Abstract
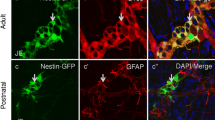
Cholecystokinin (CCK) is an early marker of both neuronal and endocrine cell lineages in the developing gastrointestinal tract. To determine the quantitative properties and the spatial distribution of the CCK-expressing myenteric neurones in early postnatal life, a transgenic mouse strain with a CCK promoter-driven red fluorescent protein (DsRedT3/CCK) was established. The cell-specific expression of DsRedT3/CCK was validated by in situ hybridization with a CCK antisense riboprobe and by in situ hybridization coupled with immunohistochemistry involving a monoclonal antibody to CCK. A gradual increase in the DsRedT3/CCK-expressing enteric neurones with clear regional differences was documented from birth until the suckling to weaning transition, in parallel with the period of rapid intestinal growth and functional maturation. To evaluate the proportion of myenteric neurones in which DsRedT3/CCK transgene expression was colocalized with the enteric neuronal marker peripherin, immunofluorescence techniques were applied. All DsRedT3/CCK neurones were peripherin-immunoreactive and the proportion of DsRedT3/CCK-expressing myenteric neurones in the duodenum was the highest after the third week of life, when the number of peripherin-immunoreactive myenteric neurones in this region had decreased. Nearly all of the DsRedT3/CCK-expressing neurones also expressed 5-hydroxytryptophan (5-HT). Thus, by utilizing a new transgenic mouse strain, we have demonstrated a small number of CCK-expressing myenteric neurones with a developmentally regulated spatiotemporal distribution. The coexistence of CCK and 5-HT in the majority of these neurones suggests their possible regulatory role in feeding at the suckling to weaning transition.





Similar content being viewed by others
References
Bagyánszki M, Torfs P, Krecsmarik M, Fekete E, Adriaensen D, Van Nassauw L, Timmermans JP, Kroese AB (2011) Chronic alcohol consumption induces an overproduction of NO by nNOS- and iNOS-expressing myenteric neurons in the murine small intestine. Neurogastroenterol Motil 23:e237–e248
Bevis BJ, Glick BS (2002) Rapidly maturing variants of the Discosoma red fluorescent protein (DsRed). Nat Biotechnol 20:83–87
Chandra R, Samsa LA, Vigna SR, Liddle RA (2010) Pseudopod-like basal cell processes in intestinal cholecystokinin cells. Cell Tissue Res 341:289–297
Crawley JN, Corwin RL (1994) Biological actions of cholecystokinin. Peptides 15:731–755
Curley JP, Jordan ER, Swaney WT, Izraelit A, Kammel S, Champagne FA (2009) The meaning of weaning: influence of the weaning period on behavioral development in mice. Dev Neurosci 31:318–331
Dockray GJ (1976) Immunochemical evidence of cholecystokinin-like peptides in brain. Nature 264:568–570
Emery DW (2011) The use of chromatin insulators to improve the expression and safety of integrating gene transfer vectors. Hum Gene Ther 22:761–774
Furness JB, Costa M, Keast JR (1984) Choline acetyltransferase- and peptide immunoreactivity of submucous neurons in the small intestine of the guinea-pig. Cell Tissue Res 237:329–336
Furness JB, Young HM, Pompolo S, Bornstein JC, Kunze WA, McConalogue K (1995) Plurichemical transmission and chemical coding of neurons in the digestive tract. Gastroenterology 108:554–563
Gabella G (1989) Fall in the number of myenteric neurons in aging guinea pigs. Gastroenterology 96:1487–1493
Giralt M, Vergara P (2000) Inhibition by CCK of ascending contraction elicited by mucosal stimulation in the duodenum of the rat. Neurogastroenterol Motil 12:173–180
Gulley S, Covasa M, Ritter RC, Sayegh AI (2005) Cholecystokinin1 receptors mediate the increase in Fos-like immunoreactivity in the rat myenteric plexus following intestinal oleate infusion. Physiol Behav 86:128–135
Hayes MR, Covasa M (2006) Dorsal hindbrain 5-HT3 receptors participate in control of meal size and mediate CCK-induced satiation. Brain Res 1103:99–107
Hayes MR, Chory FM, Gallagher CA, Covasa M (2006) Serotonin type-3 receptors mediate cholecystokinin-induced satiation through gastric distension. Am J Physiol Regul Integr Comp Physiol 291:R115–R123
Hökfelt T, Lundberg JM, Schultzberg M, Johansson O, Skirboll L, Anggård A, Fredholm B, Hamberger B, Pernow B, Rehfeld J, Goldstein M (1980) Cellular localization of peptides in neural structures. Proc R Soc Lond B Biol Sci 210:63–77
