Abstract
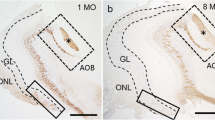
Age-related changes were examined in the distribution and severity of spontaneous lesions in the neuroepithelium and Bowman’s glands in mouse olfactory mucosa. The olfactory mucosa of female ICR mice at postnatal ages from 10 days to 16 months were investigated histologically by hematoxylin and eosin staining, high-iron diamine-Alcian blue (HID-AB) staining, and immunohistochemistry for olfactory marker protein (OMP), βIII tubulin (βIIIT), and Ki67. The lesions in the neuroepithelium and Bowman’s glands were quantitatively assessed by morphometric analyses of sections stained with anti-OMP antibody or HID-AB. The first appearance of neuroepithelial abnormality was observed in the dorsomedial portion of the olfactory mucosa in 5-month-old mice. The distribution and severity of lesions progressed with increasing age. In mildly affected epithelium in which OMP-positive olfactory receptor neurons (ORNs) were present but in smaller amounts, the numbers of βIIIT-positive and Ki67-positive neuroepithelial cells tended to be increased, indicating that neurogenesis was upregulated in these areas. In contrast, severely affected epithelium in which OMP-positive ORNs were virtually absent showed high variability in the numbers of βIIIT- and Ki67-positive cells among the areas examined, probably reflecting differences in the capacity of the basal cells remaining in the affected area to generate new neuronal cells. Histological analysis with HID-AB revealed that spontaneous lesions in Bowman’s glands also occurred in aged mouse olfactory mucosa. Lesions in the neuroepithelium and underlying Bowman’s glands tended to be spatially co-localized, suggesting a close association between pathogeneses in these two structures. Moreover, lesions in Bowman’s glands were associated with changes in the biochemical composition of mucus on the olfactory mucosa. This information should prove useful in improving the understanding of the pathogenetic mechanisms underlying age-related changes in the peripheral olfactory system.










Similar content being viewed by others
References
Bahrami F, Bergman U, Brittebo EB, Brandt I (2000) Persistent olfactory mucosal metaplasia and increased olfactory bulb glial fibrillary acidic protein levels following a single dose of methylsulfonyl-dichlorobenzene in mice: comparison of the 2,5- and 2,6-dichlorinated isomers. Toxicol Appl Pharmacol 162:49–59
Bergman U, Ostergren A, Gustafson AL, Brittebo B (2002) Differential effects of olfactory toxicants on olfactory regeneration. Arch Toxicol 76:104–112
Bihun CG, Percy DH (1995) Morphologic changes in the nasal cavity associated with sialodacryoadenitis virus infection in the Wistar rat. Vet Pathol 32:1–10
Calof AL, Hagiwara N, Holcomb JD, Mumm JS, Shou J (1996) Neurogenesis and cell death in olfactory epithelium. J Neurobiol 30:67–81
Chance DL, Mawhinney TP (2000) Carbohydrate sulfation effects on growth of Pseudomonas aeruginosa. Microbiology 146:1717–1725
Cuschieri A, Bannister LH (1974) Some histochemical observations on the mucosubstances of the nasal glands of the mouse. Histochem J 6:543–558
Doty RL, Shaman P, Applebaum SL, Giberson R, Siksorski L, Rosenberg L (1984) Smell identification ability: changes with age. Science 226:1441–1443
Ducray A, Bondier JR, Michel G, Bon K, Millot JL, Propper A, Kastner A (2002) Recovery following peripheral destruction of olfactory neurons in young and adult mice. Eur J Neurosci 15:1907–1917
Endo T, Tamaki K, Arimura Y, Itoh F, Hinoda Y, Hareyama M, Irimura T, Fujita M, Imai K (1998) Expression of sulfated carbohydrate chain and core peptides of mucin detected by monoclonal antibodies in Barrett’s esophagus and esophageal adenocarcinoma. J Gastroenterol 33:811–815
Farage MA, Miller KW, Elsner P, Maibach HI (2008) Intrinsic and extrinsic factors in skin ageing: a review. Int J Cosmet Sci 30:87–95
Farbman AI (1990) Olfactory neurogenesis: genetic or environmental controls? Trends Neurosci 13:362–365
Genter MB, Deamer NJ, Blake BL, Wesley DS, Levi PE (1995) Olfactory toxicity of methimazole: dose-response and structure-activity studies and characterization of flavin-containing monooxygenase activity in the Long-Evans rat olfactory mucosa. Toxicol Pathol 23:477–486
Getchell ML, Getchell TV (1991) Immunohistochemical localization of components of the immune barrier in the olfactory mucosae of salamanders and rats. Anat Rec 231:358–374
