Abstract
Parkinson’s disease (PD), one of the most common human neurodegenerative disorders, is characterized by the loss of dopaminergic neurons in the substantia nigra of the midbrain. Our recent case-control association study of 268 SNPs in 121 candidate genes identified α-synuclein (SNCA) as a susceptibility gene for sporadic PD (P = 1.7 × 10−11). We also replicated the association of fibroblast growth factor 20 (FGF20) with PD (P = 0.0089). To find other susceptibility genes, we added 34 SNPs to the previous screen. Of 302 SNPs in a total 137 genes, but excluding SNCA, SNPs in NDUFV2, FGF2, CALB1 and B2M showed significant association (P < 0.01; 882 cases and 938 control subjects). We replicated the association analysis for these SNPs in a second independent sample set (521 cases and 1,003 control subjects). One SNP, rs1805874 in calbindin 1 (CALB1), showed significance in both analyses (P = 7.1 × 10−5; recessive model). When the analysis was stratified relative to the SNCA genotype, the odds ratio of CALB1 tended to increase according to the number of protective alleles in SNCA. In contrast, FGF20 was significant only in the subgroup of SNCA homozygote of risk allele. CALB1 is a calcium-binding protein that widely is expressed in neurons. A relative sparing of CALB1-positive dopaminergic neurons is observed in PD brains, compared with CALB1-negative neurons. Our genetic analysis suggests that CALB1 is associated with PD independently of SNCA, and that FGF20 is associated with PD synergistically with SNCA.

Similar content being viewed by others
References
Bower JH, Maraganore DM, McDonnell SK, Rocca WA (1999) Incidence and distribution of parkinsonism in Olmsted County, Minnesota, 1976–1990. Neurology 52:1214–1220
Chard PS, Bleakman D, Christakos S et al (1993) Calcium buffering properties of calbindin D28k and parvalbumin in rat sensory neurones. J Physiol 472:341–357
Damier P, Hirsch EC, Agid Y, Graybiel AM (1999) The substantia nigra of the human brain II. Patterns of loss of dopamine-containing neurons in Parkinson’s disease patterns of loss of dopamine-containing neurons in Parkinson’s disease. Brain 122:1437–1448
de Rijk MC, Tzourio C, Breteler MMB et al (1997) Prevalence of parkinsonism and Parkinson’s disease in Europe: the EUROPARKINSON collaborative study. J Neurol Neurosurg Psychiatry 62:10–15
Goedert M (2001) Alpha-synuclein and neurodegenerative diseases. Nat Rev Neurosci 2:492–501
Haga H, Yamada R, Ohnishi Y et al (2002) Gene-based SNP discovery as part of the Japanese Millennium Genome Project: identification of 190562 genetic variations in the human genome. J Hum Genet 47:605–610
Jande SS, Maler L, Lawson DEM (1981) Immunohistochemical mapping of vitamin D-dependent calcium-binding protein in brain. Nature 294:765–767
Maraganore DM, de Andrade M, Lesnick TG et al (2005) High-resolution whole-genome association study of Parkinson disease. Am J Hum Genet 77:685–693
Maraganore DM, de Andrade M, Elbaz A et al (2006) Collaborative analysis of α-synuclein gene promoter variability and Parkinson disease. JAMA 296:661–670
Mattson MP, Rychlik B, Chu C, Christakos S (1991) Evidence for calcium-reducing and excito-protective roles for the calcium-binding protein calbindin-D28 K in cultured hippocampal neurons. Neuron 6:41–51
McMahon A, Wong BS, Iacopino AM et al (1998) Calbindin-D28 K buffers intracellular calcium and promotes resistance to degeneration in PC12 cells. Mol Brain Res 54:56–63
Mizuta I, Satake W, Nakabayashi Y et al (2006) Multiple candidate gene analysis identifies α-synuclein as a susceptibility gene for sporadic Parkinson’s disease. Hum Mol Genet 15:1151–1158
Moore DJ, West AB, Dawson VL, Dawson TM (2005) Molecular pathophysiology of Parkinson’s disease. Annu Rev Neurosci 28:57–87
Mueller JC, Fuchs J, Hofer A et al (2005) Multiple regions of α-synuclein are associated with Parkinson’s disease. Ann Neurol 57:535–541
