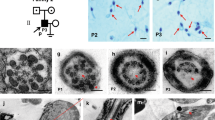
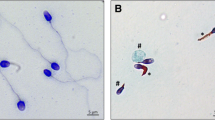
Abstract
Spermatozoa are polarized cells with a head and a flagellum joined together by the connecting piece. Flagellum integrity is critical for normal sperm function, and flagellum defects consistently lead to male infertility. Multiple morphological abnormalities of the flagella (MMAF) is a distinct sperm phenotype consistently leading to male infertility due to a reduced or absent sperm motility associated with severe morphological and ultrastructural flagellum defects. Despite numerous genes recently described to be recurrently associated with MMAF, more than half of the cases analyzed remain unresolved, suggesting that many yet uncharacterized gene defects account for this phenotype. By performing a retrospective exome analysis of the unsolved cases from our initial cohort of 167 infertile men with a MMAF phenotype, we identified one individual carrying a homozygous frameshift variant in CFAP206, a gene encoding a microtubule-docking adapter for radial spoke and inner dynein arm. Immunostaining experiments in the patient’s sperm cells demonstrated the absence of WDR66 and RSPH1 proteins suggesting severe radial spokes and calmodulin and spoke-associated complex defects. Using the CRISPR–Cas9 technique, we generated homozygous Cfap206 knockout (KO) mice which presented with male infertility due to functional, structural and ultrastructural sperm flagellum defects associated with a very low rate of embryo development using ICSI. Overall, we showed that CFAP206 is essential for normal sperm flagellum structure and function in human and mouse and that bi-allelic mutations in CFAP206 cause male infertility in man and mouse by inducing morphological and functional defects of the sperm flagellum that may also cause ICSI failures.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request. Accession numbers: The CFAP206 variant reported in this manuscript is accessible in ClinVar with the submission number SUB9294549.
References
Auguste Y, Delague V, Desvignes J-P et al (2018) Loss of calmodulin- and radial-spoke-associated complex protein CFAP251 leads to immotile spermatozoa lacking mitochondria and infertility in men. Am J Hum Genet 103:413–420. https://doi.org/10.1016/j.ajhg.2018.07.013
Beckers A, Adis C, Schuster-Gossler K et al (2020) The FOXJ1 target Cfap206 is required for sperm motility, mucociliary clearance of the airways and brain development. Development. https://doi.org/10.1242/dev.188052
Ben Khelifa M, Coutton C, Zouari R et al (2014) Mutations in DNAH1, which encodes an inner arm heavy chain dynein, lead to male infertility from multiple morphological abnormalities of the sperm flagella. Am J Hum Genet 94:95–104. https://doi.org/10.1016/j.ajhg.2013.11.017
Blackburn K, Bustamante-Marin X, Yin W et al (2017) Quantitative proteomic analysis of human airway cilia identifies previously uncharacterized proteins of high abundance. J Proteome Res 16:1579–1592. https://doi.org/10.1021/acs.jproteome.6b00972
Brown JM, Witman GB (2014) Cilia and diseases. Bioscience 64:1126–1137. https://doi.org/10.1093/biosci/biu174
Coutton C, Vargas AS, Amiri-Yekta A et al (2018) Mutations in CFAP43 and CFAP44 cause male infertility and flagellum defects in trypanosoma and human. Nat Commun 9:686. https://doi.org/10.1038/s41467-017-02792-7
Coutton C, Martinez G, Kherraf Z-E et al (2019) Bi-allelic mutations in ARMC2 lead to severe astheno-teratozoospermia due to sperm flagellum malformations in humans and mice. Am J Hum Genet 104:331–340. https://doi.org/10.1016/j.ajhg.2018.12.013
Dymek EE, Smith EF (2007) A conserved CaM- and radial spoke associated complex mediates regulation of flagellar dynein activity. J Cell Biol 179:515–526. https://doi.org/10.1083/jcb.200703107
Inaba K (2007) Molecular basis of sperm flagellar axonemes: structural and evolutionary aspects. Ann N Y Acad Sci 1101:506–526. https://doi.org/10.1196/annals.1389.017
Kherraf Z-E, Amiri-Yekta A, Dacheux D et al (2018) A Homozygous ancestral SVA-insertion-mediated deletion in WDR66 induces multiple morphological abnormalities of the sperm flagellum and male infertility. Am J Hum Genet 103:400–412. https://doi.org/10.1016/j.ajhg.2018.07.014
Kott E, Legendre M, Copin B et al (2013) Loss-of-function mutations in RSPH1 cause primary ciliary dyskinesia with central-complex and radial-spoke defects. Am J Hum Genet 93:561–570. https://doi.org/10.1016/j.ajhg.2013.07.013
Krausz C, Riera-Escamilla A (2018) Genetics of male infertility. Nat Rev Urol 15:369–384. https://doi.org/10.1038/s41585-018-0003-3
Lindemann CB, Lesich KA (2016) Functional anatomy of the mammalian sperm flagellum. Cytoskeleton (hoboken) 73:652–669. https://doi.org/10.1002/cm.21338
Liu C, He X, Liu W et al (2019a) Bi-allelic mutations in TTC29 cause male subfertility with asthenoteratospermia in humans and mice. Am J Hum Genet 105:1168–1181. https://doi.org/10.1016/j.ajhg.2019.10.010
Liu W, He X, Yang S et al (2019b) Bi-allelic mutations in TTC21A induce asthenoteratospermia in humans and mice. Am J Hum Genet 104:738–748. https://doi.org/10.1016/j.ajhg.2019.02.020