Graziadei GA, Graziadei PP (1979) Neurogenesis and neuron regeneration in the olfactory system of mammals. II. Degeneration and reconstitution of the olfactory sensory neurons after axotomy. J Neurocytol 8:197–213
Grubb BR, Rogers TD, Kulaga HM, Burns KA, Wonsetler RL, Reed RR, Ostrowski LE (2007) Olfactory epithelia exhibit progressive functional and morphological defects in CF mice. Am J Physiol Cell Physiol 293:C574–C583
Hinds JW, McNelly NA (1977) Aging of the rat olfactory bulb: growth and atrophy of constituent layers and changes in size and number of mitral cells. J Comp Neurol 72:345–367
Holbrook EH, Leopold DA, Schwob JE (2005) Abnormalities of axon growth in human olfactory mucosa. Laryngoscope 115:2144–2154
Huard JM, Youngentob SL, Goldstein BJ, Luskin MB, Schwob JE (1998) Adult olfactory epithelium contains multipotent progenitors that give rise to neurons and non-neural cells. J Comp Neurol 400:469–486
Kase H, Kodama S, Tanaka K (1999) Observations of high iron diamine-Alcian blue stain in uterine cervical glandular lesions. Gynecol Obstet Invest 48:56–60
Krishna NS, Getchell TV, Getchell ML (1994) Differential expression of alpha, mu, and pi classes of glutathione S-transferases in chemosensory mucosae of rats during development. Cell Tissue Res 275:435–450
Lee VM, Pixley SK (1994) Age and differentiation-related differences in neuron-specific tubulin immunostaining of olfactory sensory neurons. Brain Res Dev Brain Res 83:209–215
Ling G, Gu J, Genter MB, Zhuo X, Ding X (2004) Regulation of cytochrome P450 gene expression in the olfactory mucosa. Chem Biol Interact 147:247–258
Loo AT, Youngentob SL, Kent PF, Schwob JE (1996) The aging olfactory epithelium: neurogenesis, response to damage, and odorant-induced activity. Int J Dev Neurosci 14:881–900
Maruniak JA, Lin PJ, Henegar JR (1989) Effects of unilateral naris closure on the olfactory epithelia of adult mice. Brain Res 490:212–218
Matarazzo V, Zsurger N, Guillemot JC, Clot-Faybesse O, Botto JM, Dal Farra C, Crowe M, Demaille J, Vincent JP, Mazella J, Ronin C (2002) Porcine odorant-binding protein selectively binds to a human olfactory receptor. Chem Senses 27:691–701
Mellert TK, Getchell ML, Sparks L, Getchell TV (1992) Characterization of the immune barrier in human olfactory mucosa. Otolaryngol Head Neck Surg 106:181–188
Mery S, Gross EA, Joyner DR, Godo M, Morgan KT (1994) Nasal diagrams: a tool for recording the distribution of nasal lesions in rats and mice. Toxicol Pathol 22:353–372
Mirich JM, Williams NC, Berlau DJ, Brunjes PC (2002) Comparative study of aging in the mouse olfactory bulb. J Comp Neurol 454:361–372
Monti Graziadei GA (1983) Experimental studies on the olfactory marker protein. III. The olfactory marker protein in the olfactory neuroepithelium lacking connections with the forebrain. Brain Res 262:303–308
Moran DT, Jafek BW, Eller PM, Rowley JC 3rd (1992) Ultrastructural histopathology of human olfactory dysfunction. Microsc Res Tech 23:103–110
Murphy C, Schubert CR, Cruickshanks KJ, Klein BE, Klein R, Nondahl DM (2002) Prevalence of olfactory impairment in older adults. JAMA 288:2307–2312
Nagano K, Katagiri T, Aiso S, Senoh H, Sakura Y, Takeuchi T (1997) Spontaneous lesions of nasal cavity in aging F344 rats and BDF1 mice. Exp Toxicol Pathol 49:97–104
Nagao H, Yamaguchi M, Takahash Y, Mori K (2002) Grouping and representation of odorant receptors in domains of the olfactory bulb sensory map. Microsc Res Tech 58:168–175
Nakashima T, Kimmelman CP, Snow JB Jr (1984) Structure of human fetal and adult olfactory neuroepithelium. Arch Otolaryngol 110:641–646
Olofsson S, Bergstrom T (2005) Glycoconjugate glycans as viral receptors. Ann Med 37:154–172
Reden J, Mueller A, Mueller C, Konstantinidis I, Frasnelli J, Landis BN, Hummel T (2006) Recovery of olfactory function following closed head injury or infections of the upper respiratory tract. Arch Otolaryngol Head Neck Surg 132:265–269
Ressler KJ, Sullivan SL, Buck LB (1993) A zonal organization of odorant receptor gene expression in the olfactory epithelium. Cell 73:597–609
Roskams AJ, Cai X, Ronnett GV (1998) Expression of neuron-specific beta-III tubulin during olfactory neurogenesis in the embryonic and adult rat. Neuroscience 83:191–200
