Abstract
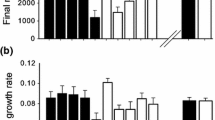
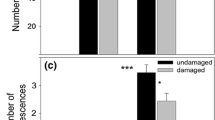
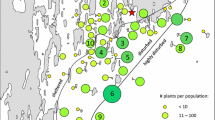
We investigated geographic patterns of herbivory and resource allocation to defense, growth, and reproduction in an invasive biennial, Alliaria petiolata, to test the hypothesis that escape from herbivory in invasive species permits enhanced growth and lower production of defensive chemicals. We quantified herbivore damage, concentrations of sinigrin, and growth and reproduction inside and outside herbivore exclusion treatments, in field populations in the native and invasive ranges. As predicted, unmanipulated plants in the native range (Hungary, Europe) experienced greater herbivore damage than plants in the introduced range (Massachusetts and Connecticut, USA), providing evidence for enemy release, particularly in the first year of growth. Nevertheless, European populations had consistently larger individuals than US populations (rosettes were, for example, eightfold larger) and also had greater reproductive output, but US plants produced larger seeds at a given plant height. Moreover, flowering plants showed significant differences in concentrations of sinigrin in the invasive versus native range, although the direction of the difference was variable, suggesting the influence of environmental effects. Overall, we observed less herbivory, but not increased growth or decreased defense in the invasive range. Geographical differences in performance and leaf chemistry appear to be due to variation in the environment, which could have masked evolved differences in allocation.




Similar content being viewed by others
References
Anderson R, Dhillion S, Kelley T (1996) Aspects of the ecology of an invasive plant, garlic mustard (Alliaria petiolata), in central Illinois. Restor Ecol 4:181–191
Bardon R, Countryman D, Hall R (1995) A reassessment of using light-sensitive diazo paper for measuring integrated light in the field. Ecology 76:1013–1016
Bazzaz F, Chiariello N, Coley P, Pitelka L (1987) Allocating resources to reproduction and defense. BioScience 37:58–67
Bernays EA, Oppenheim S, Chapman RF, Kwon H, Gould F (2000) Taste sensitivity of insect herbivores to deterrents is greater in specialists than in generalists: a behavioral test of the hypothesis with two closely related caterpillars. J Chem Ecol 26:547–563
Blossey B, Nuzzo V, Hinz HL, Gerber E (2001) Developing biological control of Alliaria petiolata (M. Bieb.) Cavara and Grande (garlic mustard). Nat Areas J 21:3570367
Blossey B, Nötzold R (1995) Evolution of increased competitive ability in invasive nonindigenous plants: a hypothesis. J Ecol 83:887–889
Booth E, Walker K, Griffiths D (1990) Effect of harvest date and pod position on glucosinolates in oilseed rape (Brassica napus). J Sci Food Agric 53:43–61
Bossdorf O, Prati D, Auge H, Schmid B (2004) Reduced competitive ability in an invasive plant. Ecol Lett 7:346–353
Byers D, Quinn J (1998) Demographic variation in Alliaria petiolata (Brassicaceae) in four contrasting habitats. J Torrey Bot Soc 125:138–149
Carson W, Root R (1999) Top-down effects of insect herbivores during early succession: influence on biomass and plant dominance. Oecologia 121:260–272
Cavers P, Heagy M, Kokron R (1979) The biology of Canadian weeds. 35. Alliaria petiolata (M. Bieb.) Cavara and Grande. Can J Plant Sci 59:217–229
Cipollini D (2002) Variation in the expression of chemical defenses in Alliaria petiolata (Brassicaceae) in the field and common garden. Am J Bot 89:1422–1430
Ciska E, Martyniak-Przybyszewska B, Kozlowska H (2000) Content of glucosinolates in cruciferous vegetables grown at the same site for two years under different climatic conditions. J Agric Food Chem 48:2862–2867
