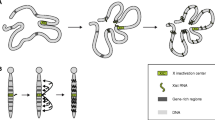
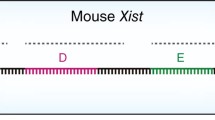
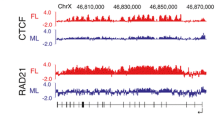
Abstract
X chromosome inactivation (XCI), the silencing of one of the two X chromosomes in XX female cells, equalises the dosage of X-linked genes relative to XY males. The process is mediated by the non-coding RNA X inactive specific transcript (Xist) that binds in cis and propagates along the inactive X chromosome elect, triggering chromosome-wide silencing. The mechanisms by which Xist RNA binds and spreads along the chromosome, and initiates Xist-mediated chromosome silencing remain poorly understood. Accumulating evidence suggests that chromosome and nuclear organisation are important in both processes. Notably, recent studies have identified specific factors, previously shown to be components of the nuclear matrix or scaffold, to play a role both in Xist RNA-binding and in Xist-mediated silencing. In this review we provide a perspective on these studies in the context of previous work on chromosome/nuclear architecture in XCI.



Similar content being viewed by others
References
Agrelo R, Souabni A, Novatchkova M, Haslinger C, Leeb M, Komnenovic V, Kishimoto H, Gresh L, Kohwi-Shigematsu T, Kenner L, Wutz A (2009) SATB1 defines the developmental context for gene silencing by Xist in lymphoma and embryonic cells. Dev Cell 16(4):507–516
Alvarez JD, Yasui DH, Niida H, Joh T, Loh DY, Kohwi-Shigematsu T (2000) The MAR-binding protein SATB1 orchestrates temporal and spatial expression of multiple genes during T-cell development. Genes Dev 14(5):521–535
Bailey JA, Carrel L, Chakravarti A, Eichler EE (2000) Molecular evidence for a relationship between LINE-1 elements and X chromosome inactivation: the Lyon repeat hypothesis. Proc Natl Acad Sci USA 97(12):6634–6639. doi:97/12/6634
Beletskii A, Hong YK, Pehrson J, Egholm M, Strauss WM (2001) PNA interference mapping demonstrates functional domains in the noncoding RNA Xist. Proc Natl Acad Sci U S A 98(16):9215–9220. doi:10.1073/pnas.161173098
Belyaev N, Keohane AM, Turner BM (1996) Differential underacetylation of histones H2A, H3 and H4 on the inactive X chromosome in human female cells. Hum Genet 97(5):573–578
Berezney R, Coffey DS (1974) Identification of a nuclear protein matrix. Biochem Biophys Res Commun 60(4):1410–1417
Bode J, Kohwi Y, Dickinson L, Joh T, Klehr D, Mielke C, Kohwi-Shigematsu T (1992) Biological significance of unwinding capability of nuclear matrix-associating DNAs. Science (New York) 255(5041):195–197
Brockdorff N (2002) X-chromosome inactivation: closing in on proteins that bind Xist RNA. Trends Genet 18(7):352–358. doi:S0168952502027178
Brown CJ, Hendrich BD, Rupert JL, Lafrenière RG, Xing Y, Lawrence J, Willard HF (1992) The human XIST gene: analysis of a 17 kb inactive X-specific RNA that contains conserved repeats and is highly localized within the nucleus. Cell 71(3):527–542
Cai S, Han HJ, Kohwi-Shigematsu T (2003) Tissue-specific nuclear architecture and gene expression regulated by SATB1. Nat Genet 34(1):42–51
Carter D, Chakalova L, Osborne CS, Dai YF, Fraser P (2002) Long-range chromatin regulatory interactions in vivo. Nat Genet 32(4):623–626. doi:10.1038/ng1051
Chaumeil J, Le Baccon P, Wutz A, Heard E (2006) A novel role for Xist RNA in the formation of a repressive nuclear compartment into which genes are recruited when silenced. Genes Dev 20(16):2223–2237
Chow JC, Hall LL, Baldry SE, Thorogood NP, Lawrence JB, Brown CJ (2007) Inducible XIST-dependent X-chromosome inactivation in human somatic cells is reversible. Proc Natl Acad Sci USA 104(24):10104–10109. doi:0610946104
Chow JC, Ciaudo C, Fazzari MJ, Mise N, Servant N, Glass JL, Attreed M, Avner P, Wutz A, Barillot E, Greally JM, Voinnet O, Heard E (2010) LINE-1 activity in facultative heterochromatin formation during X chromosome inactivation. Cell 141(6):956–969
Clemson CM, McNeil JA, Willard HF, Lawrence JB (1996) XIST RNA paints the inactive X chromosome at interphase: evidence for a novel RNA involved in nuclear/chromosome structure. J Cell Biol 132(3):259–275
