Abstract
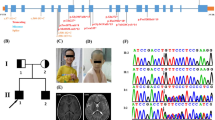
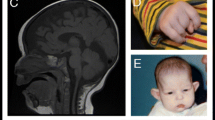
Intellectual disability (ID) has an estimated prevalence of 1.5–2%. In most affected individuals, its genetic basis remains unclear. Whole exome sequencing (WES) studies have identified a multitude of novel causative gene defects and have shown that a large proportion of sporadic ID cases results from de novo mutations. Here, we present two unrelated individuals with similar clinical features and deleterious de novo variants in FBXO11 detected by WES. Individual 1, a 14-year-old boy, has mild ID as well as mild microcephaly, corrected cleft lip and alveolus, hyperkinetic disorder, mild brain atrophy and minor facial dysmorphism. WES detected a heterozygous de novo 1 bp insertion in the splice donor site of exon 3. Individual 2, a 3-year-old boy, showed ID and pre- and postnatal growth retardation, postnatal mild microcephaly, hyperkinetic and restless behaviour, as well as mild dysmorphism. WES detected a heterozygous de novo frameshift mutation. While ten individuals with ID and de novo variants in FBXO11 have been reported as part of larger studies, only one of the reports has some additional clinical data. Interestingly, the latter individual carries the identical mutation as our individual 2 and also displays ID, intrauterine growth retardation, microcephaly, behavioural anomalies, and dysmorphisms. Thus, we confirm deleterious de novo mutations in FBXO11 as a cause of ID and start the delineation of the associated clinical picture which may also comprise postnatal microcephaly or borderline small head size and behavioural anomalies.



Similar content being viewed by others
References
Aberg KA et al (2013) A comprehensive family-based replication study of schizophrenia genes. JAMA Psychiatry 70:573–581. https://doi.org/10.1001/jamapsychiatry.2013.288
Abida WM, Nikolaev A, Zhao W, Zhang W, Gu W (2007) FBXO11 promotes the Neddylation of p53 and inhibits its transcriptional activity. J Biol Chem 282:1797–1804. https://doi.org/10.1074/jbc.M609001200
Adzhubei I, Jordan DM, Sunyaev SR (2013) Predicting functional effect of human missense mutations using PolyPhen-2. Curr Protoc Hum Genet. https://doi.org/10.1002/0471142905.hg0720s76
Bhutta MF et al (2017) A mouse-to-man candidate gene study identifies association of chronic otitis media with the loci TGIF1 and FBXO11. Sci Rep 7:12496. https://doi.org/10.1038/s41598-017-12784-8
Boot MV et al (2018) Benign and malignant tumors in Rubinstein–Taybi syndrome. Am J Med Genet A 176:597–608. https://doi.org/10.1002/ajmg.a.38603
Cenciarelli C, Chiaur DS, Guardavaccaro D, Parks W, Vidal M, Pagano M (1999) Identification of a family of human F-box proteins. Curr Biol 9:1177–1179. https://doi.org/10.1016/S0960-9822(00)80020-2
Cooper GM, Stone EA, Asimenos G, Program NCS, Green ED, Batzoglou S, Sidow A (2005) Distribution and intensity of constraint in mammalian genomic sequence. Genome Res 15:901–913. https://doi.org/10.1101/gr.3577405
Davydov EV, Goode DL, Sirota M, Cooper GM, Sidow A, Batzoglou S (2010) Identifying a high fraction of the human genome to be under selective constraint using GERP++. PLoS Comput Biol 6:e1001025. https://doi.org/10.1371/journal.pcbi.1001025
de Ligt J et al (2012) Diagnostic exome sequencing in persons with severe intellectual disability. N Engl J Med 367:1921–1929. https://doi.org/10.1056/NEJMoa1206524
Deciphering Developmental Disorders S (2017) Prevalence and architecture of de novo mutations in developmental disorders. Nature 542:433–438. https://doi.org/10.1038/nature21062
