Abstract
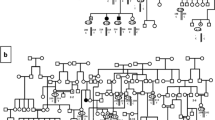
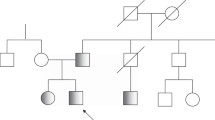
Familial cases of congenital hypothyroidism from thyroid dysgenesis (TD) (OMIM 218700) occur with a frequency 15-fold higher than by chance, FOXE1 is one of the candidate genes for this genetic predisposition and contains an alanine tract. Our purpose is to assess the influence of length of the alanine tract of FOXE1 on genetic susceptibility to TD. A case–control association study (based on 115 patients affected by TD and 129 controls genotyped by direct sequencing) and transmission disequilibrium testing (TDT) analyses were performed. The transcriptional activities of FOXE1 constructs containing 14 or 16 alanines were also studied. In the case–control association study, the 16/16 and 16/14 genotypes were inversely associated with TD (OR = 0.39, 95%CI = 0.22–0.68, P = 0.0005), strongly suggesting that the presence of 16 alanines in the tract protect against the occurrence of TD. This association was stronger in the subgroup of patients with ectopic thyroid (OR = 0.28, 95%CI = 0.13–0.58, P = 0.00015). The protection was confirmed by the TDT analysis performed in 39 trios (χ2 = 4.3, P = 0.0374). Alternatively, the presence of the 14/14 genotype is associated with an increase risk of TD (OR = 2.59, 95%CI = 1.56–4.62, P = 0.0005). The expression studies showed that the transcriptional activities of FOXE1 with 16 alanines were significantly higher (1.55-fold) than FOXE1 containing 14 alanines (P < 0.003), while the nuclear localisation of the proteins was not affected. We conclude that FOXE1 through its alanine containing stretch modulates significantly the risk of TD occurrence, enhancing a mechanism linking an alanine containing transcription factor to disease.



Similar content being viewed by others
References
Abu-Baker A, Rouleau GA (2007) Oculopharyngeal muscular dystrophy: recent advances in the understanding of the molecular pathogenic mechanisms and treatment strategies. Biochim Biophys Acta 1772(2):173–185
Albrecht A, Mundlos S (2005) The other trinucleotide repeat: polyalanine expansion disorders. Curr Opin Genet Dev 15:285–293
Amendola E, De Luca P, Macchia PE, Terracciano D, Rosica A, Chiappetta G, Kimura S, Mansouri A, Affuso A, Arra C, Macchia V, Di Lauro R, De Felice M (2005) A mouse model demonstrates a multigenic origin of congenital hypothyroidism. Endocrinology 146:5038–5044
Amiel J, Laudier B, Attie-Bitach T, Trang H, de Pontual L, Gener B, Trochet D, Etchevers H, Ray P, Simonneau M, Vekemans M, Munnich A, Gaultier C, Lyonnet S (2003) Polyalanine expansion and frameshift mutations of the paired-like homeobox gene PHOX2B in congenital central hypoventilation syndrome. Nat Genet 33:459–461
Baris I, Arisoy AE, Smith A, Agostini M, Mitchell CS, Park SM, Halefoglu AM, Zengin E, Chatterjee VK, Battaloglu E (2006) A novel missense mutation in human TTF-2 (FKHL15) gene associated with congenital hypothyroidism but not athyreosis. J Clin Endocrinol Metab 91(10):4183–4187
Brown LY, Brown SA (2004) Alanine tracts: the expanding story of human illness and trinucleotide repeats. Trends Genet 20(1):51–58
Caburet S, Demarez A, Moumne L, Fellous M, De Baere E, Veitia RA (2004) A recurrent polyalanine expansion in the transcription factor FOXL2 induces extensive nuclear and cytoplasmic protein aggregation. J Med Genet 41(12):932–936
Castanet M, Lyonnet S, Bonaiti-Pellie C, Polak M, Czernichow P, Leger J (2000) Familial forms of thyroid dysgenesis among infants with congenital hypothyroidism. N Engl J Med 343:441–442
Castanet M, Polak M, Bonaiti-Pellie C, Lyonnet S, Czernichow P Leger J (2001) Nineteen years of national screening for congenital hypothyroidism: familial cases with thyroid dysgenesis suggest the involvement of genetic factors. J Clin Endocrinol Metab 86:2009–2014
Castanet M, Park SM, Smith A, Bost M, Leger J, Lyonnet S, Pelet A, Czernichow P, Chatterjee K Polak M (2002) A novel loss-of-function mutation in TTF-2 is associated with congenital hypothyroidism, thyroid agenesis and cleft palat. Hum Mol Genet 11:2051–2059
Chadwick BP, Obermayr F, Frischauf AM (1997) FKHL15, a new human member of the forkhead gene family located on chromosome 9q22. Genomics 41:390–396
Civitareale D, Saiardi A, Falasca P (1994) Purification and characterisation of thyroid transcription factor 2. Biochem J 304:981–985