Clemson CM, Chow JC, Brown CJ, Lawrence JB (1998) Stabilization and localization of Xist RNA are controlled by separate mechanisms and are not sufficient for X inactivation. J Cell Biol 142(1):13–23
Clemson CM, Hall LL, Byron M, McNeil J, Lawrence JB (2006) The X chromosome is organized into a gene-rich outer rim and an internal core containing silenced nongenic sequences. Proc Natl Acad Sci USA 103(20):7688–7693
Costanzi C, Pehrson JR (1998) Histone macroH2A1 is concentrated in the inactive X chromosome of female mammals. Nature 393(6685):599–601
Cremer T, Cremer C (2001) Chromosome territories, nuclear architecture and gene regulation in mammalian cells. Nat Rev Genet 2(4):292–301
Cremer T, Cremer M, Dietzel S, Müller S, Solovei I, Fakan S (2006) Chromosome territories—a functional nuclear landscape. Curr Opin Cell Biol 18(3):307–316
de Belle I, Cai S, Kohwi-Shigematsu T (1998) The genomic sequences bound to special AT-rich sequence-binding protein 1 (SATB1) in vivo in Jurkat T cells are tightly associated with the nuclear matrix at the bases of the chromatin loops. J Cell Biol 141(2):335–348
Dreyfuss G, Choi YD, Adam SA (1984) Characterization of heterogeneous nuclear RNA–protein complexes in vivo with monoclonal antibodies. Mol Cell Biol 4(6):1104–1114
Dreyfuss G, Choi YD, Adam SA (1989) The ribonucleoprotein structures along the pathway of mRNA formation. Endocr Res 15(4):441–474
Dreyfuss G, Matunis MJ, Piñol-Roma S, Burd CG (1993) hnRNP proteins and the biogenesis of mRNA. Annu Rev Biochem 62:289–321
Duthie SM, Nesterova TB, Formstone EJ, Keohane AM, Turner BM, Zakian SM, Brockdorff N (1999) Xist RNA exhibits a banded localization on the inactive X chromosome and is excluded from autosomal material in cis. Hum Mol Genet 8(2):195–204
Eils R, Dietzel S, Bertin E, Schrock E, Speicher MR, Ried T, Robert-Nicoud M, Cremer C, Cremer T (1996) Three-dimensional reconstruction of painted human interphase chromosomes: active and inactive X chromosome territories have similar volumes but differ in shape and surface structure. J Cell Biol 135(6 Pt 1):1427–1440
Fackelmayer FO (2005) A stable proteinaceous structure in the territory of inactive X chromosomes. J Biol Chem 280(3):1720–1723
Fang J, Chen T, Chadwick B, Li E, Zhang Y (2004) Ring1b-mediated H2A ubiquitination associates with inactive X chromosomes and is involved in initiation of X inactivation. J Biol Chem 279(51):52812–52815
Galande S, Dickinson LA, Mian IS, Sikorska M, Kohwi-Shigematsu T (2001) SATB1 cleavage by caspase 6 disrupts PDZ domain-mediated dimerization, causing detachment from chromatin early in T-cell apoptosis. Mol Cell Biol 21(16):5591–5604
Gartler SM, Riggs AD (1983) Mammalian X-chromosome inactivation. Annu Rev Genet 17:155–190. doi:10.1146/annurev.ge.17.120183.001103
Gasser SM, Laemmli UK (1986) The organisation of chromatin loops: characterization of a scaffold attachment site. EMBO J 5(3):511–518
Glazko GV, Koonin EV, Rogozin IB, Shabalina SA (2003) A significant fraction of conserved noncoding DNA in human and mouse consists of predicted matrix attachment regions. Trends Genet TIG 19(3):119–124
Graves JAM (2006) Sex chromosome specialization and degeneration in mammals. Cell 124(5):901–914
Han SP, Tang YH, Smith R (2010) Functional diversity of the hnRNPs: past, present and perspectives. Biochem J 430(3):379–392
Hasegawa Y, Brockdorff N, Kawano S, Tsutui K, Tsutui K, Nakagawa S (2010) The matrix protein hnRNP U is required for chromosomal localization of Xist RNA. Dev Cell 19(3):469–476
Helbig R, Fackelmayer FO (2003) Scaffold attachment factor A (SAF-A) is concentrated in inactive X chromosome territories through its RGG domain. Chromosoma 112(4):173–182
Kiledjian M, Dreyfuss G (1992) Primary structure and binding activity of the hnRNP U protein: binding RNA through RGG box. EMBO J 11(7):2655–2664
Kukalev AS, Lobov IB, Percipalle P, Podgornaya OI (2009) SAF-A/hnRNP-U localization in interphase and metaphase. Cytogenet Genome Res 124(3–4):288–297
