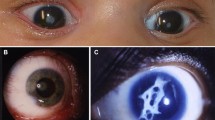
Abstract
Pediatric cataract is highly heterogeneous clinically and etiologically. While mostly isolated, cataract can be part of many multisystem disorders, further complicating the diagnostic process. In this study, we applied genomic tools in the form of a multi-gene panel as well as whole-exome sequencing on unselected cohort of pediatric cataract (166 patients from 74 families). Mutations in previously reported cataract genes were identified in 58% for a total of 43 mutations, including 15 that are novel. GEMIN4 was independently mutated in families with a syndrome of cataract, global developmental delay with or without renal involvement. We also highlight a recognizable syndrome that resembles galactosemia (a fulminant infantile liver disease with cataract) caused by biallelic mutations in CYP51A1. A founder mutation in RIC1 (KIAA1432) was identified in patients with cataract, brain atrophy, microcephaly with or without cleft lip and palate. For non-syndromic pediatric cataract, we map a novel locus in a multiplex consanguineous family on 4p15.32 where exome sequencing revealed a homozygous truncating mutation in TAPT1. We report two further candidates that are biallelically inactivated each in a single cataract family: TAF1A (cataract with global developmental delay) and WDR87 (non-syndromic cataract). In addition to positional mapping data, we use iSyTE developmental lens expression and gene-network analysis to corroborate the proposed link between the novel candidate genes and cataract. Our study expands the phenotypic, allelic and locus heterogeneity of pediatric cataract. The high diagnostic yield of clinical genomics supports the adoption of this approach in this patient group.



Similar content being viewed by others
References
Acimovic J, Rozman D (2013) Steroidal triterpenes of cholesterol synthesis. Molecules 18(4):4002–4017
Agrawal SA, Anand D, Siddam AD et al (2015) Compound mouse mutants of bZIP transcription factors Mafg and Mafk reveal a regulatory network of non-crystalline genes associated with cataract. Hum Genet 134(7):717–735
Akiyama M, Ishida N, Ogawa T et al (2005) Molecular cloning and functional analysis of a novel Cx43 partner protein CIP150. Biochem Biophys Res Commun 335(4):1264–1271
Alazami AM, Patel N, Shamseldin HE et al (2015) Accelerating novel candidate gene discovery in neurogenetic disorders via whole-exome sequencing of prescreened multiplex consanguineous families. Cell Rep 10(2):148–161
Alazami AM, Al-Qattan SM, Faqeih E et al (2016) Expanding the clinical and genetic heterogeneity of hereditary disorders of connective tissue. Hum Genet 135(5):525–540
Aldahmesh MA, Khan AO, Mohamed J, Alkuraya FS (2011) Novel recessive BFSP2 and PITX3 mutations: insights into mutational mechanisms from consanguineous populations. Genet Med 13(11):978–981
Aldahmesh MA, Khan AO, Mohamed JY et al (2012a) Identification of a truncation mutation of acylglycerol kinase (AGK) gene in a novel autosomal recessive cataract locus. Hum Mutat 33(6):960–962
Aldahmesh MA, Khan AO, Mohamed JY et al (2012b) Genomic analysis of pediatric cataract in Saudi Arabia reveals novel candidate disease genes. Genet Med 14(12):955–962
Alkuraya FS (2010) Autozygome decoded. Genet Med 12(12):765–771
Alkuraya FS (2012) Discovery of rare homozygous mutations from studies of consanguineous pedigrees. Curr Protoc Hum Genet 75:6.12.1–6.12.13
Alkuraya FS (2013) The application of next-generation sequencing in the autozygosity mapping of human recessive diseases. Hum Genet 132(11):1197–1211
Alkuraya FS (2016) Discovery of mutations for Mendelian disorders. Hum Genet 135(6):615–623
Anand D, Lachke SA (2016) Systems biology of lens development: a paradigm for disease gene discovery in the eye. Exp Eye Res. doi:10.1016/j.exer.2016.03.010
