Abstract
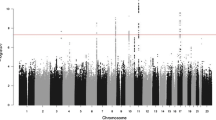
Age-adjusted mortality rates for prostate cancer are higher for African-American men compared with those of European ancestry. Recent data suggest that West African men also have elevated risk for prostate cancer relative to European men. Genetic susceptibility to prostate cancer could account for part of this difference. We conducted a genome-wide association study (GWAS) of prostate cancer in West African men in the Ghana Prostate Study. Association testing was performed using multivariable logistic regression adjusted for age and genetic ancestry for 474 prostate cancer cases and 458 population-based controls on the Illumina HumanOmni-5 Quad BeadChip. The most promising association was at 10p14 within an intron of a long non-coding RNA (lncRNA RP11-543F8.2) 360 kb centromeric of GATA3 (p = 1.29E−7). In sub-analyses, SNPs at 5q31.3 were associated with high Gleason score (≥7) cancers, the strongest of which was a missense SNP in PCDHA1 (rs34575154, p = 3.66E−8), and SNPs at Xq28 (rs985081, p = 8.66E−9) and 6q21 (rs2185710, p = 5.95E−8) were associated with low Gleason score (<7) cancers. We sought to validate our findings in silico in the African Ancestry Prostate Cancer GWAS Consortium, but only one SNP, at 10p14, replicated at p < 0.05. Of the 90 prostate cancer loci reported from studies of men of European, Asian or African-American ancestry, we were able to test 81 in the Ghana Prostate Study, and 10 of these replicated at p < 0.05. Further genetic studies of prostate cancer in West African men are needed to confirm our promising susceptibility loci.

Similar content being viewed by others
References
Adeyemo A, Rotimi C (2010) Genetic variants associated with complex human diseases show wide variation across multiple populations. Public Health Genomics 13:72–79. doi:10.1159/000218711
Akamatsu S, Takata R, Haiman CA, Takahashi A, Inoue T, Kubo M, Furihata M, Kamatani N, Inazawa J, Chen GK, Le Marchand L, Kolonel LN, Katoh T, Yamano Y, Yamakado M, Takahashi H, Yamada H, Egawa S, Fujioka T, Henderson BE, Habuchi T, Ogawa O, Nakamura Y, Nakagawa H (2012) Common variants at 11q12, 10q26 and 3p11.2 are associated with prostate cancer susceptibility in Japanese. Nat Genet 44(426–9):S1. doi:10.1038/ng.1104
Amundadottir LT, Sulem P, Gudmundsson J, Helgason A, Baker A, Agnarsson BA, Sigurdsson A, Benediktsdottir KR, Cazier JB, Sainz J, Jakobsdottir M, Kostic J, Magnusdottir DN, Ghosh S, Agnarsson K, Birgisdottir B, Le Roux L, Olafsdottir A, Blondal T, Andresdottir M, Gretarsdottir OS, Bergthorsson JT, Gudbjartsson D, Gylfason A, Thorleifsson G, Manolescu A, Kristjansson K, Geirsson G, Isaksson H, Douglas J, Johansson JE, Balter K, Wiklund F, Montie JE, Yu X, Suarez BK, Ober C, Cooney KA, Gronberg H, Catalona WJ, Einarsson GV, Barkardottir RB, Gulcher JR, Kong A, Thorsteinsdottir U, Stefansson K (2006) A common variant associated with prostate cancer in European and African populations. Nat Genet 38:652–658
Barrett JC, Fry B, Maller J, Daly MJ (2005) Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21:263–265. doi:10.1093/bioinformatics/bth457
