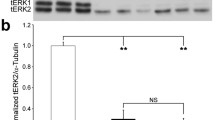
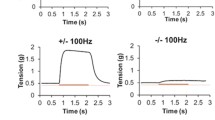
Abstract
Acetylcholinesterase (AChE) plays an essential role in neuromuscular transmission. Not surprisingly, neuromuscular transmission during repetitive nerve stimulation is severely depressed in the AChE knockout mouse (KO). However, whether this deficit in AChE leads to skeletal muscle changes is not known. We have studied the in vitro contractile properties of the postural and locomotor soleus muscles of adult KO and normal (wildtype, WT) mice, and this was completed by histological and biochemical analyses. Our results show that muscle weight, cross-sectional area of muscle fibres and absolute maximal isometric force are all reduced in KO mice compared with WT mice. Of interest, the relative amount of slow myosin heavy chain (MHC-1) in muscle homogenates and the percentage of muscle fibres expressing MHC-1 are decreased in the KO mice. Surprisingly, AChE ablation does not modify twitch kinetics, absolute maximal power, fatigue resistance or citrate synthase activity, despite the reduced number of slow muscle fibres. Thus, a deficit in AChE leads to alterations in the structure and function of muscles but these changes are not simply related to the reduced body weight of KO mice. Our results also suggest that this murine model of congenital myasthenic syndrome with endplate AChE deficiency combines alterations in both neurotransmission and intrinsic muscle properties.



Similar content being viewed by others
References
Adler M, Manley HA, Purcell AL, Deshpande SS, Hamilton TA, Kan RK, Oyler G, Lockridge O, Duysen EG, Sheridan RE (2004) Reduced acetylcholine receptor density, morphological remodeling, and butyrylcholinesterase activity can sustain muscle function in acetylcholinesterase knockout mice. Muscle Nerve 30:317–327
Aldunate R, Casar JC, Brandan E, Inestrosa NC (2004) Structural and functional organization of synaptic acetylcholinesterase. Brain Res Brain Res Rev 47:96–104
Ausoni S, Gorza L, Schiaffino S, Gundersen K, Lomo T (1990) Expression of myosin heavy chain isoforms in stimulated fast and slow rat muscles. J Neurosci 10:153–160
Belluardo N, Westerblad H, Mudo G, Casabona A, Bruton J, Caniglia G, Pastoris O, Grassi F, Ibanez CF (2001) Neuromuscular junction disassembly and muscle fatigue in mice lacking neurotrophin-4. Mol Cell Neurosci 18:56–67
Buffelli M, Pasino E, Cangiano A (1997) Paralysis of rat skeletal muscle equally affects contractile properties as does permanent denervation. J Muscle Res Cell Motil 18:683–695
Cousin X, Strahle U, Chatonnet A (2005) Are there non-catalytic functions of acetylcholinesterases? Lessons from mutant animal models. Bioessays 27:189–200
De Bleecker J, Van den Abeele K, De Reuck J (1994) Electromyography in relation to end-plate acetylcholinesterase in rats poisoned by different organophosphates. Neurotoxicology 15:331–340
Derave W, Van Den Bosch L, Lemmens G, Eijnde BO, Robberecht W, Hespel P (2003) Skeletal muscle properties in a transgenic mouse model for amyotrophic lateral sclerosis: effects of creatine treatment. Neurobiol Dis 13:264–272
Duysen EG, Stribley JA, Fry DL, Hinrichs SH, Lockridge O (2002) Rescue of the acetylcholinesterase knockout mouse by feeding a liquid diet; phenotype of the adult acetylcholinesterase deficient mouse. Brain Res Dev Brain Res 137:43–54
Edman KA (1979) The velocity of unloaded shortening and its relation to sarcomere length and isometric force in vertebrate muscle fibres. J Physiol (Lond) 291:143–159
Engel AG, Sine SM (2005) Current understanding of congenital myasthenic syndromes. Curr Opin Pharmacol 5:308–321
Fitts RH, Riley DR, Widrick JJ (2000) Physiology of a microgravity environment (invited review): microgravity and skeletal muscle. J Appl Physiol 89:823–839
Fougerousse F, Gonin P, Durand M, Richard I, Raymackers JM (2003) Force impairment in calpain 3-deficient mice is not correlated with mechanical disruption. Muscle Nerve 27:616–623
Girard E, Barbier J, Chatonnet A, Krejci E, Molgo J (2005) Synaptic remodeling at the skeletal neuromuscular junction of acetylcholinesterase knockout mice and its physiological relevance. Chem Biol Interact 157–158:87–96
Goyenvalle A, Vulin A, Fougerousse F, Leturcq F, Kaplan JC, Garcia L, Danos O (2004) Rescue of dystrophic muscle through U7 snRNA-mediated exon skipping. Science 306:1796–1799
Hamalainen N, Pette D (1996) Slow-to-fast transitions in myosin expression of rat soleus muscle by phasic high-frequency stimulation. FEBS Lett 399:220–222
Hodgson JA, Roy RR, Higuchi N, Monti RJ, Zhong H, Grossman E, Edgerton VR (2005) Does daily activity level determine muscle phenotype? J Exp Biol 208:3761–3770
Jakubiec-Puka A, Ciechomska I, Morga J, Matusiak A (1999) Contents of myosin heavy chains in denervated slow and fast rat leg muscles. Comp Biochem Physiol [B] Biochem Mol Biol 122:355–362
Karalliedde L, Baker D, Marrs TC (2006) Organophosphate-induced intermediate syndrome: aetiology and relationships with myopathy. Toxicol Rev 25:1–14