Anand D, Agrawal S, Siddam A et al (2015) An integrative approach to analyze microarray datasets for prioritization of genes relevant to lens biology and disease. Genom Data 5:223–227
Anazi S, Maddirevula S, Faqeih E et al (2016) Clinical genomics expands the morbid genome of intellectual disability and offers a high diagnostic yield. Mol Psychiatry. doi:10.1038/mp.2016.113
Audette DS, Anand D, So T et al (2016) Prox1 and fibroblast growth factor receptors form a novel regulatory loop controlling lens fiber differentiation and gene expression. Development 143(2):318–328
Charroux B, Pellizzoni L, Perkinson RA et al (2000) Gemin4 a novel component of the SMN complex that is found in both gems and nucleoli. J Cell Biol 148(6):1177–1186
Dash S, Dang CA, Beebe DC, Lachke SA (2015) Deficiency of the RNA binding protein caprin2 causes lens defects and features of Peters anomaly. Dev Dyn 244(10):1313–1327
Fischer U, Liu Q, Dreyfuss G (1997) The SMN–SIP1 complex has an essential role in spliceosomal snRNP biogenesis. Cell 90(6):1023–1029
Foster A, Gilbert C, Rahi J (1997) Epidemiology of cataract in childhood: a global perspective. J Cataract Refract Surg 23:601–604
Gautier L, Cope L, Bolstad BM, Irizarry RA (2004) affy—analysis of Affymetrix GeneChip data at the probe level. Bioinformatics 20(3):307–315
Gillespie RL, O’Sullivan J, Ashworth J et al (2014) Personalized diagnosis and management of congenital cataract by next-generation sequencing. Ophthalmology 121(11):2124–2137. e2
Gillespie RL, O’Sullivan J, Ashworth J et al (2014) Personalized diagnosis and management of congenital cataract by next-generation sequencing. Ophthalmology 121(11):2124–37 e1–2
Gillespie RL, Urquhart J, Anderson B et al (2016) Next-generation sequencing in the diagnosis of metabolic disease marked by pediatric cataract. Ophthalmology 123(1):217–220
Group SM (2015) Comprehensive gene panels provide advantages over clinical exome sequencing for Mendelian diseases. Genome Biol 16:134
Hejtmancik JF (2008) Congenital cataracts and their molecular genetics. Semin Cell Dev Biol 19(2):134–149
Hoang TV, Kumar PKR, Sutharzan S et al (2014) Comparative transcriptome analysis of epithelial and fiber cells in newborn mouse lenses with RNA sequencing. Mol Vis 20:1491–1517
Kasaikina MV, Fomenko DE, Labunskyy VM et al (2011) Roles of the 15-kDa selenoprotein (Sep15) in redox homeostasis and cataract development revealed by the analysis of Sep 15 knockout mice. J Biol Chem 286(38):33203–33212
Keber R, Motaln H, Wagner KD et al (2011) Mouse knockout of the cholesterogenic cytochrome P450 lanosterol 14alpha-demethylase (Cyp51) resembles Antley–Bixler syndrome. J Biol Chem 286(33):29086–29097
Khan AO (2012) Hereditary pediatric cataract on the Arabian Peninsula. Saudi J Ophthalmol 26(1):67–71
Khan AO (2013) Ocular genetic disease in the Middle East. Curr Opin Ophthalmol 24(5):369–378
Khan AO, Aldahmesh MA, Alkuraya FS (2015) Phenotypes of recessive pediatric cataract in a cohort of children with identified homozygous gene mutations (an American ophthalmological society thesis). Trans Am Ophthalmol Soc 113:T7-1
Lachke SA, Alkuraya FS, Kneeland SC et al (2011) Mutations in the RNA granule component TDRD7 cause cataract and glaucoma. Science 331(6024):1571–1576
Lachke SA, Ho JW, Kryukov GV et al (2012a) iSyTE: integrated Systems Tool for Eye gene discovery. Invest Ophthalmol Vis Sci 53(3):1617–1627
Lachke SA, Higgins AW, Inagaki M et al (2012b) The cell adhesion gene PVRL3 is associated with congenital ocular defects. Hum Genet 131(2):235–250
Lassen N, Bateman JB, Estey T et al (2007) Multiple and additive functions of ALDH3A1 and ALDH1A1: cataract phenotype and ocular oxidative damage in Aldh3a1(−/−)/Aldh1a1(−/−) knock-out mice. J Biol Chem 282(35):25668–25676
Lorbek G, Perse M, Jeruc J et al (2015) Lessons from hepatocyte-specific Cyp51 knockout mice: impaired cholesterol synthesis leads to oval cell-driven liver injury. Sci Rep 5:8777