Clifton-Bligh RJ, Wentworth JM, Heinz P, Crisp MS, John R, Lazarus JH, Ludgate M, Chatterjee VK (1998) Mutation of the gene encoding human TTF-2 associated with thyroid agenesis, cleft palate and choanal atresia. Nat Genet 19:399–401
Collingwood TN, Adams M, Tone Y, Chatterjee VK (1994) Spectrum of transcriptional, dimerization, and dominant negative properties of twenty different mutant thyroid hormone beta-receptors in thyroid hormone resistance syndrome. Mol Endocrinol 8:1262–1277
De Felice M, Ovitt C, Biffali E, Rodriguez-Mallon A, Arra C, Anastassiadis K, Macchia PE, Mattei MG, Mariano A, Scholer H, Macchia V, Di Lauro R (1998) A mouse model for hereditary thyroid dysgenesis and cleft palate. Nat Genet 19:395–398
De Felice M, Di Lauro R (2004) Thyroid development and its disorders: genetics and molecular mechanisms. Endocr Rev 25:722–746
De Roux N, Misrahi M, Brauner R, Houang M, Carel JC, Granier M, Le Bouc Y, Ghinea N, Boumedienne A, Toublanc JE, Milgrom E (1996) Four families with loss of function mutations of the thyrotropin receptor. J Clin Endocrinol Metab 81(12):4229–4235
Dentice M, Cordeddu V, Rosica A, Ferrara AM, Santarpia L, Salvatore D, Chiovato L, Perri A, Moschini L, Fazzini C, Olivieri A, Costa P, Stoppioni V, Baserga M, De Felice M, Sorcini M, Fenzi G, Di Lauro R, Tartaglia M, Macchia PE (2006) Missense mutation in the transcription factor NKX2-5: a novel molecular event in the pathogenesis of thyroid dysgenesis. J Clin Endocrinol Metab 91(4):1428–1433
Devriendt K, Vanhole C, Matthijs G, de Zegher F (1998) Deletion of thyroid transcription factor-1 gene in an infant with neonatal thyroid dysfunction and respiratory failure. N Engl J Med 338(18):1317–1318
Di Palma T, Nitsch R, Mascia A, Nitsch L, Di Lauro R, Zannini M, (2003) The paired domain-containing factor Pax8 and the homeodomain-containing factor TTF-1 directly interact and synergistically activate transcription. J Biol Chem 278(5):3395–3402
Eugene D, Djemli A, Van Vliet G (2005) Sexual dimorphism of thyroid function in newborns with congenital hypothyroidism. J Clin Endocrinol Metab 90:2696–2700
Freimer NB, Sabatti C (2007) Human genetics: variants in common diseases. Nature 445(7130):828–830
Gagne N, Parma J, Deal C, Vassart G, Van Vliet G (1998) Apparent congenital athyreosis contrasting with normal plasma thyroglobulin levels and associated with inactivating mutations in the thyrotropin receptor gene: are athyreosis and ectopic thyroid distinct entities? J Clin Endocrinol Metab 83:1771–1775
Hishinuma A, Ohyama Y, Kuribayashi T, Nagakubo N, Namatame T, Shibayama K, Arisaka O, Matsuura N, Ieiri T (2001) Polymorphism of the polyalanine tract of thyroid transcription factor-2 gene in patients with thyroid dysgenesis. Eur J Endocrinol 145:385–389
Lavoie H, Debeane F, Trinh QD, Turcotte JF, Corbeil-Girard LP, Dicaire MJ, Saint-Denis A, Page M, Rouleau GA, Brais B (2003) Polymorphism, shared functions and convergent evolution of genes with sequences coding for polyalanine domains. Hum Mol Genet 12:2967–2979
Macchia PE, Mattei MG, Lapi P, Fenzi G, Di Lauro R (1999) Cloning, chromosomal localization and identification of polymorphisms in the human thyroid transcription factor 2 gene (TITF2). Biochimie 81(5):433–440
Malik S, Abel L, Tooker H, Poon A, Simkin L, Girard M, Adams GJ, Starke JR, Smith KC, Graviss EA, Musser JM, Schurr E (2005) Alleles of the NRAMP1 gene are risk factors for pediatric tuberculosis disease. Proc Natl Acad Sci U S A 102:12183–12188
Mansouri A, Chowdhury K, Gruss P (1998) Follicular cells of the thyroid gland require Pax8 gene function. Nat Genet 19(1):87–90
Parlato R, Rosica A, Rodriguez-Mallon A, Affuso A, Postiglione MP, Arra C, Mansouri A, Kimura S, Di Lauro R, De Felice M (2004) An integrated regulatory network controlling survival and migration in thyroid organogenesis. Dev Biol 276(2):464–475
Perrone L, Pasca di Magliano M, Zannini M, Di Lauro R, (2000) The thyroid transcription factor 2 (TTF-2) is a promoter-specific DNA-binding independent transcriptional repressor. Biochem Biophys Res Commun 275(1):203–208
Perry R, Heinrichs C, Bourdoux P, Khoury K, Szots F, Dussault JH, Vassart G, Van Vliet G (2002) Discordance of monozygotic twins for thyroid dysgenesis: implications for screening and for molecular pathophysiology. J Clin Endocrinol Metab 87:4072–4077
