Abstract
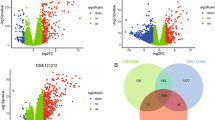
Psoriasis is a common chronic autoimmune inflammatory skin disease that involves genetic and environmental factors. To date, psoriasis is still incurable. Thus, detection of its underlying molecular mechanisms is urgent. Weighted gene co-expression network analysis (WGCNA) was performed on the basis of the RNA-Seq data of psoriatic and normal (NN) skin tissues to detect the key mRNAs and long non-coding RNAs (LncRNAs) implicated in psoriasis and to identify psoriasis-related gene modules. Subsequently, 23 independent modules were obtained, and the pink module that contained differentially expressed 212 mRNAs and 100 LncRNAs was the most remarkable. Differentially expressed genes (DEGs) between psoriasis and healthy control in other RNA-Seq and microarray datasets were integrated to identify convinced psoriasis-associated genes. A total of 312 genes in the pink module and 613 DEGs were scanned. Eleven overlapped key mRNAs were identified, including two known genes (e.g., KRT15 and CCL27) and nine novel ones (e.g., ARSF, CLDN1, DACH1, LONRF1, PAMR1, RORC, SLC26A2, STS, UNC93A). A total of 11 key mRNAs were selected to construct a co-expression network to investigate potential candidate LncRNAs. Seventy-six pairs of LncRNA–mRNA co-expression relationships were found. To validate the findings, CCL27 and LncRNA-AL162231.4 expressions were detected in psoriatic and NN skin tissues. Result of RT-qPCR showed that CCL27 and LncRNA-AL162231.4 decreased in psoriatic lesions with statistical significance (P ≤ 0.05). Our study provides a new direction for elucidating the pathogenesis of psoriasis, but further experiments are still required.




Similar content being viewed by others
References
Ahn R, Gupta R, Lai K, Chopra N, Arron ST, Liao W (2016) Network analysis of psoriasis reveals biological pathways and roles for coding and long non-coding RNAs. BMC Genomics 17:841
Antonini D, Mollo MR, Missero C (2017) Research techniques made simple: identification and characterization of long noncoding RNA in dermatological research. J Investig Dermatol 137:e21–e26
Chenguang G, Maximilian Wei-Lin P, Maquat LE (2012) Biochemical analysis of long non-coding RNA-containing ribonucleoprotein complexes. Methods 58:88–93
Diboun I, Wernisch L, Orengo CA, Koltzenburg M (2006) Microarray analysis after RNA amplification can detect pronounced differences in gene expression using limma. BMC Genomics 7:252
Ding X, Wang T, Shen Y, Wang X, Zhou C, Tian S, Liu Y, Peng G, Zhou J, Xue S, Wang R, Tang Y, Meng X, Pei G, Bai Y, Liu Q, Li H, Zhang J (2012) Prevalence of psoriasis in China: a population-based study in six cities. Eur J Dermatol 22:663–667
Dowlatshahi EA, Wakkee M, Arends LR, Nijsten T (2014) The prevalence and odds of depressive symptoms and clinical depression in psoriasis patients: a systematic review and meta-analysis. J Invest Dermatol 134:1542–1551
Gupta R, Ahn R, Lai K, Mullins E, Debbaneh M, Dimon M, Arron S, Liao W (2016) Landscape of long noncoding RNAs in psoriatic and healthy skin. J Invest Dermatol 136:603–609
Jiang S, Hinchliffe TE, Wu T (2015) Biomarkers of an autoimmune skin disease-psoriasis. Genom Proteom Bioinform 13:224–233
Kan JY, Wu DC, Yu FJ, Wu CY, Ho YW, Chiu YJ, Jian SF, Hung JY, Wang JY, Kuo PL (2015) Chemokine (C-C Motif) Ligand 5 is involved in tumor-associated dendritic cell-mediated colon cancer progression through non-coding RNA MALAT-1. J Cell Physiol 230:1883–1894
Keermann M, Koks S, Reimann E, Prans E, Abram K, Kingo K (2015) Transcriptional landscape of psoriasis identifies the involvement of IL36 and IL36RN. BMC Genomics 16:322
Langfelder P, Horvath S (2008) WGCNA: an R package for weighted correlation network analysis. BMC Bioinform 9:559
Leti F, Legendre C, Still CD, Chu X, Petrick A, Gerhard GS, DiStefano JK (2017) Altered expression of MALAT1 lncRNA in nonalcoholic steatohepatitis fibrosis regulates CXCL5 in hepatic stellate cells. Transl Res 190:25–39.e21
Li B, Pu K, Wu X (2019) Identifying novel biomarkers in hepatocellular carcinoma by weighted gene co-expression network analysis. J Cell Biochem 120:11418–11431
Nadeem A, Al-Harbi NO, Al-Harbi MM, El-Sherbeeny AM, Ahmad SF, Siddiqui N, Ansari MA, Zoheir KM, Attia SM, Al-Hosaini KA, Al-Sharary SD (2015) Imiquimod-induced psoriasis-like skin inflammation is suppressed by BET bromodomain inhibitor in mice through RORC/IL-17A pathway modulation. Pharmacol Res 99:248–257