Lorson MA, Dickson AM, Shaw DJ et al (2008) Identification and characterisation of a nuclear localisation signal in the SMN associated protein, Gemin4. Biochem Biophys Res Commun 375(1):33–37
Ma AS, Grigg JR, Ho G, Prokudin I, Farnsworth E, Holman K, Cheng A, Billson FA, Martin F, Fraser C, Mowat D, Smith J, Christodoulou J, Flaherty M, Bennetts B, Jamieson RV (2016) Sporadic and familial congenital cataracts: mutational spectrum and new diagnoses using next-generation sequencing. Hum Mutat 37(4):371–384. http://doi.org/10.1002/humu.22948
Makley LN, McMenimen KA, DeVree BT et al (2015) Pharmacological chaperone for alpha-crystalline partially restores transparency in cataract models. Science 350(6261):674–677
Manthey AL, Lachke SA, FitzGerald PG et al (2014) Loss of Sip1 leads to migration defects and retention of ectodermal markers during lens development. Mech Dev 131:86–110
Medsinge A, Nischal KK (2015) Pediatric cataract: challenges and future directions. Clin Ophthalmol (Auckland, NZ) 9:77
Musleh M, Hall G, Lloyd I et al (2016) Diagnosing the cause of bilateral paediatric cataracts: comparison of standard testing with a next-generation sequencing approach. Eye (Lond) 30(9):1175–1181
Patel N, Aldahmesh MA, Alkuraya H et al (2015) Expanding the clinical, allelic, and locus heterogeneity of retinal dystrophies. Genet Med 18(6):554–562
Paznekas WA, Boyadjiev SA, Shapiro RE et al (2003) Connexin 43 (GJA1) mutations cause the pleiotropic phenotype of oculodentodigital dysplasia. Am J Hum Genet 72(2):408–418
Rahi JS, Dezateaux C (2001) Measuring and interpreting the incidence of congenital ocular anomalies: lessons from a national study of congenital cataract in the UK. Invest Ophthalmol Vis Sci 42(7):1444–1448
Safieh LA, Khan A, Alkuraya F (2009) Identification of a novel CRYAB mutation associated with autosomal recessive juvenile cataract in a Saudi family. Mol Vis 15:980–984
Shiels A, Hejtmancik JF (2007) Genetic origins of cataract. Arch Ophthalmol 125(2):165–173
Shiels A, Hejtmancik J (2013) Genetics of human cataract. Clin Genet 84(2):120–127
Shiels A, Bennett TM, Hejtmancik JF (2010) Cat-Map: putting cataract on the map. Mol Vis 16:2007–2015
Stayte M, Reeves B, Wortham C (1993) Ocular and vision defects in preschool children. Br J Ophthalmol 77(4):228–232
Symoens S, Barnes AM, Gistelinck C et al (2015) Genetic defects in TAPT1 disrupt ciliogenesis and cause a complex lethal osteochondrodysplasia. Am J Hum Genet 97(4):521–534
Wolf L, Harrison W, Huang J et al (2013) Histone posttranslational modifications and cell fate determination: lens induction requires the lysine acetyltransferases CBP and p300. Nucleic Acids Res 41(22):10199–10214
Zhang H, Xie L, Wu X, Tian J (2012) Long-term results of pediatric cataract surgery after delayed diagnosis. J Am Assoc Pediatr Ophthalmol Strabismus 16(1):65–69
Zhao L, Chen XJ, Zhu J et al (2015) Lanosterol reverses protein aggregation in cataracts. Nature 523(7562):607–611
Acknowledgements
This study was supported by King Salman Center for Disability Research (FSA) and the National Eye Institute of the National Institutes of Health under Award Number R01EY021505 (SAL). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. SAL is a Pew Scholar in Biomedical Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
439_2016_1747_MOESM1_ESM.pdf