Pohlenz J, Dumitrescu A, Zundel D, Martine U, Schonberger W, Koo E, Weiss RE, Cohen RN, Kimura S, Refetoff S (2002) Partial deficiency of thyroid transcription factor 1 produces predominantly neurological defects in humans and mice. J Clin Invest 109:469–473
Polak M, Sura-Trueba S, Chauty A, Szinnai G, Carre A, Castanet M (2004) Molecular mechanisms of thyroid dysgenesis. Horm Res 62(Suppl 13):14–21
Santarpia L, Valenzise M, Di Pasquale G, Arrigo T, San Martino G, Ciccio MP, Trimarchi F, De Luca F, Benvenga S (2007) TTF-2/FOXE1 gene polymorphisms in Sicilian patients with permanent primary congenital hypothyroidism. J Endocrinol Invest 30(1):13–19
Shoubridge C, Cloosterman D, Parkinson-Lawerence E, Brooks D, Gecz J (2007) Molecular pathology of expanded polyalanine tract mutations in the Aristaless-related homeobox gene. Genomics 90(1):59–71
Sladek R, Rocheleau G, Rung J, Dina C, Shen L, Serre D, Boutin P, Vincent D, Belisle A, Hadjadj S, Balkau B, Heude B, Charpentier G, Hudson TJ, Montpetit A, Pshezhetsky AV, Prentki M, Posner BI, Balding DJ, Meyre D, Polychronakos C, Froguel P (2007) A genome-wide association study identifies novel risk loci for type 2 diabetes. Nature 445(7130):881–885
Spielman RS, McGinnis RE, Ewens WJ (1993) Transmission test for linkage disequilibrium: the insulin gene region and insulin-dependent diabetes mellitus (IDDM). Am J Hum Genet 52:506–516
Sura-Trueba S, Auge J, Mattei G, Etchevers H, Martinovic J, Czernichow P, Vekemans M, Polak M, Attie-Bitach T (2005) PAX8, TITF1, and FOXE1 gene expression patterns during human development: new insights into human thyroid development and thyroid dysgenesis-associated malformations. J Clin Endocrinol Metab 90:455–462
Tonacchera M, Banco M, Lapi P, Di Cosmo C, Perri A, Montanelli L, Moschini L, Gatti G, Gandini D, Massei A, Agretti P, De Marco G, Vitti P, Chiovato L, Pinchera A (2004) Genetic analysis of TTF-2 gene in children with congenital hypothyroidism and cleft palate, congenital hypothyroidism, or isolated cleft palate. Thyroid 14:584–588
Trochet D, Hong SJ, Lim JK, Brunet JF, Munnich A, Kim KS, Lyonnet S, Goridis C, Amiel J (2005) Molecular consequences of PHOX2B missense, frameshift and alanine expansion mutations leading to autonomic dysfunction. Hum Mol Genet 14:3697–3708
Van Vliet G (2005) The thyroid: a fundamental and clinical text. Lippincott Williams & Wilkins, New York, pp 1029–1047
Vilain C, Rydlewski C, Duprez L, Heinrichs C, Abramowicz M, Malvaux P, Renneboog B, Parma J, Costagliola S, Vassart G (2001) Autosomal dominant transmission of congenital thyroid hypoplasia due to loss-of-function mutation of PAX8. J Clin Endocrinol Metab 86:234–238
Watkins WJ, Harris SE, Craven MJ, Vincent AL, Winship IM, Gersak K, Shelling AN (2006) An investigation into FOXE1 polyalanine tract length in premature ovarian failure. Mol Hum Reprod 12(3):145–149
Acknowledgments
The authors would like to thank especially Josue Feingold M.D. for his help in statistics and interpretation of the data. We acknowledge Philippe Froguel and Martine Vaxillaire for the gift of DNA from control individuals. We thank Stanislas Lyonnet, Tania Attie-Bittach and Claude Ferec for helpful discussions and Arnold Munnich for his support throughout this study. We thank Krish Chatterjee, Samuel Refetoff, Stanislas Lyonnet and Gilbert Vassart for plasmids and antibody gifts. Mireille Castanet1, Aurore Carré2, Sylvia Sura-Trueba3 and Gabor Szinnai4 were supported by grants from Fondation de la Recherche Médicale1, Convention Industrielle de Formation par la Recherche in collaboration with HRA Pharma directed by Dr. André Ulmann and the Ministère de l’Education Nationale de la Recherche et de la Technologie2.3, Electricité de France (RB 200605) and Margarete und Walter Lichtenstein-Stiftung4 (Basel, Switzerland), respectively. We thank the AFDPHE (Association française pour le dépistage et la prévention des handicaps de l’enfant) for indefatigably supporting our work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Aurore Carré and Mireille Castanet contributed equally and should be considered as first joint co-authors.
Rights and permissions
About this article
Cite this article
Carré, A., Castanet, M., Sura-Trueba, S. et al. Polymorphic length of FOXE1 alanine stretch: evidence for genetic susceptibility to thyroid dysgenesis. Hum Genet 122, 467–476 (2007). https://doi.org/10.1007/s00439-007-0420-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-007-0420-5