Batra J, Lose F, Chambers S, Gardiner RA, Aitken J, Yaxley J, Clements JA, Spurdle AB, Australian Prostate Cancer B (2011) A replication study examining novel common single nucleotide polymorphisms identified through a prostate cancer genome-wide association study in a Japanese population. Am J Epidemiol 174:1391–1395. doi:10.1093/aje/kwr271
Boyle AP, Hong EL, Hariharan M, Cheng Y, Schaub MA, Kasowski M, Karczewski KJ, Park J, Hitz BC, Weng S, Cherry JM, Snyder M (2012) Annotation of functional variation in personal genomes using RegulomeDB. Genome Res 22:1790–1797. doi:10.1101/gr.137323.112
Bryc K, Auton A, Nelson MR, Oksenberg JR, Hauser SL, Williams S, Froment A, Bodo JM, Wambebe C, Tishkoff SA, Bustamante CD (2010) Genome-wide patterns of population structure and admixture in West Africans and African Americans. Proc Natl Acad Sci USA 107:786–791. doi:10.1073/pnas.0909559107
Campbell MC, Tishkoff SA (2008) African genetic diversity: implications for human demographic history, modern human origins, and complex disease mapping. Annu Rev Genomics Hum Genet 9:403–433
Center MM, Jemal A, Lortet-Tieulent J, Ward E, Ferlay J, Brawley O, Bray F (2012) International variation in prostate cancer incidence and mortality rates. Eur Urol 61:1079–1092. doi:10.1016/j.eururo.2012.02.054
Chang BL, Isaacs SD, Wiley KE, Gillanders EM, Zheng SL, Meyers DA, Walsh PC, Trent JM, Xu J, Isaacs WB (2005) Genome-wide screen for prostate cancer susceptibility genes in men with clinically significant disease. Prostate 64:356–361. doi:10.1002/pros.20249
Chokkalingam AP, Yeboah ED, Demarzo A, Netto G, Yu K, Biritwum RB, Tettey Y, Adjei A, Jadallah S, Li Y, Chu LW, Chia D, Niwa S, Partin A, Thompson IM, Roehrborn C, Hoover RN, Hsing AW (2012) Prevalence of BPH and lower urinary tract symptoms in West Africans. Prostate Cancer Prostatic Dis 15:170–176. doi:10.1038/pcan.2011.43
Chu LW, Ritchey J, Devesa SS, Quraishi SM, Zhang H, Hsing AW (2011) Prostate cancer incidence rates in Africa. Prostate Cancer 2011:947870. doi:10.1155/2011/947870
Chung CC, Chanock SJ (2011) Current status of genome-wide association studies in cancer. Hum Genet 130:59–78. doi:10.1007/s00439-011-1030-9
Clarke L, Zheng-Bradley X, Smith R, Kulesha E, Xiao C, Toneva I, Vaughan B, Preuss D, Leinonen R, Shumway M, Sherry S, Flicek P, Genomes Project C (2012) The 1000 Genomes Project: data management and community access. Nat Methods 9:459–462. doi:10.1038/nmeth.1974
Conrad DF, Jakobsson M, Coop G, Wen X, Wall JD, Rosenberg NA, Pritchard JK (2006) A worldwide survey of haplotype variation and linkage disequilibrium in the human genome. Nat Genet 38:1251–1260. doi:10.1038/ng1911
Donnelly P (2008) Progress and challenges in genome-wide association studies in humans. Nature 456:728–731. doi:10.1038/nature07631
Database of Single Nucleotide Polymorphisms (dbSNP). Bethesda (MD): National Center for Biotechnology Information, National Library of Medicine. (dbSNP Build ID: 137). Available from: http://www.ncbi.nlm.nih.gov/SNP/
ENCODE Project Consortium; Bernstein BE, Birney E, Dunham I, Green ED, Gunter C, Snyder M (2012) An integrated encyclopedia of DNA elements in the human genome. Nature 489(7414):57–74. http://www.nature.com/nature/journal/v489/n7414/abs/nature11247.html#supplementary-information