Figure S1. CYP51A1 is a novel candidate gene for syndromic cataract. (A) Pedigree for family 16DG0226. (B) Agile mapper ideogram reveals a single shared haplotype in the two affected (dark blue) that is not present in the unaffected individuals (pink) on Chr7. * Denotes a haplotype that is not unique to the affected individuals. (C) DNA sequence chromatogram of the missense mutation found in CYP51A1 that segregates within the family. Figure S2. TAPT1 is a novel candidate gene for isolated cataract. (A) Pedigree for family 12DG2657. (B) Agile mapper ideogram reveals a single locus on Chr4 (dark blue). (C) Genomic DNA sequence chromatogram of TAPT1 shows 1 bp insertion downstream of exon 6 that segregates with the family. (D) cDNA sequence chromatogram shows deletion of part of exon 5 and skipping of exon 6 in the aberrant band compared to the wild-type allele. Figure S3. RIC1 is a novel candidate gene for syndromic cataract. (A) Pedigree for two individual families with syndromic cataract. (B) Agile mapper ideogram reveals a locus for RIC1 on Chr9 (dark blue) (*denotes a haplotype that is not unique to the affected individuals). (C) Chromatogram of RIC1 shows missense change G>C that segregates with the family. Figure S4. TAF1A is a novel candidate gene for syndromic pediatric cataract. (A) Pedigree for family 11DG2176. (B) Sequence chromatogram for frameshift deletion mutation in TAF1A that segregates within the family. Figure S5. WDR87 is a novel candidate gene for pediatric cataract. (A) Pedigree for family 12DG2386. (B) Sequence chromatogram for stop-gain mutation in WDR87 that segregates within the family. Figure S6. Lens-expression curated PPI-network for non-syndromic cataract genes TAPT1 and RIC1 identifies candidates with known function or expression in the eye. (A, B) Integrated analysis-derived network using PPI data from String database (human interactions) and expression data from iSyTE E10.5 lens expression database identifies TAPT1 network with 22 direct connections and their interacting partners. Of these 22 interacting candidates, 15 are expressed in the lens, of which 7 are lens-enriched. iSyTE overlay identifies candidates with high lens expression such as SUCO, ANXA5, CALR. Proteins with known function in the lens, such as CDKN1A, CDKN1B, NOTCH1, SMAD3 are connected to direct partners of TAPT1 and are expressed in the network at E10.5. (C, D) Integrated analysis-derived network using PPI data from String database (human interactions) and lens expression identifies 39 direct interactors of RIC1, of which 32 candidates exhibit lens expression, and 14 are lens-enriched. GJA1, which is linked to microphthalmia and cataracts in humans is directly connected to RIC1 in the network. Color key represents high (deep red) to low (green) enrichment score and high (deep red) to low (white) expression scores. Genes absent in iSyTE are color-coded as grey. Figure S7. Lens-expression curated PPI-network for the syndromic cataract genes GEMIN4 and CYP51A1 identifies candidates with known function or expression in the eye. Integrated analysis-derived network using PPI data from String database (human interactions) and expression data from iSyTE E10.5 lens gene expression database identifies direct interacting proteins of GEMIN4 and CYP51A1. (A, B) GEMIN4 is connected to 53 protein-protein interacting partners, of which 50 candidates exhibit lens expression and 37 exhibit lens-enrichment. This analysis identifies a cluster of lens-expressed proteins such as GEMIN5, GEMIN6, GEMIN7, GEMIN8. (C, D) Network analysis demonstrates that CYP51A1 is directly connected to 51 interacting proteins, of which 35 candidates exhibit lens expression and 22 exhibit lens-enrichment. CYP51A1 is directly connected to ALDH1A1 and MAFG, both of which are linked to cataracts in mice. Several candidates for sterol biosynthetic process (GO:0016126) categories are enriched and expressed in the network. Color key represents high (deep red) to low (green) enrichment score and high (deep red) to low (white) expression scores. Genes absent in iSyTE are color-coded as grey. (PDF 3128 kb)
Rights and permissions
About this article
Cite this article
Patel, N., Anand, D., Monies, D. et al. Novel phenotypes and loci identified through clinical genomics approaches to pediatric cataract. Hum Genet 136, 205–225 (2017). https://doi.org/10.1007/s00439-016-1747-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-016-1747-6