Abstract
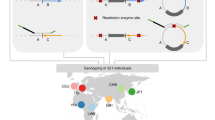
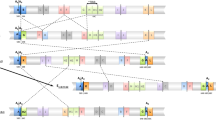
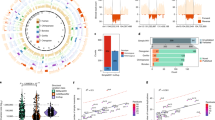
A significant proportion of both pericentric and paracentric inversions have recurrent breakpoints and so could either have arisen through multiple independent events or be identical by descent (IBD) with a single common ancestor. Of two common variant inversions previously studied, the inv(2)(p11q13) was genuinely recurrent while the inv(10)(p11.2q21.2) was IBD in all cases tested. Excluding these two variants we have ascertained 257 autosomal inversion probands at the Wessex Regional Genetics Laboratory. There were 104 apparently recurrent inversions, representing 35 different breakpoint combinations and we speculated that at least some of these had arisen on more than one occasion. However, haplotype analysis identified no recurrent cases among eight inversions tested, including the variant inv(5)(p13q13). The cases not IBD were shown to have different breakpoints at the molecular cytogenetic level. No crossing over was detected within any of the inversions and the founder haplotypes extended for variable distances beyond the inversion breakpoints. Defining breakpoint intervals by FISH mapping identified no obvious predisposing elements in the DNA sequence. In summary the vast majority of human inversions arise as unique events. Even apparently recurrent inversions, with the exception of the inv(2)(p12q13), are likely to be either derived from a common ancestor or to have subtly different breakpoints. Presumably the lack of selection against most inversions allows them to accumulate and disperse amongst different populations over time.

Similar content being viewed by others
References
Allderdice PW, Browne N, Murphy DP (1975) Chromosome 3 duplication q21->qter deletion p25->pter syndrome in children of carriers of a pericentric inversion inv(3)(p25q31). Am J Hum Genet 27:699–718
Baptista J, Mercer C, Prigmore E, Gribble SM, Carter NP, Maloney V, Thomas NS, Jacobs PA, Crolla JA (2008) Breakpoint mapping and array CGH in translocations: comparison of a phenotypically normal and an abnormal cohort. Am J Hum Genet 82:927–936
Beiraghi S, Zhou M, Talmadge CB, Went-Sumegi CB, Davis JR, Huang D, Saal H, Seemayer TA, Sumegi J (2003) Identification and characterisation of a novel gene disrupted by a pericentric inversion inv(4)(p13.1q21.1) in a family with cleft lip. Gene 309:11–21
Bhatt S, Moradkhani K, Mrasek K, Puechberty J, Lefort G, Lespinasse J, Sarda P, Liehr T, Hamamah S, Pellestor F (2007) Breakpoint characterisation: a new approach for segregation analysis of paracentric inversion in human sperm. Hum Reprod 13:751–756
Chodirker BN, Greenberg CR, Pabello PD, Chundley AE (1992) Paracentric inversion 11q in Canadian Hutterites. Hum Genet 89:450–452
Daniel A, Hook EB, Wulf G (1989) Risks of unbalanced progeny at amniocentesis to carriers of chromosome rearrangements: data from United States and Canadian laboratories. Am J Med Genet 431:14–53
Fantes JA, Boland E, Ramsay J, Donnai D, Splitt M, Goodship JA, Stewart H, Whiteford M, Gautier P, Harewood L, Holloway S, Sharkey F, Maher E, van Heyningen V, Clayton-Smith J, Fitzpatrick DR, Black GCM (2008) FISH mapping of de novo apparently balanced chromosome rearrangements identified characteristics associated with phenotypic abnormality. Am J Hum Genet 82:916–926
Fickelscher I, Liehr T, Watts K, Bryant V, Barber JCK, Heidemann S, Siebert R, Hertz JM, Tumer Z, Thomas NS (2007) The variant inv(2)(p11.2q13) is a genuinely recurrent rearrangement but displays some breakpoint heterogeneity. Am J Hum Genet 81:847–856
Gardner RJM, Sutherland GT (1996) Chromosome abnormalities and genetic counselling, 2nd edn. Oxford University Press, New York, pp 115–182
Gilling M, Dullinger JS, Gesk S, Metzke-Heidemann S, Siebert R, Meyer T, Brondum-Nielsen K, Tommerup N, Ropers HH, Tumer Z, Kalscheuer VM, Thomas NS (2006) Breakpoint cloning and haplotype analysis indicate a single origin of the common inv(10)(p11.2q21.2) among Northern Europeans. Am J Hum Genet 78:878–883
Graw SL, Sample T, Bleskan J, Sujansky E, Patterson D (2000) Cloning, sequencing and analysis of Inv8 chromosome breakpoints associated with recombinant 8 syndrome. Am J Hum Genet 66:1138–1144
