Abstract
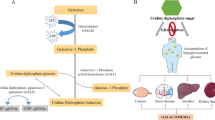
Fabry disease comprises classic and variant phenotypes. The former needs early enzyme replacement therapy, and galactose infusion is effective for some variant cases. Attempts of early diagnosis before manifestations appear will begin in the near future. However, it is difficult to predict the phenotype, to determine the therapeutic approach, only from genetic information. Thus we attempted structural analysis from a novel viewpoint. We built structural models of mutant α-galactosidases resulting from 161 missense mutations (147 classic and 14 variant), and evaluated the influence of each replacement on the structure by calculating the numbers of atoms affected. Among them, 11 mutants, biochemically characterized, were further investigated by color imaging of the influenced atoms. In the variant group, the number of atoms influenced by amino-acid replacement was small, especially in the main chain. In 85% of the cases, less than three atoms in the main chain are influenced. In this group, small structural changes, located apart from the active site, result in destabilization of the mutant enzymes, but galactose can stabilize them. Structural changes caused by classic Fabry mutations are generally large or are located in functionally important regions. In 82% of the cases, three atoms or more in the main chain are affected. The classic group comprises dysfunctional and unstable types, and galactose is not expected to stabilize the mutant enzymes. This study demonstrated the correlation of structural changes, and clinical and biochemical phenotypes. Structural investigation is useful for elucidating the bases of Fabry disease and clinical treatment.



Similar content being viewed by others
References
Altarescu GM, Goldfarb LG, Park KY, Kaneski C, Jeffries N, Litvak S, Nagle JW, Schiffmann R (2001) Identification of 15 novel mutations and genotype-phenotype relationship in Fabry disease. Clin Genet 60:46–51
Desnick RJ, Ioannou YA, Eng CM (2001) α-Galactosidase A deficiency: Fabry disease. In: Scriver CR, Beaudet AL, Sly WS, Valle D (eds) The metabolic and molecular bases of inherited disease, 8th edn. McGraw-Hill, New York, pp 3733–3774
Elleder M, Bradova V, Smid F, Budesinsky M, Harzer K, Kustermann-Kuhn B, Ledvinova J, Belohlavek, Kral V, Dorazilova V (1990) Cardiocyte storage and hypertrophy as a sole manifestation of Fabry’s disease. Report on a case simulating hypertrophic non-obstructive cardiomyopathy. Virchows Arch A Pathol Anat Histopathol 417:449–455
Eng CM, Guffon N, Wilcox WR, Germain DP, Lee P, Waldek S, Caplan L, Linthorst GE, Desnick RJ (2001) Safety and efficacy of recombinant human α-galactosidase A replacement therapy in Fabry’s disease. N Engl J Med 345:9–16
Frustaci A, Chimenti C, Ricci R, Natale L, Russo MA, Pieroni M, Eng CM, Desnick RJ (2001) Improvement in cardiac function in the cardiac variant of Fabry’s disease with galactose-infusion therapy. N Engl J Med 345:25–32
Fukushima M, Tsuchiyama Y, Nakato T, Yokoi T, Ikeda H, Yoshida S, Kusumoto T, Itoh K, Sakuraba H (1995) A female heterozygous patient with Fabry’s disease with renal accumulation of trihexosylceramide detected with a monoclonal antibody. Am J Kidney Dis 26:952–955
Garman SC, Garboczi DN (2004) The molecular defect leading to Fabry disease: structure of human α-galactosidase. J Mol Biol 337:319–335
Germain DP, Poenaru L (1999) Fabry disease: identification of novel α-galactosidase A mutations and molecular carrier detection by use of fluorescent chemical cleavage mismatches. Biochem Biophys Res Commun 257:708–713
Ishii S, Sakuraba H, Suzuki Y (1992) Point mutations in the upstream region of the α-galactosidase A exon 6 in an atypical variant of Fabry disease. Hum Genet 89:29–32
Ishii S, Kase R, Sakuraba H, Suzuki Y (1993) Characterization of a mutant α-galactosidase gene product for the late-onset cardiac form of Fabry disease. Biochem Biophys Res Commun 197:1585–1589
Itoh K, Kotani M, Tai T, Suzuki H, Utsunomiya T, Inoue H, Yamada H, Sakuraba H, Suzuki Y (1993) Immunofluorescence imaging diagnosis of Fabry heterozygotes using confocal laser scanning microscopy. Clin Genet 44:302–306
Itoh K, Takenaka T, Nakao S, Setoguchi M, Tanaka H, Suzuki T, Sakuraba H (1996) Immunofluorescence analysis of trihexosylceramide accumulated in the hearts of variant hemizygotes and heterozygotes with Fabry disease. Am J Cardiol 78:116–117
Kabsh W (1976) A solution for the best rotation to relate two sets of vectors. Acta Crystallogr A 32:827–828
Kabsh W (1978) A discussion of the solution for the best rotation to relate two sets of vectors. Acta Crystallogr A 34:922–923
Kase R, Bierfreund U, Klein A, Kolter T, Utsumi K, Itoh K, Sandhoff K, Sakuraba H (2000) Characterization of two α-galactosidase mutants (Q279E and R301Q) found in an atypical variant of Fabry disease. Biochim Biophys Acta 1501:227–235