Rosli Y, Breckenridge LJ, Smith RA (1999) An ultrastructural study of age-related changes in mouse olfactory epithelium. J Electron Microsc (Tokyo) 48:77–84
Schulz BL, Sloane AJ, Robinson LJ, Prasad SS, Lindner RA, Robinson M, Bye PT, Nielson DW, Harry JL, Packer NH, Karlsson NG (2007) Glycosylation of sputum mucins is altered in cystic fibrosis patients. Glycobiology 17:698–712
Schwob JE (2002) Neural regeneration and the peripheral olfactory system. Anat Rec 269:33–49
Schwob JE, Szumowski KE, Stasky AA (1992) Olfactory sensory neurons are trophically dependent on the olfactory bulb for their prolonged survival. J Neurosci 12:3896–3919
Schwob JE, Saha S, Youngentob SL, Jubelt B (2001) Intranasal inoculation with the olfactory bulb line variant of mouse hepatitis virus causes extensive destruction of the olfactory bulb and accelerated turnover of neurons in the olfactory epithelium of mice. Chem Senses 26:937–952
Spicer SS (1965) Diamine methods for differentialing mucosubstances histochemically. J Histochem Cytochem 13:211–234
Suzuki Y, Takeda M, Obara N, Suzuki N, Takeichi N (2000) Olfactory epithelium consisting of supporting cells and horizontal basal cells in the posterior nasal cavity of mice. Cell Tissue Res 299:313–325
Temmel AF, Quint C, Schickinger-Fischer B, Klimek L, Stoller E, Hummel T (2002) Characteristics of olfactory disorders in relation to major causes of olfactory loss. Arch Otolaryngol Head Neck Surg 128:635–641
Trojanowski JQ, Newman PD, Hill WD, Lee VM (1991) Human olfactory epithelium in normal aging, Alzheimer’s disease, and other neurodegenerative disorders. J Comp Neurol 310:365–376
Vassar R, Ngai J, Axel R (1993) Spatial segregation of odorant receptor expression in the mammalian olfactory epithelium. Cell 74:309–318
Vedin V, Slotnick B, Berghard A (2004) Zonal ablation of the olfactory sensory neuroepithelium of the mouse: effects on odorant detection. Eur J Neurosci 20:1858–1864
Verhaagen J, Oestreicher AB, Grillo M, Khew-Goodall YS, Gispen WH, Margolis FL (1990) Neuroplasticity in the olfactory system: differential effects of central and peripheral lesions of the primary olfactory pathway on the expression of B-50/GAP43 and the olfactory marker protein. J Neurosci Res 26:31–44
Watanabe K, Kondo K, Takeuchi N, Nibu K, Kaga K (2006) Age-related changes in cell density and the proliferation rate of olfactory ensheathing cells in the lamina propria of postnatal mouse olfactory mucosa. Brain Res 1116:82–92
Watanabe K, Kondo K, Takeuchi N, Okano H, Yamasoba T (2007) Musashi-1 expression in postnatal mouse olfactory epithelium. Neuroreport 18:641–644
Weiler E, Farbman AI (1997) Proliferation in the rat olfactory epithelium: age-dependent changes. J Neurosci 17:3610–3622
Whitby-Logan GK, Weech M, Walters E (2004) Zonal expression and activity of glutathione S-transferase enzymes in the mouse olfactory mucosa. Brain Res 995:151–157
Yamagishi M, Fujiwara M, Nakamura H (1994) Olfactory mucosal findings and clinical course in patients with olfactory disorders following upper respiratory viral infection. Rhinology 32:113–118
Youngentob SL, Schwob JE, Sheehe PR, Youngentob LM (1997) Odorant threshold following methyl bromide-induced lesions of the olfactory epithelium. Physiol Behav 62:1241–1252
Yu TT, McIntyre JC, Bose SC, Hardin D, Owen MC, McClintock TS (2005) Differentially expressed transcripts from phenotypically identified olfactory sensory neurons. J Comp Neurol 483:251–262
Acknowledgements
We thank Dr. Frank Margolis, Department of Anatomy and Neurobiology/Medicine, University of Maryland School of Medicine, for providing the OMP antiserum, and Mr. Yoshiro Mori, Ms. Yukari Kurasawa, and Mr. Koichi Miyazawa for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by grants from the Japanese Ministry of Education, Culture, Sports, Science, and Technology (nos. 14770886, 16790987 and 18799002; K. Kondo) and a grant from the Japanese Ministry of Health, Labour, and Welfare (Comprehensive Research on Aging and Welfare, no. H13-choju-012; K. Nibu).
Rights and permissions
About this article
Cite this article
Kondo, K., Watanabe, K., Sakamoto, T. et al. Distribution and severity of spontaneous lesions in the neuroepithelium and Bowman’s glands in mouse olfactory mucosa: age-related progression. Cell Tissue Res 335, 489–503 (2009). https://doi.org/10.1007/s00441-008-0739-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-008-0739-9