifying the relative reproductive rates of larger (older) versus smaller (younger) animals within the keelback population.


Similar content being viewed by others
References
Andrews RM (1991) Population stability of a tropical lizard. Ecology 72:1204–1217
Andrews RM, Wright SJ (1994) Long-term population fluctuations of a tropical lizard: a test of causality. In: Vitt LJ, Pianka ER (eds) Lizard ecology: historical and experimental perspectives. Princeton University Press, Princeton, NJ, pp 267–285
Bolton BL, Newsome AE, Merchant J (1982) Reproduction in the agile wallaby, Macropus agilis (Gould), in the tropical lowlands of the Northern Territory: opportunism in a seasonal environment. Aust J Ecol 7:261–277
Brown GP, Shine R (2002) Reproductive ecology of a tropical natricine snake, Tropidonophis mairii (Colubridae). J Zool Lond 258:63–72
Brown GP, Shine R (2006) Why do most tropical animals reproduce seasonally? Testing alternative hypotheses on the snake Tropidonophis mairii (Colubridae). Ecology 87:133–143
Brown GP, Shine R (2007) Repeatability and heritability of reproductive traits in free-ranging snakes. J Evol Biol 20:588–596
Brown GP, Shine R, Madsen T (2001) Responses of three sympatric snake species to tropical seasonality in northern Australia. J Trop Ecol 18:549–558
Bull JJ, Shine R (1979) Iteroparous animals that skip opportunities for reproduction. Am Nat 114:296–316
Cezilly F, Boy V, Green RE, Hirons GMJ, Johnson AR (1995) Interannual variation in Greater Flamingo breeding success in relation to water levels. Ecology 76:20–26
Cogger HG (2000) Reptiles and amphibians of Australia, 6th edn. Reed New Holland, Sydney
Du W, Ji X, Shine R (2005) Does body-volume constrain reproductive output in lizards? Biol Lett 1:98–100
Duellman WE, Trueb L (1986) Biology of amphibians. McGraw-Hill, New York
Fitch HS (1970) Reproductive cycles in lizards and snakes. Univ Kansas Mus Nat Hist Misc Publ 52:1–247
Forsman A (1996) Body size and net energy gain in gape-limited predators: a model. J Herpetol 30:307–319
Forsman A (1997) Growth and survival of Vipera berus in a variable environment. Symp Zool Soc Lond 70:143–145
Forsman A, Lindell L (1997) Responses of a predator to variation in prey abundance: survival and emigration of adders in relation to vole density. Can J Zool 75:1099–1108
Frith HJ, Davies SJJF (1961) Ecology of the magpie goose, Anseranas semipalmata Latham (Anatidae). Wildl Res 6:91–141
Gibbons JW, Greene JL (1990) Reproduction in the slider and other species of turtles. In: Gibbons JW (ed) Life history and ecology of the slider turtle. Smithsonian Institution Press, Washington, DC, pp 124–134
Kennett R (1999) Reproduction of two species of freshwater turtle, Chelodina rugosa and Elseya dentata, from the wet-dry tropics of northern Australia. J Zool Lond 247:457–473
Luiselli L, Capula M, Shine R (1997) Food habits, growth rates, and reproductive biology of grass snakes, Natrix natrix (Colubridae) in the Italian Alps. J Zool Lond 241:371–380
Madsen T, Shine R (1999a) The adjustment of reproductive threshold to prey abundance in a capital breeder. J Anim Ecol 68:571–580
Madsen T, Shine R (1999b) Rainfall and rats: climatically-driven dynamics of a tropical rodent population. Aust J Ecol 24:80–89
Madsen T, Shine R (2000) Rain, fish and snakes: climatically driven population dynamics of Arafura filesnakes in tropical Australia. Oecologia 124:208–215
Madsen T, Ujvari B, Shine R, Olsson M, Loman J (2006) Rain, rats and pythons: climate-driven population dynamics of predators and prey in tropical Australia. Austral Ecol 31:30–37
Malnate EV, Underwood G (1988) Australasian natricine snakes of the genus Tropidonophis. Proc Acad Nat Sci Philadelphia 140:59–201
Morton SR, Brennan KG (1991) Birds. In: Haynes CD, Ridpath MG, Williams MAJ (eds) Monsoonal Australia: landscape, ecology and man in the northern lowlands. AA Balkema, Rotterdam, pp 133–149