Myhre R, Toft M, Kachergus J, Hulihan MM, Aasly JO, Klungland H, Farrer MJ (2008) Multiple alpha-synuclein gene polymorphisms are associated with Parkinson’s disease in a Norwegian population. Acta Neurol Scand (in press)
Persechini A, Moncrief ND, Kretsinger RH (1989) The EF-hand family of calcium-modulated proteins. Trends Neurosci 12:462–467
Polymeropoulos MH, Lavedan C, Leroy E et al (1997) Mutation in the α-synuclein gene identified in families with Parkinson’s disease. Science 276:2045–2047
Rascol O, Payoux P, Ory F et al (2003) Limitations of current Parkinson’s disease therapy. Ann Neurol 53(Suppl 3):S3–S12
Satake W, Mizuta I, Suzuki S et al (2007) Fibroblast growth factor 20 gene and Parkinson’s disease in the Japanese population. Neuroreport 18:937–940
Shults CW (2006) Lewy bodies. Proc Natl Acad Sci USA 103:1661–1668
Spillantini MG, Schmidt ML, Lee VM-Y et al (1997) α-Synuclein in Lewy bodies. Nature 388:839–840
The International HapMap Consortium (2005) A haplotype map of the human genome. Nature 437:1299–1320
Thomas B, Beal MF (2007) Parkinson’s disease. Hum Mol Genet 16:R183–R194
Wang G, van der Walt JM, Mayhew G et al (2008) Variation in the miRNA-433 binding site of FGF20 confers risk for Parkinson disease by overexpression of α-synuclein. Am J Hum Genet 82:283–289
Warner TT, Schapira AH (2003) Genetic and environmental factors in the cause of Parkinson’s disease. Ann Neurol 53(Suppl 3):S16–S23
Wasserman RH, Corradino RA, Taylor AN (1968) Vitamin D-dependent calcium-binding protein. Purification and some properties. J Biol Chem 243:3978–3986
Yamada T, McGeer PL, Baimbridge KG, McGeer EG (1990) Relative sparing in Parkinson’s disease of substantia nigra dopamine neurons containing calbindin-D28K. Brain Res 526:303–307
Yenari MA, Minami M, Sun GH et al (2001) Calbindin D28K overexpression protects striatal neurons from transient focal cerebral ischemia. Stroke 32:1028–1035
Zabetian CP, Hutter CM, Factor SA et al (2007) Association analysis of MAPT H1 haplotype and subhaplotypes in Parkinson’s disease. Ann Neurol 62:137–144
Acknowledgments
We are grateful to the PD patients who participated in this study. We also thank Chiyomi Ito, Satoko Suzuki, and Dr. Yoshio Momose for help in performing the study; Drs. Akira Oka, Hidetoshi Inoko, and Katsushi Tokunaga for control samples; and Dr. Jennifer Logan for editing the manuscript. This work was supported by a grant from Core Research for Evolutional Science and Technology (CREST), Japan Science and Technology Agency (JST); by the twenty-first Century COE program and KAKENHI (17019044 and 19590990), both from the Ministry of Education, Culture, Sports, Science, and Technology of Japan; and by the Grant-in-Aid for “the Research Committee for the Neurodegenerative Diseases” of the Research on Measures for Intractable Diseases and Research Grant (H19-Genome-Ippan-001), all from the Ministry of Health, Labor, and Welfare of Japan.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 4 Information of SNPs tagged by rs1805874 and four neighboring SNPs (DOC 68 kb).

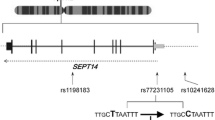
439_2008_525_Fig2_ESM.jpg
Distribution of SNPs tagged by PD-associated SNP rs1805874 and four closely neighboring ones showing no association (rs2074596, rs12549239, rs12171665, and rs12541465). The five tag SNPs are presented by vertical lines. The SNPs tagged by rs1805874 are shown in red. All the SNP IDs are listed in Supplementary Table 4 (JPEG 26 kb).
Rights and permissions
About this article
Cite this article
Mizuta, I., Tsunoda, T., Satake, W. et al. Calbindin 1, fibroblast growth factor 20, and α-synuclein in sporadic Parkinson’s disease. Hum Genet 124, 89–94 (2008). https://doi.org/10.1007/s00439-008-0525-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-008-0525-5