Abstract
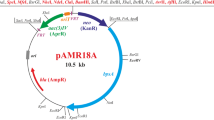
Transformation of Mucor circinelloides is routinely achieved by using a plasmid containing the wild-type leuA gene to complement the leucine requirement of an auxotrophic host strain. As is the case for other zygomycetes, the transforming DNA is usually not integrated into the genome of M. circinelloides, but is maintained as an autonomously replicating plasmid. However, even under selective conditions, the plasmid is segregationally unstable, resulting in a rather low number of cells carrying the plasmid. We report here on a new transformation vector based on a dominant selection marker conferring resistance to geneticin, which allows for plasmid maintenance in high copy numbers. The vector was also used to transform Mucor rouxii and Rhizomucor pusillus, and should therefore be a valuable tool for gene expression studies in zygomycetes. The functionality and regulatory properties of the promoter of the M. circinelloides gpd1 gene (which codes for glyceraldehyde-3P-dehydrogenase) were demonstrated in R. pusillus using geneticin selection. In this work, we have also determined the molecular basis of the Leu- phenotype of the M. circinelloides host strain R7B. The leucine requirement is due to a single point mutation in the leuA gene that results in the replacement of a glutamic acid by a lysine residue.






Similar content being viewed by others
References
Anaya N, Roncero MIG (1991) Transformation of a methionine auxotrophic mutant of Mucor circinelloides by direct cloning of the corresponding wild-type gene. Mol Gen Genet 230:449-455
Arnau J, Strøman P (1993) Gene replacement and ectopic integration in the zygomycete Mucor circinelloides. Curr Genet 23:542–546
Bartnicki-Garcia S, Nickerson WJ (1962) Induction of yeast-like development in Mucor by carbon dioxide. J Bacteriol 84:829–840
Houghton-Larsen J, Pedersen PA (2003a) Cloning and characterisation of a glucoamylase gene ( glaM) from the dimorphic zygomycete Mucor circinelloides. Appl Microbiol Biotechnol 62:210–217
Houghton-Larsen J, Pedersen PA (2003b) Functional expression of rat adenosine A1 receptor in the dimorphic zygomycete Mucor circinelloides. Appl Microbiol Biotechnol 63:64–67
Larsen GG, Appel KF, Wolff AM, Nielsen J, Arnau J (2004) Characterisation of the Mucor circinelloides regulated promoter gpd1P. Curr Genet 45:225–234
Lasker BA, Borgia PT (1980) High-frequency heterokaryon formation by Mucor circinelloides. J Bacteriol 141:565–569
Liou CM, Yanai K, Horiuchi H, Takagi M (1992) Transformation of a Leu- mutant of Rhizopus niveus with the leuA gene of Mucor circinelloides. Biosci Biotechnol Biochem 56:1503–1504
Lübbehusen T, Gonzalez Polo V, Rossi S, Nielsen J, Moreno S, McIntyre M, Arnau J (2004) Protein kinase A is involved in the control of morphology and branching during aerobic growth of Mucor circinelloides. Microbiology 150:143–150
Mackenzie DA, Wongwathanarat P, Carter AT, Archer DB (2000) Isolation and use of a homologous histone H4 promoter and a ribosomal DNA region in a transformation vector for the oil-producing fungus Mortierella alpina. Appl Environ Microbiol 66:4655–4661
Obraztsova IN, Prados N, Holzmann K, Avalos J, Cerdá-Olmedo E (2004) Genetic damage following introduction of DNA in Phycomyces. Fungal Genet Biol 41:168–180
Roncero MIG, Jepsen LP, Strøman P, van Heeswijck R (1989) Characterization of a leuA gene and an ARS element from Mucor circinelloides. Gene 84:335–343
Sambrook J, Frisch EF, Maniatis T (1989) Molecular cloning: a laboratory manual (2nd edn). Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
Torres-Guzman JC, Arreola-Garcia GA, Zazueta-Sandoval R, Carrillo-Rayas T, Martinez-Cadena G, Gutierrez-Corona F (1994) Genetic evidence for independence between fermentative metabolism (ethanol accumulation) and yeast-cell development in the dimorphic fungus Mucor rouxii. Curr Genet 26:166–171
Van Heeswijck R, Roncero MIG (1984) High frequency transformation of Mucor with recombinant plasmid DNA. Carlsberg Res Commun 49:691–702
Van Heeswijck R, Roncero MIG, Jepsen LP (1988) Genetic analysis and manipulation of Mucor species by DNA mediated transformation. In: Linskens JH, Jackson JF (eds) Modern methods of plant analysis, vol VII. Springer Verlag, Berlin, pp 207–220
Wada M, Beppu T, Horinouchi S (1996) Integrative transformation of the zygomycete Rhizomucor pusillus by homologous recombination. Appl Microbiol Biotechnol 45:652–657
Wolff AM, Arnau J (2002) Cloning of glyceraldehyde-3-phosphate dehydrogenase-encoding genes in Mucor circinelloides (syn. racemosus) and use of the gpd1 promoter for recombinant protein production. Fungal Gen Biol 35:21–29
Wöstemeyer J, Burmester A, Wiegel C (1987) Neomycin resistance as a dominantly selectable marker for transformation of the zygomycete Absidia glauca. Curr Genet 12:625–627
Acknowledgements
The work described here was partially funded by the Danish Agency for Trade and Industry through the EUKA-Centre, a collaborative industrial project. Ulla Poulsen is thanked for excellent technical assistance. The M. rouxii strain IM-80 was kindly provided by Prof. Felix Gutierrez-Corona (University of Guanajuato, Mexico). R. pusillus strain B49-7 was kindly provided by Dr. Sueharu Horinouchi (University of Tokyo, Japan)
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by E. Cerdà-Olmedo
On 1 January, 2004, the Biotechnological Institute became Bioneer A/S (http://www.bioneer.dk)
Rights and permissions
About this article
Cite this article
Appel, K.F., Wolff, A.M. & Arnau, J. A multicopy vector system for genetic studies in Mucor circinelloides and other zygomycetes. Mol Genet Genomics 271, 595–602 (2004). https://doi.org/10.1007/s00438-004-1008-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-004-1008-6