Engelhardt BE, Stephens M (2010) Analysis of population structure: a unifying framework and novel methods based on sparse factor analysis. PLoS Genet 6. doi:10.1371/journal.pgen.1001117
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM (2010) Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 127:2893–2917. doi:10.1002/ijc.25516
Franks LM (1954) Latent carcinoma of the prostate. J Pathol Bacteriol 68:603–616
Freedman ML, Haiman CA, Patterson N, McDonald GJ, Tandon A, Waliszewska A, Penney K, Steen RG, Ardlie K, John EM, Oakley-Girvan I, Whittemore AS, Cooney KA, Ingles SA, Altshuler D, Henderson BE, Reich D (2006) Admixture mapping identifies 8q24 as a prostate cancer risk locus in African-American men. Proc Natl Acad Sci USA 103:14068–14073. doi:10.1073/pnas.0605832103
Gaj P, Maryan N, Hennig EE, Ledwon JK, Paziewska A, Majewska A, Karczmarski J, Nesteruk M, Wolski J, Antoniewicz AA, Przytulski K, Rutkowski A, Teumer A, Homuth G, Starzynska T, Regula J, Ostrowski J (2012) Pooled sample-based GWAS: a cost-effective alternative for identifying colorectal and prostate cancer risk variants in the Polish population. PLoS ONE 7:e35307. doi:10.1371/journal.pone.0035307
Grisanzio C, Werner L, Takeda D, Awoyemi BC, Pomerantz MM, Yamada H, Sooriakumaran P, Robinson BD, Leung R, Schinzel AC, Mills I, Ross-Adams H, Neal DE, Kido M, Yamamoto T, Petrozziello G, Stack EC, Lis R, Kantoff PW, Loda M, Sartor O, Egawa S, Tewari AK, Hahn WC, Freedman ML (2012) Genetic and functional analyses implicate the NUDT11, HNF1B, and SLC22A3 genes in prostate cancer pathogenesis. Proc Natl Acad Sci USA 109:11252–11257. doi:10.1073/pnas.1200853109
Haas GP, Delongchamps N, Brawley OW, Wang CY, de la Roza G (2008) The worldwide epidemiology of prostate cancer: perspectives from autopsy studies. Can J Urol 15:3866–3871
Haiman CA, Patterson N, Freedman ML, Myers SR, Pike MC, Waliszewska A, Neubauer J, Tandon A, Schirmer C, McDonald GJ, Greenway SC, Stram DO, Le Marchand L, Kolonel LN, Frasco M, Wong D, Pooler LC, Ardlie K, Oakley-Girvan I, Whittemore AS, Cooney KA, John EM, Ingles SA, Altshuler D, Henderson BE, Reich D (2007) Multiple regions within 8q24 independently affect risk for prostate cancer. Nat Genet 39:638–644
Haiman CA, Chen GK, Blot WJ, Strom SS, Berndt SI, Kittles RA, Rybicki BA, Isaacs WB, Ingles SA, Stanford JL, Diver WR, Witte JS, Chanock SJ, Kolb S, Signorello LB, Yamamura Y, Neslund-Dudas C, Thun MJ, Murphy A, Casey G, Sheng X, Wan P, Pooler LC, Monroe KR, Waters KM, Le Marchand L, Kolonel LN, Stram DO, Henderson BE (2011a) Characterizing genetic risk at known prostate cancer susceptibility loci in African Americans. PLoS Genet 7:e1001387. doi:10.1371/journal.pgen.1001387
Haiman CA, Chen GK, Blot WJ, Strom SS, Berndt SI, Kittles RA, Rybicki BA, Isaacs WB, Ingles SA, Stanford JL, Diver WR, Witte JS, Hsing AW, Nemesure B, Rebbeck TR, Cooney KA, Xu J, Kibel AS, Hu JJ, John EM, Gueye SM, Watya S, Signorello LB, Hayes RB, Wang Z, Yeboah E, Tettey Y, Cai Q, Kolb S, Ostrander EA, Zeigler-Johnson C, Yamamura Y, Neslund-Dudas C, Haslag-Minoff J, Wu W, Thomas V, Allen GO, Murphy A, Chang BL, Zheng SL, Leske MC, Wu SY, Ray AM, Hennis AJ, Thun MJ, Carpten J, Casey G, Carter EN, Duarte ER, Xia LY, Sheng X, Wan P, Pooler LC, Cheng I, Monroe KR, Schumacher F, Le Marchand L, Kolonel LN, Chanock SJ, Berg DV, Stram DO, Henderson BE (2011b) Genome-wide association study of prostate cancer in men of African ancestry identifies a susceptibility locus at 17q21. Nat Genet 43:570–573. doi:10.1038/ng.839
