Abstract
Porcine adrenergic receptor beta 2 (ADRB2) gene exhibits differential allelic expression in skeletal muscle, and its genetic variation has been associated with muscle pH. Exploring the molecular–genetic background of expression variation for porcine ADRB2 will provide insight into the mechanisms driving its regulatory divergence and may also contribute to unraveling the genetic basis of muscle-related traits in pigs. In the present study, we therefore examined haplotype effects on the expression of porcine ADRB2 in four tissues: longissimus dorsi muscle, liver, subcutaneous fat, and spleen. The diversity and structure of haplotypes of the proximal gene region segregating in German commercial breeds were characterized. Seven haplotypes falling into three clades were identified. Two clades including five haplotypes most likely originated from introgression of Asian genetics during formation of modern breeds. Expression analyses revealed that the Asian-derived haplotypes increase expression of the porcine ADRB2 compared to the major, wild-type haplotype independently of tissue type. In addition, several tissue-specific differences in the expression of the Asian-derived haplotypes were found. Inspection of haplotype sequences showed that differentially expressed haplotypes exhibit polymorphisms in a polyguanine tract located in the core promoter region. These findings demonstrate that expression variation of the porcine ADRB2 has a complex genetic basis and suggest that the promoter polyguanine tract is causally involved. This study highlights the challenges of finding causal genetic variants underlying complex traits.





Similar content being viewed by others
References
Andersson L (2012) How selective sweeps in domestic animals provide new insight into biological mechanisms. J Intern Med 271:1–14
Babbitt CC, Silverman JS, Haygood R, Reininga JM, Rockman MV, Wray GA (2010) Multiple functional variants in cis modulate PDYN expression. Mol Biol Evol 27:465–479
Barnes PJ (1995) Beta-adrenergic receptors and their regulation. Am J Respir Crit Care Med 152:838–860
Brodde OE, Leineweber K (2005) Beta2-adrenoceptor gene polymorphisms. Pharmacogenet Genomics 15:267–275
Cagliani R, Fumagalli M, Pozzoli U, Riva S, Comi GP, Torri F, Macciardi F, Bresolin N, Sironi M (2009) Diverse evolutionary histories for beta-adrenoreceptor genes in humans. Am J Hum Genet 85:64–75
Chikuni K, Horiuchi A, Ide H, Shibata M, Hayashi T, Nakajima I, Oe M, Muroya S (2008) Nucleotide sequence polymorphisms of beta1-, beta2-, and beta3-adrenergic receptor genes on Jinhua, Meishan, Duroc and Landrace pigs. Anim Sci J 79:665–672
Clement M, Posada D, Crandall KA (2000) TCS: a computer program to estimate gene genealogies. Mol Ecol 9:1657–1659
Cornett LE, Hiller FC, Jacobi SE, Cao W, McGraw DW (1998) Identification of a glucocorticoid response element in the rat beta2-adrenergic receptor gene. Mol Pharmacol 54:1016–1023
Cowles CR, Hirschhorn JN, Altshuler D, Lander ES (2002) Detection of regulatory variation in mouse genes. Nat Genet 32:432–437
Dimas AS, Deutsch S, Stranger BE, Montgomery SB, Borel C, Attar-Cohen H, Ingle C, Beazley C, Gutierrez AM, Sekowska M, Gagnebin M, Nisbett J, Deloukas P, Dermitzakis ET, Antonarakis SE (2009) Common regulatory variation impacts gene expression in a cell type-dependent manner. Science 325:1246–1250
Excoffier L, Slatkin M (1995) Maximum-likelihood estimation of molecular haplotype frequencies in a diploid population. Mol Biol Evol 12:921–927
Flicek P, Ahmed I, Amode MR, Barrell D, Beal K et al (2013) Ensembl 2013. Nucleic Acids Res 41:D48–D55
Forton JT, Udalova IA, Campino S, Rockett KA, Hull J, Kwiatkowski DP (2007) Localization of a long-range cis-regulatory element of IL13 by allelic transcript ratio mapping. Genome Res 17:82–87
Gaffney DJ, Veyrieras JB, Degner JF, Pique-Regi R, Pai AA, Crawford GE, Stephens M, Gilad Y, Pritchard JK (2012) Dissecting the regulatory architecture of gene expression QTLs. Genome Biol 13:R7
Ge B, Gurd S, Gaudin T, Dore C, Lepage P, Harmsen E, Hudson TJ, Pastinen T (2005) Survey of allelic expression using EST mining. Genome Res 15:1584–1591
Gemayel R, Vinces MD, Legendre M, Verstrepen KJ (2010) Variable tandem repeats accelerate evolution of coding and regulatory sequences. Annu Rev Genet 44:445–477
Groenen MA, Archibald AL, Uenishi H, Tuggle CK, Takeuchi Y et al (2012) Analyses of pig genomes provide insight into porcine demography and evolution. Nature 491:393–398
Hayes B, Goddard ME (2001) The distribution of the effects of genes affecting quantitative traits in livestock. Genet Sel Evol 33:209–229
Jiang L, Gao B, Kunos G (1996) DNA elements and protein factors involved in the transcription of the beta 2-adrenergic receptor gene in rat liver. The negative regulatory role of C/EBP alpha. Biochemistry 35:13136–13146
Johnatty SE, Abdellatif M, Shimmin L, Clark RB, Boerwinkle E (2002) Beta 2 adrenergic receptor 5′ haplotypes influence promoter activity. Br J Pharmacol 137:1213–1216