O’Shea M (1996) A guide to the snakes of Papua New Guinea. Independent Publishing, Port Moresby, Papua New Guinea
Owen-Smith N (1990) Demography of a large herbivore, the Greater Kudu Tragelaphus strepsiceros, in relation to rainfall. J Anim Ecol 59:893–913
Polis GA, Hurd SD, Jackson CT, Pinero FS (1997) El Niño effects on the dynamics and control of an island ecosystem in the Gulf of California. Ecology 78:1884–1897
Pough FH (1980) The advantages of ectothermy for tetrapods. Am Nat 115:92–112
Redhead TD (1979) On the demography of Rattus sordidus colletti in monsoonal Australia. Aust J Ecol 4:115–136
Ridpath MG (1985) Ecology in the wet-dry tropics: how different? Proc Ecol Soc Aust 13:3–20
Seigel RA, Fitch HS (1985) Annual variation in reproduction in snakes in a fluctuating environment. J Anim Ecol 54:497–505
Shine R (1991) Strangers in a strange land: ecology of the Australian colubrid snakes. Copeia 1991:120–131
Shine R (1992) Relative clutch mass and body shape in lizards and snakes: is reproductive investment constrained or optimized? Evolution 46:828–833
Shine R, Brown GP (2007) Adapting to the unpredictable: reproductive biology of vertebrates in the Australian wet-dry tropics. Phil Trans Roy Soc (in press)
Shine R, Greer AE (1991) Why are clutch sizes more variable in some species than in others? Evolution 45:1696–1706
Shine R, Madsen T (1997) Prey abundance and predator reproduction: rats and pythons on a tropical Australian floodplain. Ecology 78:1078–1086
Shine R, Harlow PS, Keogh JS, Boeadi (1998) The influence of sex and body size on food habits of a giant tropical snake, Python reticulatus. Funct Ecol 12:248–258
Singer FJ, Harting A, Symonds KK, Coughenour MB (1997) Density dependence, compensation, and environmental effects on calf mortality in Yellowstone National Park. J Wildl Manage 61:12–25
Smith GR, Ballinger RE, Rose BR (1995) Reproduction in Sceloporus virgatus from the Chiricahua Mountains of southeast Arizona with emphasis on annual variation. Herpetologica 51:342–349
Stewart M (1995) Climate driven population fluctuation in rain forest frogs. J Herpetol 29:437–446
Taylor JA, Tulloch D (1985) Rainfall in the wet-dry tropics: extreme events at Darwin and similarities between years during the period 1870–1983. Aust J Ecol 10:281–295
Tyler MJ (1989) Australian frogs. Viking O’Neill, Ringwood, VIC, Australia
Vitt LJ, Congdon JD (1978) Body shape, reproductive effort, and relative clutch mass in lizards: resolution of a paradox. Am Nat 112:595–608
Webb GJW, Buckworth R, Manolis SC (1983) Crocodylus johnstoni in the McKinlay River area, NT. III. Growth, movement and the population age structure. Aust Wildl Res 10:383–401
Wikelski M, Romero LM (2003) Body size, performance and fitness in Galapagos marine iguanas. Integr Comp Biol 43:376–386
Wikelski M, Thom C (2000) Marine iguanas shrink to survive El Niño. Nature 403:37–38
Williams CK, Newsome AE (1991) Adaptation in native mammals. In: Haynes CD, Ridpath MG, Williams MAJ (eds) Monsoonal Australia: landscape, ecology and man in the northern lowlands. AA Balkema, Rotterdam, pp 151–167
Acknowledgments
We thank Cathy Shilton, Ben Phillips and the other members of TeamBufo for advice and encouragement, Melanie Elphick for help with manuscript preparation, the managers and staff of Beatrice Hill Farm for logistical assistance, and the Australian Research Council for funding.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Anssi Laurila.
Rights and permissions
About this article
Cite this article
Brown, G.P., Shine, R. Rain, prey and predators: climatically driven shifts in frog abundance modify reproductive allometry in a tropical snake. Oecologia 154, 361–368 (2007). https://doi.org/10.1007/s00442-007-0842-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-007-0842-8