Izbéki F, Wittman T, Rosztóczy A, Linke N, Bódi N, Fekete E, Bagyánszki M (2008) Immediate insulin treatment prevents gut motility alterations and loss of nitrergic neurons in the ileum and colon of rats with streptozotocin-induced diabetes. Diabetes Res Clin Pract 80:192–198
Larsson LI, Rehfeld JF (1979) Localization and molecular heterogeneity of cholecystokinin in the central and peripheral nervous system. Brain Res 165:201–218
Lay JM, Gillespie PJ, Samuelson LC (1999) Murine prenatal expression of cholecystokinin in neural crest, enteric neurons, and enteroendocrine cells. Dev Dyn 216:190–200
Lay JM, Bane G, Brunkan CS, Davis J, Lopez-Diaz L, Samuelson LC (2004) Enteroendocrine cell expression of a cholecystokinin gene construct in transgenic mice and cultured cells. Am J Physiol Gastrointest Liver Physiol 288:G354–G361
Lee EC, Yu D, Martinez de Velasco J, Tessarollo L, Swing DA, Court DL, Jenkins NA, Copeland NG (2001) A highly efficient Escherichia coli-based chromosome engineering system adapted for recombinogenic targeting and subcloning of BAC DNA. Genomics 73:56–65
Messenger JP, Furness JB (1990) Projections of chemically-specified neurons in the guinea-pig colon. Arch Histol Cytol 53:467–495
Muller JE, Straus E, Yalow RS (1977) Cholecystokinin and its COOH-terminal octapeptide in the pig brain. Proc Natl Acad Sci USA 74:3035–3037
Powley TL, Phillips RJ (2004) Gastric satiation is volumetric, intestinal satiation is nutritive. Physiol Behav 82:69–74
Raybould HE, Glatzle J, Robin C, Meyer JH, Phan T, Wong H, Sternini C (2003) Expression of 5-HT3 receptors by extrinsic duodenal afferents contribute to intestinal inhibition of gastric emptying. Am J Physiol Gastrointest Liver Physiol 284:G367–G372
Recillas-Targa F, Valadez-Graham V, Farrell CM (2004) Prospects and implications of using chromatin insulators in gene therapy and transgenesis. Bioessays 26:796–807
Román V, Bagyánszki M, Krecsmarik M, Horváth A, Resch BA, Fekete E (2004) Spatial pattern analysis of nitrergic neurons in the developing myenteric plexus of the human fetal intestine. Cytometry A 57:108–112
Santer RM (1994) Survival of the population of NADPH-diaphorase stained myenteric neurons in the small intestine of aged rats. J Auton Nerv Syst 49:115–121
Scarpignato C, Varga G, Corradi C (1993) Effect of CCK and its antagonists on gastric emptying. J Physiol (Paris) 87:291–300
Schultzberg M, Hökfelt T, Nilsson G, Terenius L, Rehfeld JF, Brown M, Elde R, Goldstein M, Said S (1980) Distribution of peptide- and catecholamine-containing neurons in the gastro-intestinal tract of rat and guinea-pig: immunohistochemical studies with antisera to substance P, vasoactive intestinal polypeptide, enkephalins, somatostatin, gastrin/cholecystokinin, neurotensin and dopamine beta-hydroxylase. Neuroscience 5:689–744
Schutte IW, Hollestein KB, Akkermans LM, Kroese AB (1997) Evidence for a role of cholecystokinin as neurotransmitter in the guinea-pig enteric nervous system. Neurosci Lett 236:155–158
Storr M, Sattler D, Hahn A, Schusdziarra V, Allescher HD (2003) Endogenous CCK depresses contractile activity within the ascending myenteric reflex pathway of rat ileum. Neuropharmacology 44:524–532
Vintersten K, Monetti C, Gertsenstein M, Zhang P, Laszlo L, Biechele S, Nagy A (2004) Mouse in red: red fluorescent protein expression in mouse ES cells, embryos, and adult animals. Genesis 40:241–246
Washington MC, Coggeshall J, Sayegh AI (2011) Cholecystokinin-33 inhibits meal size and prolongs the subsequent intermeal interval. Peptides 32:971–797
Weller A (2006) The ontogeny of postingestive inhibitory stimuli: examining the role of CCK. Dev Psychobiol 48:368–379
Xiang Z, Burnstock G (2004) Development of nerves expressing P2X3 receptors in the myenteric plexus of rat stomach. Histochem Cell Biol 122:111–119
Author information
Authors and Affiliations
Corresponding author
Additional information
Zoltán Máté and Marietta Zita Poles equally contributed to this work.
Rights and permissions
About this article
Cite this article
Máté, Z., Poles, M.Z., Szabó, G. et al. Spatiotemporal expression pattern of DsRedT3/CCK gene construct during postnatal development of myenteric plexus in transgenic mice. Cell Tissue Res 352, 199–206 (2013). https://doi.org/10.1007/s00441-013-1552-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-013-1552-7