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lorès P, Kheraff Z-E, Amiri-Yekta A et al (2021) A missense mutation in IFT74, encoding for an essential component for intraflagellar transport of Tubulin, causes asthenozoospermia and male infertility without clinical signs of Bardet-Biedl syndrome. Hum Genet. https://doi.org/10.1007/s00439-021-02270-7
Martinez G, Beurois J, Dacheux D et al (2020) Biallelic variants in MAATS1 encoding CFAP91, a calmodulin-associated and spoke-associated complex protein, cause severe astheno-teratozoospermia and male infertility. J Med Genet 57:708–716. https://doi.org/10.1136/jmedgenet-2019-106775
McLaren W, Gil L, Hunt SE et al (2016) The ensembl variant effect predictor. Genome Biol 17:122. https://doi.org/10.1186/s13059-016-0974-4
Morimoto Y, Yoshida S, Kinoshita A et al (2019) Nonsense mutation in CFAP43 causes normal-pressure hydrocephalus with ciliary abnormalities. Neurology 92:e2364–e2374. https://doi.org/10.1212/WNL.0000000000007505
Rachev E, Schuster-Gossler K, Fuhl F et al (2020) CFAP43 modulates ciliary beating in mouse and Xenopus. Dev Biol 459:109–125. https://doi.org/10.1016/j.ydbio.2019.12.010
Ron-El R, Liu J, Nagy Z et al (1995) Intracytoplasmic sperm injection in the mouse. Hum Reprod 10:2831–2834. https://doi.org/10.1093/oxfordjournals.humrep.a135802
Touré A, Martinez G, Kherraf Z-E et al (2021) The genetic architecture of morphological abnormalities of the sperm tail. Hum Genet 140:21–42. https://doi.org/10.1007/s00439-020-02113-x
Urbanska P, Song K, Joachimiak E et al (2015) The CSC proteins FAP61 and FAP251 build the basal substructures of radial spoke 3 in cilia. Mol Biol Cell 26:1463–1475. https://doi.org/10.1091/mbc.E14-11-1545
Vasudevan KK, Song K, Alford LM et al (2015) FAP206 is a microtubule-docking adapter for ciliary radial spoke 2 and dynein c. Mol Biol Cell 26:696–710. https://doi.org/10.1091/mbc.E14-11-1506
Wambergue C, Zouari R, Fourati Ben Mustapha S et al (2016) Patients with multiple morphological abnormalities of the sperm flagella due to DNAH1 mutations have a good prognosis following intracytoplasmic sperm injection. Hum Reprod 31:1164–1172. https://doi.org/10.1093/humrep/dew083
Wang G, Guo Y, Zhou T et al (2013) In-depth proteomic analysis of the human sperm reveals complex protein compositions. J Proteomics 79:114–122. https://doi.org/10.1016/j.jprot.2012.12.008
Wang Y, Yang J, Jia Y et al (2014) Variability in the morphologic assessment of human sperm: use of the strict criteria recommended by the World Health Organization in 2010. Fertil Steril. https://doi.org/10.1016/j.fertnstert.2013.12.047
Yang H, Wang H, Jaenisch R (2014) Generating genetically modified mice using CRISPR/Cas-mediated genome engineering. Nat Protoc 9:1956–1968. https://doi.org/10.1038/nprot.2014.134
Acknowledgements
We thank the patient for their participation in this study as well as all the referring physicians.
Funding
This work was mainly supported by the following grants by the Agence Nationale de la Recherche (ANR) MASFLAGELLA (ANR-14-CE15-0002) and FLAGEL-OME (ANR-19-CE17-0014). This work was also supported by the National Key R&D Program of China [Grant number 2019YFC1005106]; Supported by the Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences (2019PT310002; the National Natural Science Foundation of China [Grant numbers 81901541 and 81971441].
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. CCo, YXC, PFR, FZ, XJH and XA designed and supervised the study. AA-Y, VS recruited the patients, performed clinical analysis and characterization. NTM, ZEK, PFR, TC and CCo performed exome and bioinformatics analysis. GM, JB, CCa, MB and TC performed patient’s experimental work. HBL and QSS provided Cfap206 knockout mice. BS, MRL and QSS performed animal’s experimental work. QSS and DL performed ICSI of mice. GM, CCA, KKL, FBT and XJH performed data illustrations. XA, FZ, PFR, AT, MW, CA, HW and CCo analyzed the data. XA, GM, QSS and CCo wrote the manuscript. The first draft of the manuscript was written by XA, GM and CCo, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethical approval
The study was approved by local ethics committees, and samples were then stored in the CRB Germethèque (certification under ISO-9001 and NF-S 96-900) following a standardized procedure or were part of the Fertithèque collection declared to the French Ministry of health (DC-2015-2580) and the French Data Protection Authority (DR-2016-392).
Animal research
All animal procedures were run according to the Chinese guidelines on the use of animals in scientific investigations with the approval of the animal Ethics Committee at the First Affiliated Hospital of Anhui Medical University (P2020-12-36).
Consent to participate
Informed consent was obtained from all individuals included in the study.
Consent to publish
Agreement for data publication was obtained from all individuals included in the study.
Web resources
ClinVar, https://www.ncbi.nlm.nih.gov/clinvar/
gnomAD Browser, http://gnomad.broadinstitute.org
GTEx, https://gtexportal.org
Uniprot, https://www.uniprot.org/
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shen, Q., Martinez, G., Liu, H. et al. Bi-allelic truncating variants in CFAP206 cause male infertility in human and mouse. Hum Genet 140, 1367–1377 (2021). https://doi.org/10.1007/s00439-021-02313-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-021-02313-z