Clossais-Besnard N, Larher F (1991) Physiological role of glucosinolates in Brassica napus. Concentration and distribution pattern of glucosinolates among plant organs during a complete life cycle. J Sci Food Agric 56:25–38
Crawley M (1987) What makes a community invasible? In: Gray A, Crawley M, Edwards P (eds) Colonization, succession, and stability. Blackwell,, Oxford, pp 429–453
Cruden R, McClain A, Shrivastava G (1996) Pollination biology and breeding system of Alliaria petiolata (Brassicaceae). Bull Torrey Bot Club 123:273–280
Daehler C (1998) The taxonomic distribution of invasive angiosperm plants: ecological insights and comparison to agricultural weeds. Biol Conserv 84:167–180
Daehler C, Strong D (1997) Reduced herbivore resistance in introduced smooth cordgrass (Spartina alterniflora) after a century of herbivore-free growth. Oecologia 110:99–108
Daxenbichler M, Spencer G, Carlson D, Rose G, Brinker A, Powell R (1991) Glucosinolate composition of seeds from 297 species of wild plants. Phytochemistry 30:2623–2638
Elton C (2000) The ecology of invasions by animals and plants, 2nd edn. University of Chicago Press, Chicago
Feeny P, Rosenberry L (1982) Seasonal variation in the glucosinolate content of North American Brassica nigra and Dentaria species. Biochem Syst Ecol 10:23–32
Friend D (1961) A simple method of measuring integrated light values in the field. Ecology 42:577–580
Hacia H, Vitale CS, Arkin MA, McKittrick DA (1991) Climates of the World. In: National oceanic and atmospheric administration, Asheville, North Carolina, pp 40
Herms D, Mattson W (1992) The dilemma of plants: to grow or defend. Q Rev Biol 67:283–335
Huang X, Renwick J (1994) Relative activities of glucosinolates as oviposition stimulants for Pieris rapae and P. napi oleracea. J Chem Ecol 20:1025–1037
Hunter M, Schultz J (1995) Fertilization mitigates chemical induction and herbivore responses within damaged oak trees. Ecology 76:1226–1232
Jalas J, Suominen J (eds) (1994) Atlas Florae Europaeae: distribution of vascular plants in Europe. Committee for Mapping the Flora of Europe, Helsinki
Louda S, Collinge S (1992) Plant resistance to insect herbivores: a field test of the environmental stress hypothesis. Ecology 73:153–169
Louda S, Mole S (1991) Glucosinolates: chemistry and ecology. In: Rosenthal G, Berenbaum M (eds) Herbivores: their interactions with secondary plant metabolites, vol 1: The Chemical Participants, 2nd edn. Academic, San Diego, pp 123–164
Maron JL, Vilá M, Bommarco R, Elmendorf S, Beardsley P (2004) Rapid evolution of an invasive plant. Ecol Monogr 74:261–280
McGlynn T (1999) The worldwide transfer of ants: geographical distribution and ecological invasions. J Biogeogr 26:535–548
Meekins J, McCarthy B (1999) Competitive ability of Alliaria petiolata (garlic mustard, Brassicaceae), an invasive, nonindigenous forest herb. Int J Plant Sci 160:743–752
Mitchell C, Power A (2003) Release of invasive plants from fungal and viral pathogens. Nature 421:625–627
Nichols-Orians C, Schultz J (1990) Interactions among leaf toughness, chemistry, and harvesting by attine ants. Ecol Entomol 15:311–320
Nuzzo V (1993) Distribution and spread of the invasive biennial Alliaria petiolata (garlic mustard) in North America. In: McKnight B (ed) Biological pollution: the control and impact of invasive exotic species. Indiana Academy of Science, Indianapolis, pp 137–145
Pivnick K, Lamb R, Reed D (1992) Response of flea beetles, Phyllotreta spp., to mustard oils and nitriles in field trapping experiments. J Chem Ecol 18:863–873
Rejmánek M (2000) Invasive plants: approaches and predictions. Aust Ecol 25:497–506
Renwick JAA, Zhang W, Haribal M, Attygalle AB, Lopez KD (2001) Dual chemical barriers protect a plant against different larval stages of an insect. J Chem Ecol 27:1575–1583
Siemann E, Rogers W (2001) Genetic differences in growth of an invasive tree species. Ecol Lett 4:514–518
