Abstract
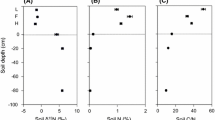
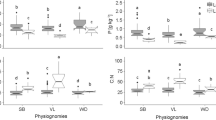
A large number of herbaceous and woody plants from tropical woodland, savanna, and monsoon forest were analysed to determine the impact of environmental factors (nutrient and water availability, fire) and biological factors (microbial associations, systematics) on plant δ15N values. Foliar δ15N values of herbaceous and woody species were not related to growth form or phenology, but a strong relationship existed between mycorrhizal status and plant δ15N. In woodland and savanna, woody species with ectomycorrhizal (ECM) associations and putative N2-fixing species with ECM/arbuscular (AM) associations had lowest foliar δ15N values (1.0–0.6‰), AM species had mostly intermediate δ15N values (average +0.6‰), while non-mycorrhizal Proteaceae had highest δ15N values (+2.9 to +4.1‰). Similar differences in foliar δ15N were observed between AM (average 0.1 and 0.2‰) and non-mycorrhizal (average +0.8 and +0.3‰) herbaceous species in woodland and savanna. Leguminous savanna species had significantly higher leaf N contents (1.8–2.5% N) than non-fixing species (0.9–1.2% N) indicating substantial N acquisition via N2 fixation. Monsoon forest species had similar leaf N contents (average 2.4% N) and positive δ15N values (+0.9 to +2.4‰). Soil nitrification and plant NO3 − use was substantially higher in monsoon forest than in woodland or savanna. In the studied communities, higher soil N content and nitrification rates were associated with more positive soil δ15N and plant δ15N. In support of this notion, Ficus, a high NO3 − using taxa associated with NO3 − rich sites in the savanna, had the highest δ15N values of all AM species in the savanna. δ15N of xylem sap was examined as a tool for studying plant δ15N relations. δ15N of xylem sap varied seasonally and between differently aged Acacia and other savanna species. Plants from annually burnt savanna had significantly higher δ15N values compared to plants from less frequently burnt savanna, suggesting that foliar 15N natural abundance could be used as marker for assessing historic fire regimes. Australian woodland and savanna species had low leaf δ15N and N content compared to species from equivalent African communities indicating that Australian biota are the more N depauperate. The largest differences in leaf δ15N occurred between the dominant ECM Australian and African savanna (miombo) species, which were depleted and enriched in 15N, respectively. While the depleted δ15N of Australian ECM species are similar to those of previous reports on ECM species in natural plant communities, the 15N-enriched δ15N of African ECM species represent an anomaly.





Similar content being viewed by others
References
Austin AT, Sala OE (1999) Foliar δ15N is negatively correlated with rainfall along with the IGBP transect in Australia. Aust J Plant Physiol 26:293–295
Bowman DMJS, Fensham RJ (1991) Response of a monsoon forest-savanna boundary to fire protection in Weipa, northern Australia. Aust J Ecol 16:111–118
Brock J (1988) Top end native plants. John Brook, Darwin, Australia
Cook GD (1994) The fate of nutrients during fires in a tropical savanna. Aust J Ecol 19:359–365
Cook GD (2001) Effects of frequent fires and grazing on stable nitrogen isotope ratios in northern Australia. Aust Ecol 26:630–636
Evans RD (2001) Physiological mechanisms influencing plant nitrogen isotope composition. Trends Plant Sci 6:121–126
Handley LL, Raven JA (1992) The use of natural abundance of N isotopes in plant physiology and ecology. Plant Cell Environ 15:965–985
Handley LL, Scrimgeour C (1997) Terrestrial plant ecology and 15N natural abundance: the present limits to interpretation for uncultivated systems with original data from a Scottish old field. Adv Ecol Res 27:133–212
Handley LL, Austin AT, Robinson D, Scrimgeour CM, Raven JA, Heaton THE, Schmidt S, Stewart GR (1999) The 15N natural abundance (δ15N) of ecosystem samples reflects measures of water availability. Aust J Plant Physiol 26:185–199
Hobbie EA, Macko SA, Williams W (2000) Correlations between foliar δ15N and nitrogen concentrations may indicate plant-mycorrhizal interactions. Oecologia 122:273–283
Högberg P (1990) 15N natural abundance as a possible marker of the ectomycorrhizal habit of trees in mixed African woodlands. New Phytol 115:483–486
Högberg P (1997) Tansley review no. 95. 15N natural abundance in soil-plant systems. New Phytol 137:179–203
Högberg P, Alexander IJ (1995) Roles of root symbioses in African woodland and forest: evidence from 15N abundance and foliar analysis. J Ecol 83:217–224
