Abstract
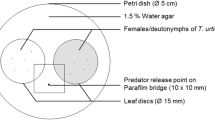
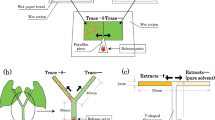
We studied whether volatiles released by putative host plants affect the antipredator response of an herbivorous mite, Tetranychus urticae, when the patch was invaded by Phytoseiulus persimilis. Tetranychus urticae laid a lower number of eggs on tomato leaves than on lima bean leaves, suggesting that lima bean is a preferred host food source for T. urticae. In addition, T. urticae preferred lima bean plant volatiles to tomato plant volatiles in a Y-tube olfactometer test. To investigate the antipredator response of T. urticae, we examined the migration of T. urticae from a lima bean leaf disc to a neighbouring leaf disc (either a tomato or lima bean leaf disc) when ten predators were introduced into the original lima bean disc. A Parafilm bridge allowed for migration between the leaf discs. No migrations occurred between leaf discs when there were no predators introduced to the original leaf disc. However, when predators were introduced migrations did occur. When the neighbouring leaf disc was upwind of the original disc, the migration rate of the mite from original lima bean leaf disc to a neighbouring tomato leaf disc was significantly lower than that to a neighbouring lima bean leaf disc. By contrast, when the neighbouring leaf disc was downwind of the original leaf disc, there was no difference in the migration rates between lima bean leaf discs and tomato leaf discs. The number of T. urticae killed by P. persimilis for each treatment was not different, and this clearly shows that the danger was the same in all treatments regardless of the decision made by T. urticae. From these results, we conclude that T. urticae change their antipredator response by evaluating the difference in host plant volatiles in the patch they inhabit.


Similar content being viewed by others
References
Abrahams M, Dill LM (1989) A determination of the energetic equivalence of the risk of predation. Ecology 70:999–1007
Aina OJ, Rodriguez JG, Knavel DE (1972) Characterizing resistance to Tetranychus urticae in tomato. J Econ Entomol 65:641–643
Athias-Binche F (1993) Dispersal in varying environments: the case of phoretic uropodid mites. Can J Zool 71:1793–1798
Bernays EA, Chapman RF (1994) Host-plant selection by phytophagous insects. Chapman and Hall, London
Blackwood JS, Schausberger P, Croft BA (2001) Prey-stage preference in generalist and specialist phytoseiid mites (Acari: Phytoseiidae) when offered Tetranychus urticae (Acari: Tetranychidae) eggs and larvae. Environ Entomol 30:1103–1111
Bolland H, Gutierrez J, Flechtmann C (1998) World catalogue of the spider mite family. Brill, Leiden
Chatzivasileiadis EA, Sabelis MW (1997) Toxicity of methyl ketones from tomato trichomes to Teranychus urticae Koch. Exp Appl Acarol 21:473–484
Chatzivasileiadis EA, Boon JJ, Sabelis MW (1999) Accumulation and turnover of 2-tridecanone in Teranychus urticae and its consequences for resistance of wild and cultivated tomatoes. Exp Appl Acarol 23:1011–1021
Dicke M (1986) Volatile spider-mite pheromone and host-plant kairomone, involved in spaced-out gregariousness in the spider mite Tetranychus urticae. Physiol Entomol 11:251–262
Dicke M (2000) Chemical ecology of host-plant selection by herbivorous arthropods: a multiple perspectives. Biochem Syst Ecol 28:601–617
Grand TC, Dill LM (1997) The energetic equivalence of cover to juvenile coho salmon (Oncorhynchus kisutch): ideal free distribution theory applied. Behav Ecol 8:437–447
Grostal P, Dicke M (1999) Direct and indirect cues of predation risk influence behavior and reproduction of prey: a case for acarine interactions. Behav Ecol 10:422–427
Holte AE, Houck MA, Collie NC (2001) Potential role of parasitism in the evolution of mutualism in astigmatid mites: Hemisarcoptes cooremani as a model. Exp Appl Acarol 25:97–107
Horiuchi J, Arimura G, Ozawa R, Shimoda T, Dicke M, Takabayashi J, Nishioka T (2003) A comparison of the response of Tetranychus urticae (Acari: Tetranychidae) and Phytoseiulus persimilis (Acari: Phytoseiidae) to volatiles emitted from lima bean leaves with different levels of damage made by T. urticae or Spodoptera exigua (Lepidoptera: Noctuidae). Appl Entomol Zool 38:365–368
Kishida O, Nishimura K (2004) Bulgy tadpoles: inducible defense morph. Oecologia 140:414–421
Kondo A, Takafuji A (1985) Resource utilization pattern of two species of tetranychid mites (Avarina: Tetranychidae). Res Popul Ecol 27:145–157
Li J, Margolies DC (1993) Quantitative genetics of aerial dispersal behaviour and life-history traits in Tetranychus urticae. Heredity 70: 544–552
Lima SL (1998) Stress and decision making under the risk of predation: recent developments from behavioral, reproductive, and ecological perspectives. Adv Study Behav 27:215–290
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68:619–640
Losey JE, Denno RF (1998a) The escape response of pea aphid to foliar-foraging predators: factors affecting dropping behaviour. Ecol Entomol 23:53–61
Losey JE, Denno RF (1998b) Interspecific variation in the escape responses of aphids: effect on risk of predation from foliar-foraging and ground-foraging predators. Oecologia 115:245–252
Magalhães S, Janssen A, Hanna R, Sabelis MW (2002) Flexible antipredator behaviour in herbivorous mites through vertical migration in a plant. Oecologia 132:143–149
Matsumoto T, Itioka T, Nishida T (2002) Fitness cost of parasitoid avoidance behavior in the arrowhead scale, Unaspis yanonensis Kuwana. Entomol Exp Appl 105:83–88
Pallini A, Janssen A, Sabelis MW (1999) Spider mites avoid plants with predators. Exp Appl Acarol 23:803–815
Relyea RA (2003) How prey respond to combined predators: a review and an empirical test. Ecology 84:1827–1839
Takabayashi J, Dicke M (1992) Response of predatory mites with different rearing histories to volatiles of uninfested plants. Entomol Exp Appl 64:187–193
Tollrian R (1995) Predator-induced morphological defenses: costs, life history shifts, and maternal effects in Daphnia pulex. Ecology 76:1691–1705
Tollrian R, Harvell CD (1999) The ecology and evolution of inducible defenses. Princeton University Press, Princeton
Villagra CA, Ramírez CC, Niemeyer HM (2002) Antipredator responses of aphids to parasitoids change as a function of aphid physiological state. Anim Behav 64:677–683
Visser JH (1986) Host odor perception in phytophagous insects. Annu Rev Entomol 31:121–144
Yano S (2004) Does Tetranychus urticae (Acari: Tetranichidae) use flying insects as vectors for phoretic dispesal? Exp Appl Acarol 32:243–248
Acknowledgment
This study was supported by Core Research for Evolutional Science and Technology of the Japan Science and Technology Corporation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Stefan Scheu.
Rights and permissions
About this article
Cite this article
Choh, Y., Takabayashi, J. Predator avoidance in phytophagous mites: response to present danger depends on alternative host quality. Oecologia 151, 262–267 (2007). https://doi.org/10.1007/s00442-006-0590-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-006-0590-1