Launay T, Noirez P, Butler-Browne G, Agbulut O (2006) Expression of slow myosin heavy chain during muscle regeneration is not always dependent on muscle innervation and calcineurin phosphatase activity. Am J Physiol Regul Integr Comp Physiol 290:R1508–R1514
Lewis DM, al-Amood WS, Schmalbruch H (1997) Effects of long-term phasic electrical stimulation on denervated soleus muscle: guinea-pig contrasted with rat. J Muscle Res Cell Motil 18:573–586
Marechal G, Beckers-Bleukx G (1993) Force-velocity relation and isomyosins in soleus muscles from two strains of mice (C57 and NMRI). Pflugers Arch 424:478–487
Maselli RA, Leung C (1993) Analysis of neuromuscular transmission failure induced by anticholinesterases. Ann N Y Acad Sci 681:402–404
Mendias CL, Marcin JE, Calerdon DR, Faulkner JA (2006) Contractile properties of EDL and soleus muscles of myostatin-deficient mice. J Appl Physiol 101:898–905
Merkulova T, Dehaupas M, Nevers MC, Creminon C, Alameddine H, Keller A (2000) Differential modulation of alpha, beta and gamma enolase isoforms in regenerating mouse skeletal muscle. Eur J Biochem 267:3735–3743
Midrio M (2006) The denervated muscle: facts and hypotheses. A historical review. Eur J Appl Physiol 98:1–21
Milatovic D, Zivin M, Hustedt E, Dettbarn WD (2000) Spin trapping agent phenyl-N-tert-butylnitrone prevents diisopropylphosphorofluoridate-induced excitotoxicity in skeletal muscle of the rat. Neurosci Lett 278:25–28
Minic J, Chatonnet A, Krejci E, Molgo J (2003) Butyrylcholinesterase and acetylcholinesterase activity and quantal transmitter release at normal and acetylcholinesterase knockout mouse neuromuscular junctions. Br J Pharmacol 138:177–187
Mouisel E, Blondet B, Escourrou P, Chatonnet A, Molgo J, Ferry A (2006) Outcome of acetylcholinesterase deficiency for neuromuscular functioning. Neurosci Res 55:389–396
Muller JS, Mihaylova V, Abicht A, Lochmuller H (2007) Congenital myasthenic syndromes: spotlight on genetic defects of neuromuscular transmission. Expert Rev Mol Med 9:1–20
Panenic R, Gisiger V, Gardiner PF (1999) Fatigability of rat hindlimb muscles after acute irreversible acetylcholinesterase inhibition. J Appl Physiol 87:1455–1462
Roy RR, Zhong H, Monti RJ, Vallance KA, Edgerton VR (2002) Mechanical properties of the electrically silent adult rat soleus muscle. Muscle Nerve 26:404–412
Schiaffino S, Reggiani C (1996) Molecular diversity of myofibrillar proteins: gene regulation and functional significance. Physiol Rev 76:371–423
Schiaffino S, Sandri M, Murgia M (2007) Activity-dependent signaling pathways controlling muscle diversity and plasticity. Physiology (Bethesda) 22:269–278
Shavlakadze T, Grounds M (2006) Of bears, frogs, meat, mice and men: complexity of factors affecting skeletal muscle mass and fat. Bioessays 28:994–1009
Srere PA (1969) Citrate synthase. Methods Enzymol 13:3–11
Thiermann H, Eyer P, Worek F, Szinicz L (2005) Effects of oximes on muscle force and acetylcholinesterase activity in isolated mouse hemidiaphragms exposed to paraoxon. Toxicology 214:190–197
Van der Kloot W, Molgo J (1994) Quantal acetylcholine release at the vertebrate neuromuscular junction. Physiol Rev 74:899–991
Ventura-Clapier R, Kuznetsov AV, d'Albis A, Deursen J van, Wieringa B, Veksler VI (1995) Muscle creatine kinase-deficient mice. I. Alterations in myofibrillar function. J Biol Chem 270:19914–19920
Witzemann V, Schwarz H, Koenen M, Berberich C, Villarroel A, Wernig A, Brenner HR, Sakmann B (1996) Acetylcholine receptor epsilon-subunit deletion causes muscle weakness and atrophy in juvenile and adult mice. Proc Natl Acad Sci USA 93:13286–13291
Xie W, Stribley JA, Chatonnet A, Wilder PJ, Rizzino A, McComb RD, Taylor P, Hinrichs SH, Lockridge O (2000) Postnatal developmental delay and supersensitivity to organophosphate in gene-targeted mice lacking acetylcholinesterase. J Pharmacol Exp Ther 293:896–902
Yang D, He F, Li T (2001) Repetitive nerve stimulation and stimulation single fiber electromyography studies in rats intoxicated with single or mixed insecticides. Toxicology 161:111–116
Zhao J, Brault JJ, Schild A, Cao P, Sandri M, Schiaffino S, Lecker SH, Goldberg AL (2007) FoxO3 coordinately activates protein degradation by the autophagic/lysosomal and proteasomal pathways in atrophying muscle cells. Cell Metab 6:472–483
Acknowledgements
We thank Angélica Keller (UMR 7149, Créteil) in whose laboratory the enzyme activity measurements were carried out and Stéphanie Gay and Muriel Durand for excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by the Association Française contre les Myopathies, INSERM, University Pierre et Marie Curie, European Commission 6th Framework programme and the Myores Network of Excellence (contract 5111978).
Rights and permissions
About this article
Cite this article
Vignaud, A., Fougerousse, F., Mouisel, E. et al. Genetic inactivation of acetylcholinesterase causes functional and structural impairment of mouse soleus muscles. Cell Tissue Res 333, 289–296 (2008). https://doi.org/10.1007/s00441-008-0640-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-008-0640-6