Nair RP, Duffin KC, Helms C, Ding J, Stuart PE, Goldgar D, Gudjonsson JE, Li Y, Tejasvi T, Feng BJ, Ruether A, Schreiber S, Weichenthal M, Gladman D, Rahman P, Schrodi SJ, Prahalad S, Guthery SL, Fischer J, Liao W, Kwok PY, Menter A, Lathrop GM, Wise CA, Begovich AB, Voorhees JJ, Elder JT, Krueger GG, Bowcock AM, Abecasis GR (2009) Genome-wide scan reveals association of psoriasis with IL-23 and NF-kappaB pathways. Nat Genet 41:199–204
Nestle FO, Kaplan DH, Barker J (2009) Psoriasis. N Engl J Med 361:496–509
Oldham MC, Konopka G, Iwamoto K, Langfelder P, Kato T, Horvath S, Geschwind DH (2008) Functional organization of the transcriptome in human brain. Nat Neurosci 11:1271–1282
Pitule P, Vycital O, Bruha J, Novak P, Hosek P, Treska V, Hlavata I, Soucek P, Kralickova M, Liska V (2013) Differential expression and prognostic role of selected genes in colorectal cancer patients. Anticancer Res 33:4855–4865
Powe DG, Dhondalay GK, Lemetre C, Allen T, Habashy HO, Ellis IO, Rees R, Ball GR (2014) DACH1: its role as a classifier of long term good prognosis in luminal breast cancer. PLoS ONE 9:e84428
Quaranta M, Knapp B, Garzorz N, Mattii M, Pullabhatla V, Pennino D, Andres C, Traidl-Hoffmann C, Cavani A, Theis FJ, Ring J, Schmidt-Weber CB, Eyerich S, Eyerich K (2014) Intraindividual genome expression analysis reveals a specific molecular signature of psoriasis and eczema. Sci Transl Med 6:244ra90
Riis JL, Johansen C, Vestergaard C, Bech R, Kragballe K, Iversen L (2011) Kinetics and differential expression of the skin-related chemokines CCL27 and CCL17 in psoriasis, atopic dermatitis and allergic contact dermatitis. Exp Dermatol 20:789–794
Scardoni G, Petterlini M, Laudanna C (2009) Analyzing biological network parameters with CentiScaPe. Bioinformatics 25:2857–2859
Trowbridge RM, Pittelkow MR (2014) Epigenetics in the pathogenesis and pathophysiology of psoriasis vulgaris. J Drugs Dermatol 13:111–118
Van Nas A, Guhathakurta D, Wang SS, Yehya N, Horvath S, Zhang B, Ingram-Drake L, Chaudhuri G, Schadt EE, Drake TA, Arnold AP, Lusis AJ (2009) Elucidating the role of gonadal hormones in sexually dimorphic gene coexpression networks. Endocrinology 150:1235–1249
Xia M, Hu S, Fu Y, Jin W, Yi Q, Matsui Y, Yang J, McDowell MA, Sarkar S, Kalia V, Xiong N (2014) CCR10 regulates balanced maintenance and function of resident regulatory and effector T cells to promote immune homeostasis in the skin. J Allergy Clin Immunol 134:634–644.e10
Yang J, Hu S, Zhao L, Kaplan DH, Perdew GH, Xiong N (2016) Selective programming of CCR10(+) innate lymphoid cells in skin-draining lymph nodes for cutaneous homeostatic regulation. Nat Immunol 17:48–56
Zhang Y, Pitchiaya S, Cieslik M, Niknafs YS, Tien JC, Hosono Y, Iyer MK, Yazdani S, Subramaniam S, Shukla SK, Jiang X, Wang L, Liu TY, Uhl M, Gawronski AR, Qiao Y, Xiao L, Dhanasekaran SM, Juckette KM, Kunju LP, Cao X, Patel U, Batish M, Shukla GC, Paulsen MT, Ljungman M, Jiang H, Mehra R, Backofen R, Sahinalp CS, Freier SM, Watt AT, Guo S, Wei JT, Feng FY, Malik R, Chinnaiyan AM (2018) Analysis of the androgen receptor-regulated lncRNA landscape identifies a role for ARLNC1 in prostate cancer progression. Nat Genet 50:814–824
Zhao W, Langfelder P, Fuller T, Dong J, Li A, Hovarth S (2010) Weighted gene coexpression network analysis: state of the art. J Biopharm Stat 20:281–300
Acknowledgments
The authors would like to thank the Dermatology Hospital of Southern Medical University for their contribution to the support for the experiment.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest.
Ethical approval statement
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Communicated by Stefan Hohmann.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
438_2020_1654_MOESM1_ESM.tif
Supplementary file1 (TIF 237 kb) Analysis of the main components of skin lesions and healthy control samples. The blue triangle represents the psoriasis group sample, whereas the red circle represents the control group sample.
Rights and permissions
About this article
Cite this article
Li, H., Yang, C., Zhang, J. et al. Identification of potential key mRNAs and LncRNAs for psoriasis by bioinformatic analysis using weighted gene co-expression network analysis. Mol Genet Genomics 295, 741–749 (2020). https://doi.org/10.1007/s00438-020-01654-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-020-01654-0