Soerensen H (1990) Glucosinolates- structure, properties, function. In: Shahidi F (ed) Canola and rapeseed. van Nostrand Reinhold, New York, pp 149–172
Szentesi Á (1990) Controversial components of plant apparency in Alliaria petiolata Cavara & Grande (Cruciferae). In: Szentesi Á, Jermy T (eds) Insects-plants ‘89, vol 39. Akadémiai Kiadó, Budapest, pp 237–244
Thébaud C, Simberloff D (2001) Are plants really larger in their introduced ranges? Am Nat 157:231–236
Torchin M, Lafferty K, Dobson A, McKenzie V, Kurls A (2003) Introduced species and their missing parasites. Nature 421:628–630
Traw MB, Dawson TE (2002) Reduced performance of two specialist herbivores (Lepidoptera: Pieridae, Coleoptera: Chrysomelidae) on new leaves of damaged black mustard plants. Environ Entomol 31:714–722
Troyer J, Stephenson K, Fahey J (2001) Analysis of glucosinolates from broccoli and other cruciferous vegetables by hydrophilic interaction liquid chromatography. J Chromatogr A 919:299–304
USDA, NRCS (2002) The PLANTS Database, Version 3.5. In, vol. 2003. National Plant Data Center, Baton Rouge, LA 70874–4490 USA
van Kleunen M, Schmid B (2003) No evidence for an evolutionary increased competitive ability in an invasive plant. Ecology 84:2816–2823
Vaughn S, Berhow M (1999) Allelochemicals isolated from tissues of the invasive weed garlic mustard (Alliaria petiolata). J Chem Ecol 25:2495–2504
Weatherbee P, Somers P, Simmons T (1998) A guide to invasive plants in Massachusetts. In. Massachusetts Biodiversity Initiative, Massachusetts Division of Fisheries and Wildlife, Boston
Williamson M, Fitter A (1996) The characters of successful invaders. Biol Conserv 78:163–170
Willis A, Thomas M, Lawton J (1999) Is the increased vigour of invasive weeds explained by a trade-off between growth and herbivore resistance? Oecologia 120:632–640
Willis AJ, Memmott J, Forrester RI (2000) Is there evidence for the post-invasion evolution of increased size among invasive plant species? Ecol Lett 3:275–283
Wolfe L (2002) Why alien invaders succeed: support for the escape-from-enemy hypothesis. Am Nat 160:705–711
Zangerl A, Bazzaz F (1992) Theory and pattern in plant defense allocation. In: Fritz R, Simms E (eds) Plant resistance to herbivores and pathogens: ecology, evolution and genetics. Chicago University Press, Chicago, pp 363–391
Acknowledgements
This work was funded by a NSF Graduate Student Fellowship and an EPA STAR Fellowship to KCL, as well as a Harvard OEB Student Dissertation Grant and the Massachusetts Natural Heritage and Endangered Species Program. Peter Ashton, Missy Holbrook, Brooke Parry-Hecht, Naomi Pierce, Bernhard Schmid, Amity Wilczek and Kelly Wolfe-Bellin and four anonymous reviewers provided helpful comments on previous versions of this manuscript. We thank the Duna-Ipoly and Bükk National Park directorates, Trustees of Reservations, and National Audubon, and Harvard Forest for use of research sites. Francie Chew provided assistance with the HPLC-MS column and chemical extraction protocol. We thank Sylvan Kaufman, János Nagy, András Schmotzer, Kristina Stinson, Rich Stomberg, and Zoltán Tuba for assistance with the field studies. Aaron Ellison provided feedback on statistical analyses, but any errors are the authors’ own. Special thanks to our research assistants: Dan Flynn, Daria Hinz, Julia Mansfield, and Luke McKneally. Samples were transported under USDA Permit # 37–83868.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Judith Bronstein
Rights and permissions
About this article
Cite this article
Lewis, K.C., Bazzaz, F., Liao, Q. et al. Geographic patterns of herbivory and resource allocation to defense, growth, and reproduction in an invasive biennial, Alliaria petiolata . Oecologia 148, 384–395 (2006). https://doi.org/10.1007/s00442-006-0380-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-006-0380-9