Harrow J, Frankish A, Gonzalez JM, Tapanari E, Diekhans M, Kokocinski F, Aken BL, Barrell D, Zadissa A, Searle S, Barnes I, Bignell A, Boychenko V, Hunt T, Kay M, Mukherjee G, Rajan J, Despacio-Reyes G, Saunders G, Steward C, Harte R, Lin M, Howald C, Tanzer A, Derrien T, Chrast J, Walters N, Balasubramanian S, Pei B, Tress M, Rodriguez JM, Ezkurdia I, van Baren J, Brent M, Haussler D, Kellis M, Valencia A, Reymond A, Gerstein M, Guigo R, Hubbard TJ (2012) GENCODE: the reference human genome annotation for The ENCODE Project. Genome Res 22:1760–1774. doi:10.1101/gr.135350.111
Hemminki K, Lorenzo Bermejo J, Forsti A (2006) The balance between heritable and environmental aetiology of human disease. Nat Rev Genet 7:958–965. doi:10.1038/nrg2009
Henn BM, Gravel S, Moreno-Estrada A, Acevedo-Acevedo S, Bustamante CD (2010) Fine-scale population structure and the era of next-generation sequencing. Hum Mol Genet 19:R221–R226. doi:10.1093/hmg/ddq403
Hindorff LA, Sethupathy P, Junkins HA, Ramos EM, Mehta JP, Collins FS, Manolio TA (2009) Potential etiologic and functional implications of genome-wide association loci for human diseases and traits. Proc Natl Acad Sci USA 106:9362–9367. doi:10.1073/pnas.0903103106
Ioannidis JP (2007) Non-replication and inconsistency in the genome-wide association setting. Hum Hered 64:203–213. doi:10.1159/000103512
Jin W, Wang S, Wang H, Jin L, Xu S (2012) Exploring population admixture dynamics via empirical and simulated genome-wide distribution of ancestral chromosomal segments. Am J Hum Genet 91:849–862. doi:10.1016/j.ajhg.2012.09.008
Kraft P, Hunter DJ (2009) Genetic risk prediction—are we there yet? N Engl J Med 360:1701–1703. doi:10.1056/NEJMp0810107
Li J, German R, King J, Joseph D, Thompson T, Wu XC, Ajani U, Tai E (2012) Recent trends in prostate cancer testing and incidence among men under age of 50. Cancer Epidemiol 36:122–127. doi:10.1016/j.canep.2011.10.014
Liu M, Wang J, Xu Y, Wei D, Shi X, Yang Z (2012) Risk loci on chromosome 8q24 are associated with prostate cancer in northern Chinese men. J Urol 187:315–321. doi:10.1016/j.juro.2011.09.011
Ntzani EE, Liberopoulos G, Manolio TA, Ioannidis JPA (2012) Consistency of genome-wide associations across major ancestral groups. Hum Genet 131:1057–1071. doi:10.1007/s00439-011-1124-4
Park JH, Wacholder S, Gail MH, Peters U, Jacobs KB, Chanock SJ, Chatterjee N (2010) Estimation of effect size distribution from genome-wide association studies and implications for future discoveries. Nat Genet 42:570–575. doi:10.1038/ng.610
Parkin DM, Ferlay J, Curado MP, Bray F, Edwards B, Shin HR, Forman D (2010) Fifty years of cancer incidence: CI5 I-IX. Int J Cancer 127:2918–2927. doi:10.1002/ijc.25517
Pemberton TJ, Absher D, Feldman MW, Myers RM, Rosenberg NA, Li JZ (2012) Genomic patterns of homozygosity in worldwide human populations. Am J Hum Genet 91:275–292. doi:10.1016/j.ajhg.2012.06.014