den Dunnen JT, Antonarakis SE (2000) Mutation nomenclature extensions and suggestions to describe complex mutations: a discussion. Hum Mutat 15:7–12. https://doi.org/10.1002/(SICI)1098-1004(200001)15:1<7::AID-HUMU4>3.0.CO;2-N
Duan S et al (2012) FBXO11 targets BCL6 for degradation and is inactivated in diffuse large B-cell lymphomas. Nature 481:90–93. https://doi.org/10.1038/nature10688
Froyen G et al (2012) Copy-number gains of HUWE1 due to replication- and recombination-based rearrangements. Am J Hum Genet 91:252–264. https://doi.org/10.1016/j.ajhg.2012.06.010
Hamdan FF et al (2014) De novo mutations in moderate or severe intellectual disability. PLoS Genet 10:e1004772. https://doi.org/10.1371/journal.pgen.1004772
Hanus C, Schuman EM (2013) Proteostasis in complex dendrites. Nat Rev Neurosci 14:638–648. https://doi.org/10.1038/nrn3546
Hardisty-Hughes RE et al (2006) A mutation in the F-box gene, Fbxo11, causes otitis media in the Jeff mouse. Hum Mol Genet 15:3273–3279. https://doi.org/10.1093/hmg/ddl403
He X et al (2013) Integrated model of de novo and inherited genetic variants yields greater power to identify risk genes. PLoS Genet 9:e1003671. https://doi.org/10.1371/journal.pgen.1003671
Kircher M, Witten DM, Jain P, O’Roak BJ, Cooper GM, Shendure J (2014) A general framework for estimating the relative pathogenicity of human genetic variants. Nat Genet 46:310–315. https://doi.org/10.1038/ng.2892
Kochinke K et al (2016) Systematic phenomics analysis deconvolutes genes mutated in intellectual disability into biologically coherent modules. Am J Hum Genet 98:149–164
Kumar P, Henikoff S, Ng PC (2009) Predicting the effects of coding non-synonymous variants on protein function using the SIFT algorithm. Nat Protoc 4:1073–1081. https://doi.org/10.1038/nprot.2009.86
Lek M et al (2016) Analysis of protein-coding genetic variation in 60,706 humans. Nature 536:285–291. https://doi.org/10.1038/nature19057
Lelieveld SH et al (2016) Meta-analysis of 2,104 trios provides support for 10 new genes for intellectual disability. Nat Neurosci 19:1194–1196. https://doi.org/10.1038/nn.4352
Leonard H, Wen X (2002) The epidemiology of mental retardation: challenges and opportunities in the new millennium. Ment Retard Dev Disabil Res Rev 8:117–134. https://doi.org/10.1002/mrdd.10031
Louros SR, Osterweil EK (2016) Perturbed proteostasis in autism spectrum disorders. J Neurochem 139:1081–1092. https://doi.org/10.1111/jnc.13723
Martínez F, Caro-Llopis A, Rosello M, Oltra S, Mayo S, Monfort S, Orellana C (2017) High diagnostic yield of syndromic intellectual disability by targeted next-generation sequencing. J Med Genet 54:87–92. https://doi.org/10.1136/jmedgenet-2016-103964
Miller RW, Rubinstein JH (1995) Tumors in Rubinstein–Taybi syndrome. Am J Med Genet 56:112–115. https://doi.org/10.1002/ajmg.1320560125
Moortgat S et al (2018) HUWE1 variants cause dominant X-linked intellectual disability: a clinical study of 21 patients. Eur J Hum Genet 26:64–74. https://doi.org/10.1038/s41431-017-0038-6
Najmabadi H et al (2011) Deep sequencing reveals 50 novel genes for recessive cognitive disorders. Nature 478:57–63. https://doi.org/10.1038/nature10423
Nguyen HT et al (2017) Integrated Bayesian analysis of rare exonic variants to identify risk genes for schizophrenia and neurodevelopmental disorders. Genome Med 9:114. https://doi.org/10.1186/s13073-017-0497-y
Pasqualucci L et al (2011) Inactivating mutations of acetyltransferase genes in B-cell lymphoma. Nature 471:189–195. https://doi.org/10.1038/nature09730
Plon SE et al (2008) Sequence variant classification and reporting: recommendations for improving the interpretation of cancer susceptibility genetic test results. Hum Mutat 29:1282–1291. https://doi.org/10.1002/humu.20880