Högberg P, Högbom L, Schinkel H, Högberg M, Johannisson C, Wallmark H (1996) 15N natural abundance of surface soils, roots and mycorrhizas in profiles of European forest soils. Oecologia 108:207–214
Holt JA, Coventry RJ (1990) Nutrient cycling in Australian savanna. J Biogeogr 17:427–432
Mariotti A, Mariotti F, Amarger N, Pizelle G, N'Gambi JM, Champigny ML, Moyse A (1980) Fractionnement isotopique de l'azote lors des processsus d'absorption des nitrates et de la fixation de l'azote atmosphérique par les plantes. Physiol Veg 18:163–181
Michelsen A, Quarmby C, Sleep D, Jonasson S (1998) Vascular plan 15N natural abundance in heath and forest tundra ecosystems is closely correlated with presence and type of mycorrhizal fungi in roots. Oecologia 115:406–418
Mordelet P, Cook G, Abbadie L, Grably M, Mariotti A (1996) Natural 15N abundance of vegetation and soil in the Kapalga savanna, Australia. Aust J Ecol 21:336–340
Mott JJ, Williams J, Andrew MH, Gillison AN (1985) Australian savanna ecosystems. In: Tothill JC, Mott JJ (eds) Ecology and management of the world's savannas. Australian Academy of Science, Canberra, pp 56–82
Nadelhoffer KJ, Shaver G, Fry B, Giblin A, Johnson L, McKane R (1996) 15N natural abundance and N use by tundra plants. Oecologia 107:386–394
Pate JS, Stewart GR, Unkovich M (1993) 15N natural abundance of plant and soil components of a Banksia woodland ecosystem in relation to nitrate utilisation, life form, mycorrhizal status and N2-fixing abilities of component species. Plant Cell Environ 16:365–373
Reddell P, Milnes AR (1992) Mycorrhizas and rehabilitation of waste rock dumps. Aust J Bot 40:233–242
Robinson D (2001) δ15N as an integrator of the nitrogen cycle. Trends Ecol Evol 16:153–162
Schmidt S, Stewart GR (1997) Waterlogging and fire impacts on N availability in a subtropical wet heathland (wallum). Plant Cell Environ 20:1231–1241
Schmidt S, Stewart GR (1998) Transport, storage and mobilization of N by trees and shrubs in the wet/dry tropics of northern Australia. Tree Physiol 18:403–410
Schmidt S, Stewart GR, Turnbull MH, Erskine PD, Ashwath N (1998) N relations of natural and disturbed plant communities in tropical Australia. Oecologia 117:95–104
Schulze E-D, Gebauer G, Ziegler H, Lange OL (1991) Estimates of nitrogen fixation by trees on an aridity gradient in Namibia. Oecologia 88:451–455
Schulze E-D, Williams RJ, Farquhar GD, Schulze W, Langridge J, Miller JM, Walker BH (1998) Carbon and nitrogen isotope discrimination and nitrogen nutrition of trees along a rainfall gradient in northern Australia. Aust J Plant Physiol 25:413–425
Schulze E-D, Farquhar GD, Miller JM, Schulze W, Walker BH, Williams RJ (1999) Interpretation of increased foliar δ15N in woody species along a rainfall gradient in northern Australia. Aust J Plant Physiol 26:296–298
Stewart GR, Schmidt S (1999) The evolution and ecology of plant mineral nutrition. In: Press MC, Scholes JD, Barker MG (eds) Physiological plant ecology. Blackwell, Oxford, pp 91–114
Stock WD, Wienand KT, Baker AC (1995) Impacts of invading N2-fixing Acacia species on patterns of nutrient cycling in two Cape ecosystems: evidence from soil incubation studies and 15N natural abundance values. Oecologia 101:375–382
Yoneyama T, Omata T, Nakata S, Yazaki J (1991) Fractionation of N isotopes during the uptake and assimilation of ammonia by plants. Plant Cell Physiol 32:1211–1217
Yoneyama T, Matsumaru T, Usui K, Engelaar WMHG (2001) Discrimination of nitrogen isotopes during absorption of ammonium and nitrate at different nitrogen concentration by rice (Oryza sativa L.) plants. Plant Cell Environ 24:133–139
Acknowledgements
We thank EPA of the Supervising Scientist, Jabiru, for logistic support and CSIRO Tropical Ecosystems Research Centre for access to the Kapalga fire trial. We are indebted to Drs. N. Ashwath, L. Hutley, M. Turnbull, P. Erskine, G. Woodall, I. Biggs and S. Richards for helping with field and laboratory work. G. Moss and J. Stewart provided excellent expert technical support with mass spectrometry. Many thanks to Prof. Peter Högberg for supplying additional data for the African communities. The Australian Research Council supported this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schmidt, S., Stewart, G.R. δ15N values of tropical savanna and monsoon forest species reflect root specialisations and soil nitrogen status. Oecologia 134, 569–577 (2003). https://doi.org/10.1007/s00442-002-1150-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-002-1150-y