Lobov IB, Tsutsui K, Mitchell AR, Podgornaya OI (2000) Specific interaction of mouse major satellite with MAR-binding protein SAF-A. Eur J Cell Biol 79(11):839–849
Lyon MF (1961) Gene action in the X-chromosome of the mouse (Mus musculus L.). Nature 190:372–373
Lyon MF (1998) X-chromosome inactivation: a repeat hypothesis. Cytogenet Cell Genet 80(1–4):133–137
Naughton C, Sproul D, Hamilton C, Gilbert N (2010) Analysis of active and inactive X chromosome architecture reveals the independent organization of 30 nm and large-scale chromatin structures. Mol Cell 40(3):397–409
Navarro P, Avner P (2010) An embryonic story: analysis of the gene regulative network controlling Xist expression in mouse embryonic stem cells. Bioessays 32(7):581–588. doi:10.1002/bies.201000019
Pederson T (2000) Half a century of “the nuclear matrix”. Mol Biol Cell 11(3):799–805
Popova BC, Tada T, Takagi N, Brockdorff N, Nesterova TB (2006) Attenuated spread of X-inactivation in an X; autosome translocation. Proc Natl Acad Sci USA 103(20):7706–7711. doi:0602021103
Pullirsch D, Härtel R, Kishimoto H, Leeb M, Steiner G, Wutz A (2010) The Trithorax group protein Ash2l and Saf-A are recruited to the inactive X chromosome at the onset of stable X inactivation. Development (Cambridge, England) 137(6):935–943
Rego A, Sinclair PB, Tao W, Kireev I, Belmont AS (2008) The facultative heterochromatin of the inactive X chromosome has a distinctive condensed ultrastructure. J Cell Sci 121(Pt 7):1119–1127
Romig H, Fackelmayer FO, Renz A, Ramsperger U, Richter A (1992) Characterization of SAF-A, a novel nuclear DNA binding protein from HeLa cells with high affinity for nuclear matrix/scaffold attachment DNA elements. EMBO J 11(9):3431–3440
Sarma K, Levasseur P, Aristarkhov A, Lee JT (2010) Locked nucleic acids (LNAs) reveal sequence requirements and kinetics of Xist RNA localization to the X chromosome. Proc Natl Acad Sci USA 107(51):22196–22201. doi:1009785107
Savarese F, Flahndorfer K, Jaenisch R, Busslinger M, Wutz A (2006) Hematopoietic precursor cells transiently reestablish permissiveness for X inactivation. Mol Cell Biol 26(19):7167–7177. doi:26/19/7167
Senner CE, Nesterova TB, Norton S, Dewchand H, Godwin J, Mak W, Brockdorff N (2011) Disruption of a conserved region of Xist exon 1 impairs Xist RNA localisation and X-linked gene silencing during random and imprinted X chromosome inactivation. Development 138(8):1541–1550. doi:dev.056812
Smith KP, Byron M, Clemson CM, Lawrence JB (2004) Ubiquitinated proteins including uH2A on the human and mouse inactive X chromosome: enrichment in gene rich bands. Chromosoma 113(6):324–335. doi:10.1007/s00412-004-0325-1
Tang YA, Huntley D, Montana G, Cerase A, Nesterova TB, Brockdorff N (2010) Efficiency of Xist-mediated silencing on autosomes is linked to chromosomal domain organisation. Epigenet Chromatin 3(1):10. doi:1756-8935-3-10
Wang TY, Han ZM, Chai YR, Zhang JH (2010) A mini review of MAR-binding proteins. Mol Biol Rep 37(7):3553–3560
Wutz A, Jaenisch R (2000) A shift from reversible to irreversible X inactivation is triggered during ES cell differentiation. Mol Cell 5(4):695–705
Wutz A, Rasmussen TP, Jaenisch R (2002) Chromosomal silencing and localization are mediated by different domains of Xist RNA. Nat Genet 30(2):167–174
Yamasaki K, Akiba T, Yamasaki T, Harata K (2007) Structural basis for recognition of the matrix attachment region of DNA by transcription factor SATB1. Nucleic Acids Res 35(15):5073–5084
Yasui D, Miyano M, Cai S, Varga-Weisz P, Kohwi-Shigematsu T (2002) SATB1 targets chromatin remodelling to regulate genes over long distances. Nature 419(6907):641–645
Zbarskii IB, Debov SS (1951) Protein fractions in the cell nuclei. Biokhimiia (Moscow, Russia) 16(5):390–395
Acknowledgments
AT was supported by the Stiftung der Deutschen Wirtschaft (SDW) and the Medical Research Council (MRC).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tattermusch, A., Brockdorff, N. A scaffold for X chromosome inactivation. Hum Genet 130, 247–253 (2011). https://doi.org/10.1007/s00439-011-1027-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-011-1027-4