Keane TM, Goodstadt L, Danecek P, White MA, Wong K, Yalcin B, Heger A, Agam A, Slater G, Goodson M, Furlotte NA, Eskin E, Nellaker C, Whitley H, Cleak J, Janowitz D, Hernandez-Pliego P, Edwards A, Belgard TG, Oliver PL, McIntyre RE, Bhomra A, Nicod J, Gan X, Yuan W, van der Weyden L, Steward CA, Bala S, Stalker J, Mott R, Durbin R, Jackson IJ, Czechanski A, Guerra-Assuncao JA, Donahue LR, Reinholdt LG, Payseur BA, Ponting CP, Birney E, Flint J, Adams DJ (2011) Mouse genomic variation and its effect on phenotypes and gene regulation. Nature 477:289–294
Maurano MT, Humbert R, Rynes E, Thurman RE, Haugen E, Wang H, Reynolds AP, Sandstrom R, Qu H, Brody J, Shafer A, Neri F, Lee K, Kutyavin T, Stehling-Sun S, Johnson AK, Canfield TK, Giste E, Diegel M, Bates D, Hansen RS, Neph S, Sabo PJ, Heimfeld S, Raubitschek A, Ziegler S, Cotsapas C, Sotoodehnia N, Glass I, Sunyaev SR, Kaul R, Stamatoyannopoulos JA (2012) Systematic localization of common disease-associated variation in regulatory DNA. Science 337:1190–1195
McDonald MJ, Wang WC, Huang HD, Leu JY (2011) Clusters of nucleotide substitutions and insertion/deletion mutations are associated with repeat sequences. PLoS Biol 9:e1000622
Murani E, Ponsuksili S, Srikanchai T, Maak S, Wimmers K (2009) Expression of the porcine adrenergic receptor beta 2 gene in longissimus dorsi muscle is affected by cis-regulatory DNA variation. Anim Genet 40:80–89
Murani E, Reyer H, Ponsuksili S, Fritschka S, Wimmers K (2012) A substitution in the ligand binding domain of the porcine glucocorticoid receptor affects activity of the adrenal gland. PLoS One 7:e45518
Nicolae DL, Gamazon E, Zhang W, Duan S, Dolan ME, Cox NJ (2010) Trait-associated SNPs are more likely to be eQTLs: annotation to enhance discovery from GWAS. PLoS Genet 6:e1000888
Nonogaki K (2000) New insights into sympathetic regulation of glucose and fat metabolism. Diabetologia 43:533–549
Panebra A, Schwarb MR, Glinka CB, Liggett SB (2007) Allele-specific binding of airway nuclear extracts to polymorphic beta2-adrenergic receptor 5′ sequence. Am J Respir Cell Mol Biol 36:654–660
Panebra A, Wang WC, Malone MM, Pitter DR, Weiss ST, Hawkins GA, Liggett SB (2010) Common ADRB2 haplotypes derived from 26 polymorphic sites direct beta2-adrenergic receptor expression and regulation phenotypes. PLoS One 5:e11819
Ponsuksili S, Du Y, Murani E, Schwerin M, Wimmers K (2012) Elucidating molecular networks that either affect or respond to plasma cortisol concentration in target tissues of liver and muscle. Genetics 192:1109–1122
Scott MG, Swan C, Wheatley AP, Hall IP (1999) Identification of novel polymorphisms within the promoter region of the human beta2 adrenergic receptor gene. Br J Pharmacol 126:841–844
Tao H, Cox DR, Frazer KA (2006) Allele-specific KRT1 expression is a complex trait. PLoS Genet 2:e93
Templeton AR, Crandall KA, Sing CF (1992) A cladistic analysis of phenotypic associations with haplotypes inferred from restriction endonuclease mapping and DNA sequence data. III. Cladogram estimation. Genetics 132:619–633
The ENCODE Project Consortium (2012) An integrated encyclopedia of DNA elements in the human genome. Nature 489:57–74
Veyrieras JB, Kudaravalli S, Kim SY, Dermitzakis ET, Gilad Y, Stephens M, Pritchard JK (2008) High-resolution mapping of expression-QTLs yields insight into human gene regulation. PLoS Genet 4:e1000214
Vinces MD, Legendre M, Caldara M, Hagihara M, Verstrepen KJ (2009) Unstable tandem repeats in promoters confer transcriptional evolvability. Science 324:1213–1216
Wittkopp PJ, Kalay G (2011) Cis-regulatory elements: molecular mechanisms and evolutionary processes underlying divergence. Nat Rev Genet 13:59–69
Wray GA (2007) The evolutionary significance of cis-regulatory mutations. Nat Rev Genet 8:206–216
Acknowledgments
The authors thank Angela Garve, Hannelore Tychsen, and Marlies Fuchs for excellent technical help. The authors are also grateful to Aline Foury and Pierre Morméde (INRA, France) and Do Van Khoa for providing Meishan and Muong Khuong tissue samples, respectively. This research was supported by the German Research Foundation (Deutsche Forschungsgemeinschaft, Grant MU2965/2-1) and by matched funding from the FBN.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Fredholm.
Electronic supplementary material
Below is the link to the electronic supplementary material.
438_2013_776_MOESM2_ESM.tif
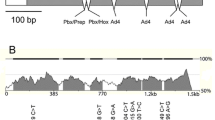
Figure S1. Position of polymorphisms in the porcine ADRB2 and flanking regions. Coding region is shown in blue, untranslated regions in yellow. Vertical arrows show position of polymorphisms. Horizontal double-headed arrows depict amplicons used for resequencing. STR – short tandem repeat, I – insertion/deletion, A – nonsynonymous variant (TIFF 267 kb)
Rights and permissions
About this article
Cite this article
Murani, E., Ponsuksili, S., Reyer, H. et al. Expression variation of the porcine ADRB2 has a complex genetic background. Mol Genet Genomics 288, 615–625 (2013). https://doi.org/10.1007/s00438-013-0776-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-013-0776-2