Perez-Losada J, Castellanos-Martin A, Mao JH (2011) Cancer evolution and individual susceptibility. Integr Biol (Camb) 3:316–328. doi:10.1039/c0ib00094a
Rebbeck TR, Devesa SS, Chang BL, Bunker CH, Cheng I, Cooney K, Eeles R, Fernandez P, Giri VN, Gueye SM, Haiman CA, Henderson BE, Heyns CF, Hu JJ, Ingles SA, Isaacs W, Jalloh M, John EM, Kibel AS, Kidd LR, Layne P, Leach RJ, Neslund-Dudas C, Okobia MN, Ostrander EA, Park JY, Patrick AL, Phelan CM, Ragin C, Roberts RA, Rybicki BA, Stanford JL, Strom S, Thompson IM, Witte J, Xu J, Yeboah E, Hsing AW, Zeigler-Johnson CM (2013) Global patterns of prostate cancer incidence, aggressiveness, and mortality in men of African descent. Prostate Cancer 2013:560857. doi:10.1155/2013/560857
Reich D, Price AL, Patterson N (2008) Principal component analysis of genetic data. Nat Genet 40:491–492. doi:10.1038/ng0508-491
Sakr WA, Haas GP, Cassin BF, Pontes JE, Crissman JD (1993) The frequency of carcinoma and intraepithelial neoplasia of the prostate in young male patients. J Urol 150:379–385
Sherry ST, Ward MH, Kholodov M, Baker J, Phan L, Smigielski EM, Sirotkin K (2001) dbSNP: the NCBI database of genetic variation. Nucleic Acids Res 29:308–311
Takata R, Akamatsu S, Kubo M, Takahashi A, Hosono N, Kawaguchi T, Tsunoda T, Inazawa J, Kamatani N, Ogawa O, Fujioka T, Nakamura Y, Nakagawa H (2010) Genome-wide association study identifies five new susceptibility loci for prostate cancer in the Japanese population. Nat Genet 42:751–754. doi:10.1038/ng.635
Tishkoff SA, Reed FA, Friedlaender FR, Ehret C, Ranciaro A, Froment A, Hirbo JB, Awomoyi AA, Bodo JM, Doumbo O, Ibrahim M, Juma AT, Kotze MJ, Lema G, Moore JH, Mortensen H, Nyambo TB, Omar SA, Powell K, Pretorius GS, Smith MW, Thera MA, Wambebe C, Weber JL, Williams SM (2009) The genetic structure and history of Africans and African Americans. Science 324:1035–1044. doi:10.1126/science.1172257
Tolnay SE (2003) The African American “Great Migration” and beyond. Annu Rev Sociol 29:209–232. doi:10.1146/annurev.soc.29.010202.100009
Tomlinson IP, Webb E, Carvajal-Carmona L, Broderick P, Howarth K, Pittman AM, Spain S, Lubbe S, Walther A, Sullivan K, Jaeger E, Fielding S, Rowan A, Vijayakrishnan J, Domingo E, Chandler I, Kemp Z, Qureshi M, Farrington SM, Tenesa A, Prendergast JG, Barnetson RA, Penegar S, Barclay E, Wood W, Martin L, Gorman M, Thomas H, Peto J, Bishop DT, Gray R, Maher ER, Lucassen A, Kerr D, Evans DG, Consortium C, Schafmayer C, Buch S, Volzke H, Hampe J, Schreiber S, John U, Koessler T, Pharoah P, van Wezel T, Morreau H, Wijnen JT, Hopper JL, Southey MC, Giles GG, Severi G, Castellvi-Bel S, Ruiz-Ponte C, Carracedo A, Castells A, Consortium E, Forsti A, Hemminki K, Vodicka P, Naccarati A, Lipton L, Ho JW, Cheng KK, Sham PC, Luk J, Agundez JA, Ladero JM, de la Hoya M, Caldes T, Niittymaki I, Tuupanen S, Karhu A, Aaltonen L, Cazier JB, Campbell H, Dunlop MG, Houlston RS (2008) A genome-wide association study identifies colorectal cancer susceptibility loci on chromosomes 10p14 and 8q23.3. Nat Genet 40:623–630. doi:10.1038/ng.111
Torres JB, Doura MB, Keita SO, Kittles RA (2012) Y chromosome lineages in men of west African descent. PLoS ONE 7:e29687. doi:10.1371/journal.pone.0029687