Hysert M, Bruyere H, Cote GB, Dawson AJ, Dolling J-A, Fetni R, Hrynchak M, Lavoie J, McGowan-Jordan J, Tihy F, Duncan AMV (2006) Prenatal cytogenetic assessment and inv(2)(p11.2q13). Prenat Diagn 26:810–813
Iida A, Emi M, Matsuoka R, Hiratsuka R, Okui K, Ohashi H, Inazawa J, Fukushima Y, Imai T, Nakamura Y (2000) Identification of a gene disrupted by inv(11)(q13.5q25) in a patient with left–right axis malformation. Hum Genet 106:277–287
Jacobs PA, Frackiewicz A, Law P, Hilditch J, Morton NE (1975) The effect of structural aberrations of the chromosomes on reproductive fitness in man. II. Results. Clin Genet 8:169–178
Kleinjan DA, van Heyningen V (2005) Long-range control of gene expression: emerging mechanisms and disruption in disease. Am J Hum Genet 76:8–32
Madan K, Pieters MHEC, Kuyt LP, van Asperen CJ, de Pater JM, Hammers AJH, Gerssen-Schoorl KBJ, Hustinx TWJ, Breed ASPM, Van Hemel JO, Smeets DFCM (1990) Paracentric inversion inv(11)(q21q23) in the Netherlands. Hum Genet 85:15–20
Morel F, Laudier B, Guerif F, Couet ML, Royere D, Roux C, Bresson JL, Amice V, De Braekeleer M, Douet-Guilbert N (2007) Meiotic segregation analysis in spermatozoa of pericentric inversion carriers using fluorescence in-situ hybridization. Hum Reprod 22:136–141
Pettenati MJ, Rao PN, Phelan MC, Grass F, Rao KW, Cosper P, Carroll AJ, Elder F, Smith JL, Higgins MD, Lanman JT, Higgins RR, Butler MG, Luthardt F, Keitges E, Jackson-Cook C, Brown J, Schwartz S, Van Dyke DL, Palmer GG (1995) Paracentric inversions in Humans: a review of 446 paracentric inversions with presentation of 120 new cases. Am J Med Genet 55:171–187
Saito-Ohara H, Fukuda Y, Ito M, Agarwala KL, Hayashi M, Matsuo M, Imoto I, Yamakawa K, Nakamura Y, Inazawa J (2002) The Xq22 inversion breakpoint interrupted a novel Ras-Like GTPase gene in a patient with Duchenne muscular dystrophy and profound mental retardation. Am J Hum Genet 71:637–645
Sakharkar MK, Chow VT, Kangueane P (2004) Distribution of exons and introns in the human genome. In Silico Biol 4:387–393
Shaw CJ, Lupski JR (2004) Implications of human genome architecture for rearrangement-based disorders: the genomic basis of disease. Hum Mol Genet 13:R57–R64
Sherman SL, Iselius L, Gallano P, Buckton K, Collyer S, De Mey R, Kristoffersson U, Lindsten J, Mikkelsen M, Morton NE, Newton M, Nordensson I, Petersen MB, Wahlstrom J (1986) Segregation analysis of balanced pericentric inversions in pedigree data. Clin Genet 30:87–94
Smith AC, Spuhler K, Williams TM, McConnell T, Sujansky E, Robinson A (1987) Genetic risk for recombinant 8 syndrome and the transmission rate of balanced inversion 8 in the Hispanic population of the south western United States. Am J Hum Genet 41:1083–1103
Sood R, Bader PI, Speer MC, Edwards YH, Eddings EM, Blair RT, Hu P, Faruque MU, Robbins CM, Zhang H, Leuders J, Morrison K, Thompson D, Schartzberg PL, Meltzer PS, Trent JM (2004) Cloning and characterisation of an inversion breakpoint at 6q23.3 suggests a role for Map7 in sacral dysgenesis. Cytogenet Genome Res 106:61–67
Tadin-Strapps M, Warburton D, Baumeister FA, Fischer SG, Yonan J, Gilliam TC, Christiano AM (2004) Cloning of the breakpoints of a de novo inversion of chromosome 8, inv(8)(p11.2q23.1) in a patient with Ambras syndrome. Genome Res 107:68–76
Youings S, Ellis K, Ennis S, Barber J, Jacobs P (2004) A study of reciprocal translocations and inversions detected by light microscopy with special reference to origin, segregation and recurrent abnormalities. Am J Med Genet 126A:46–60
Warburton D (1991) De novo balanced chromosome rearrangements and extra marker chromosomes identified at prenatal diagnosis: clinical significance and distribution of breakpoints. Am J Hum Genet 49:995–1013
Acknowledgments
We are very grateful to Mrs. Barbara O’Prey for obtaining patient samples and Dr. Katherine Lachlan for supplying clinical information for phenotype–genotype correlations. Dr. Claire Scott (claire.scott@orh.nhs.uk) kindly provided inversion data from the Chromosome Abnormality Database (funded by BDF Newlife) and additional DNA samples were obtained from Dr. Leema Robert and Dr. Caroline MacKie-Ogilvie.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Thomas, N.S., Bryant, V., Maloney, V. et al. Investigation of the origins of human autosomal inversions. Hum Genet 123, 607–616 (2008). https://doi.org/10.1007/s00439-008-0510-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-008-0510-z