Kobori M (2000) Three-dimensional structure of human α-galactosidase, and structural and biological studies on Fabry disease (in Japanese). Doctoral thesis, Faculty of Pharmaceutical Sciences, Graduate School of the University of Tokyo
Leinekugel P, Michel S, Conzelmann E, Sandhoff K (1992) Quantitative correlation between the residual activity of β-hexosaminidase A and arylsulfatase A and severity of the resulting lysosomal storage disease. Hum Genet 88:513–523
Matsuzawa F, Aikawa S, Sakuraba H, Lan HT, Tanaka A, Ohno K, Sugimoto Y, Ninomiya H, Doi H (2003) Structural basis of the GM2 gangliosidosis B variant. J Hum Genet 48:582–589
Mehta A, Ricci R, Widmer U, Dehout F, Garcia de Lorenzo A, Kampmann C, Linhart A, Sunder-Plassmann G, Ries M, Beck M (2004) Fabry disease defined: baseline clinical manifestations of 366 patients in the Fabry Outcome Survey. Eur J Clin Invest 34:236–242
Nakao S, Takenaka T, Maeda M, Kodama C, Tanaka A, Tahara M, Yoshida A, Kuriyama M, Hayashibe H, Sakuraba H, Tanaka H (1995) An atypical variant of Fabry’s disease in men with left ventricular hypertrophy. N Engl J Med 333:288–293
Okumiya T, Ishii S, Kase R, Kamei S, Sakuraba H, Suzuki Y (1995a) α-Galactosidase gene mutations in Fabry disease: heterogeneous expressions of mutant enzyme proteins. Hum Genet 95:557–561
Okumiya T, Ishii S, Takenaka T, Kase R, Kamei S, Sakuraba H, Suzuki Y (1995b) Galactose stabilizes various missense mutants of α-galactosidase in Fabry disease. Biochem Biophys Res Commun 214:1219–1224
Okumiya T, Kawamura O, Itoh K, Kase R, Ishii S, Kamei S, Sakuraba H (1998) Novel missense mutation (M72V) of α-galactosidase gene and its expression product in an atypical Fabry hemizygote. Hum Mut (Suppl 1):S213–S216
Sakuraba H, Yanagawa Y, Igarashi T, Suzuki Y, Suzuki T, Watanabe K, Ieki K, Shimoda K, Yamanaka T (1986) Cardiovascular manifestations in Fabry’s disease. A high incidence of mitral valve prolapse in hemizygotes and heterozygotes. Clin Genet 29:276–283
Sakuraba H, Oshima A, Fukuhara Y, Shimmoto M, Nagao Y, Bishop DF, Desnick RJ, Suzuki Y (1990) Identification of point mutations in the α-galactosidase A gene in classical and atypical hemizygotes with Fabry disease. Am J Hum Genet 47:784–789
Sakuraba H, Matsuzawa F, Aikawa S, Doi H, Kotani M, Lin H, Ohno K, Tanaka A, Yamada H, Uyama E (2000) Molecular and structural studies of the GM2 gangliosidosis 0 variant. J Hum Genet 47:176–183
Sakuraba H, Matsuzawa F, Aikawa S, Doi H, Kotani M, Nakada H, Fukushige T, Kanzaki T (2004) Structural and immunocytochemical studies on α-N-acetylgalactosaminidase deficiency (Schindler/Kanzaki disease). J Hum Genet 49:1–8
Sawada K, Mizoguchi K, Hishida A, Kaneko E, Koide Y, Nishimura K, Kimura M (1996) Point mutation in the α-galactosidase A gene of atypical Fabry disease with only nephropathy. Clin Nephrol 45:289–294
von Scheidt W, Eng CM, Fitzmaurice TF, Erdmann E, Hubner G, Olsen EG, Christomanou H, Kandolf R, Bishop DF, Desnick RJ (1991) An atypical variant of Fabry’s disease with manifestations confined to the myocardium. N Engl J Med 324:395–399
Schiffmann R, Murray GJ, Treco D, Daniel P, Sellos-Moura M, Myers M, Quirk JM, Zirzow GC, Borowski M, Loveday K, Anderson T, Gillespie F, Oliver KL, Jeffries NO, Doo E, Liang TJ, Kreps C, Gunter K, Frei K, Crutchfield K, Selden RF, Brady RO (2000) Infusion of α-galactosidase A reduces tissue globotriaosylceramide storage in patients with Fabry disease. Proc Natl Acad Sci USA 97:365–370
Thurberg BL, Rennke H, Colvin RB, Dikman S, Gordon RE, Collins AB, Desnick RJ, O‘Callaghan M (2002) Globotriaosylceramide accumulation in the Fabry kidney is cleared from multiple cell types after enzyme replacement therapy. Kidney Int 62:1933–1946
Utsumi K, Yamamoto N, Kase R, Takata T, Okumiya T, Saito H, Suzuki T, Uyama E, Sakuraba H (1997) High incidence of thrombosis in Fabry’s disease. Int Med 36:3227–3229
Yanagawa Y, Sakuraba H (1988) Cardiovascular manifestations in Fabry’s disease—age-related changes in hemizygotes and heterozygotes. Acta Paediatr Jpn 30:38–48
Acknowledgements
We wish to thank Prof. K. Itoh (Department of Medical Biotechnology, Graduate School of Pharmaceutical Sciences, The University of Tokushima), and Drs. M. Kotani and Y. Tajima (Department of Clinical Genetics, The Tokyo Metropolitan Institute of Medical Science) for their valuable comments. This work was partly supported by grants from the Tokyo Metropolitan Government, the Japan Society for the Promotion of Science, CREST, the Japan Science and Technology Agency, the Ministry of Education, Science, Sports and Culture of Japan, and the Ministry of Health, Labor and Welfare of Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Matsuzawa, F., Aikawa, Si., Doi, H. et al. Fabry disease: correlation between structural changes in α-galactosidase, and clinical and biochemical phenotypes. Hum Genet 117, 317–328 (2005). https://doi.org/10.1007/s00439-005-1300-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-005-1300-5