Vijai J, Kirchhoff T, Gallagher D, Hamel N, Guha S, Darvasi A, Lencz T, Foulkes WD, Offit K, Klein RJ (2011) Genetic architecture of prostate cancer in the Ashkenazi Jewish population. Br J Cancer 105:864–869. doi:10.1038/bjc.2011.307
Wang M, Liu F, Hsing AW, Wang X, Shao Q, Qi J, Ye Y, Wang Z, Chen H, Gao X, Wang G, Chu LW, Ding Q, OuYang J, Gao X, Huang Y, Chen Y, Gao YT, Zhang ZF, Rao J, Shi R, Wu Q, Zhang Y, Jiang H, Zheng J, Hu Y, Guo L, Lin X, Tao S, Jin G, Sun J, Lu D, Zheng SL, Sun Y, Mo Z, Yin C, Zhang Z, Xu J (2012) Replication and cumulative effects of GWAS-identified genetic variations for prostate cancer in Asians: a case-control study in the ChinaPCa consortium. Carcinogenesis 33:356–360. doi:10.1093/carcin/bgr279
Ward LD, Kellis M (2012) HaploReg: a resource for exploring chromatin states, conservation, and regulatory motif alterations within sets of genetically linked variants. Nucleic Acids Res 40:D930–D934. doi:10.1093/nar/gkr917
Welch HG, Black WC (2010) Overdiagnosis in cancer. J Natl Cancer Inst 102:605–613. doi:10.1093/jnci/djq099
Xu Z, Bensen JT, Smith GJ, Mohler JL, Taylor JA (2011) GWAS SNP Replication among African American and European American men in the North Carolina-Louisiana prostate cancer project (PCaP). Prostate 71:881–891. doi:10.1002/pros.21304
Zakharia F, Basu A, Absher D, Assimes TL, Go AS, Hlatky MA, Iribarren C, Knowles JW, Li J, Narasimhan B, Sidney S, Southwick A, Myers RM, Quertermous T, Risch N, Tang H (2009) Characterizing the admixed African ancestry of African Americans. Genome Biol 10. doi: 10.1186/gb-2009-10-12-r141
Acknowledgments
The authors thank Ms. Vicky Okyne for her expert help in coordinating the study; consultants/resident urologists, pathologists, nurses, and interviewers of Korle‐Bu Hospital and University of Ghana Medical School for their assistance with subject enrollment, screening, and clinical examination; the study participants for their contribution toward a better understanding of prostate disease; A. DeMarzo and G. Netto of Johns Hopkins University for pathology review; Ms. Violet Devairakkam, Ms. Norma Kim, and Mr. John Heinrich of Research Triangle Institute (RTI) for their expert study management; Prof. Rosalind A. Eeles and her team for cross-checking published prostate cancer loci specified in Table 3; and members of the African Ancestry Prostate Cancer GWAS Consortium for looking-up our most promising associations from our African GWAS. This research was supported by the Intramural Research Program of the National Cancer Institute, National Institutes of Health. Intramural Program of the National Cancer Institute, National Institutes of Health, Department of Health and Human Services including Contract No. HHSN261200800001E.
Conflict of interest
There are no financial disclosures from any of the authors.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
M. B. Cook, Z. Wang and E. D. Yeboah are co-first authors.
A. W. Hsing and S. J. Chanock are co-last authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cook, M.B., Wang, Z., Yeboah, E.D. et al. A genome-wide association study of prostate cancer in West African men. Hum Genet 133, 509–521 (2014). https://doi.org/10.1007/s00439-013-1387-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-013-1387-z