Rauch A et al (2012) Range of genetic mutations associated with severe non-syndromic sporadic intellectual disability: an exome sequencing study. Lancet 380:1674–1682. https://doi.org/10.1016/S0140-6736(12)61480-9
Roy A, Yang J, Zhang Y (2012) COFACTOR: an accurate comparative algorithm for structure-based protein function annotation. Nucleic Acids Res 40:W471–W477. https://doi.org/10.1093/nar/gks372
Rye MS, Blackwell JM, Jamieson SE (2012) Genetic susceptibility to otitis media in childhood. Laryngoscope 122:665–675. https://doi.org/10.1002/lary.22506
Sanders SJ et al (2012) De novo mutations revealed by whole-exome sequencing are strongly associated with autism. Nature 485:237–241. https://doi.org/10.1038/nature10945
Schäfgen J et al (2016) De novo nonsense and frameshift variants of TCF20 in individuals with intellectual disability and postnatal overgrowth. Eur J Hum Genet 24:1739–1745. https://doi.org/10.1038/ejhg.2016.90
Schneider C et al (2016) FBXO11 inactivation leads to abnormal germinal-center formation and lymphoproliferative disease. Blood 128:660–666. https://doi.org/10.1182/blood-2015-11-684357
Schwarz JM, Rodelsperger C, Schuelke M, Seelow D (2010) MutationTaster evaluates disease-causing potential of sequence alterations. Nat Methods 7:575–576. https://doi.org/10.1038/nmeth0810-575
Villani A et al (2017) Recommendations for cancer surveillance in individuals with RASopathies and other rare genetic conditions with increased cancer risk. Clin Cancer Res 23:e83–e90. https://doi.org/10.1158/1078-0432.CCR-17-0631
Wright CF et al (2015) Genetic diagnosis of developmental disorders in the DDD study: a scalable analysis of genome-wide research data. Lancet 385:1305–1314. https://doi.org/10.1016/S0140-6736(14)61705-0
Xie Y, Varshavsky A (1999) The E2-E3 interaction in the N-end rule pathway: the RING-H2 finger of E3 is required for the synthesis of multiubiquitin chain. EMBO J 18:6832–6844. https://doi.org/10.1093/emboj/18.23.6832
Yang J, Zhang Y (2015) I-TASSER server: new development for protein structure and function predictions. Nucleic Acids Res 43:W174-181. https://doi.org/10.1093/nar/gkv342
Zhang Y (2009) I-TASSER: fully automated protein structure prediction in CASP8. Proteins 77(Suppl 9):100–113. https://doi.org/10.1002/prot.22588
Acknowledgements
We thank the patients and their families for their participation in this study. This work was supported in part by the German Ministry of Research and Education/Bundesministerium für Bildung und Forschung BMBF (Grant Numbers 01GS08164, 01GS08167, 01GS08163 to HE, DW and TMS, German Mental Retardation Network) as part of the National Genome Research Network and through the Juniorverbund in der Systemmedizin “mitOmics” (01ZX1405C to TBH). This study makes use of data generated by the DECIPHER community. A full list of centres who contributed to the generation of the data is available from http://decipher.sanger.ac.uk and via email from decipher@sanger.ac.uk. Funding for the project was provided by the Wellcome Trust.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
Informed consent was obtained for all individual participants included in the study. Additional informed consent was obtained for all individual participants for whom identifying information is included in this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fritzen, D., Kuechler, A., Grimmel, M. et al. De novo FBXO11 mutations are associated with intellectual disability and behavioural anomalies. Hum Genet 137, 401–411 (2018). https://doi.org/10.1007/s00439-018-1892-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-018-1892-1