Abstract
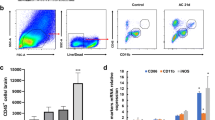
Angiostrongyliasis caused by Angiostrongylus cantonensis (A. cantonensis) is an emerging food-borne parasitic disease, which refers basically to eosinophilic meningitis. Chitinase-like protein 3 (Chil3), a member of chitinase-like protein family which has chemotactic activity for eosinophils, is reported to be highly upregulated in brain of mouse infected with A. cantonensis. The mechanisms of high expression of Chil3 and the association between A. cantonensis and Chil3 are rarely reported. In order to understand the mechanism of high expression of Chil3 in A. cantonensis-infected mouse, we measured the level of Chil3 in RAW 264.7 and BV2 cell lines stimulated with soluble antigen of A. cantonensis by qPCR and ELISA. To explore the role of Chil3 in inflammation caused by A. cantonensis, we extracted and cultured brain mononuclear cells (BMNCs) and detected the eosinophil chemotactic activity of Chil3 using transwell assay and flow cytometer. Furthermore, we treated the infected mice by injection with rmChil3 and then counted the number of larvae in brains of infected mice and treated mice to examine the association between the worm and Chil3. Our results showed the soluble antigen from A. cantonensis could promote the Chil3 expression in macrophage and microglial cell lines induced by interleukin-13. In conclusion, we supposed that high expression of Chil3 enhanced by soluble antigens from A. cantonensis might be the reason of serious eosinophil infiltration in mouse brain after A. cantonensis infection.








Similar content being viewed by others
References
Arora M, Chen L, Paglia M, Gallagher I, Allen JE, Vyas YM, Ray A, Ray P (2006) Simvastatin promotes Th2-type responses through the induction of the chitinase family member Ym1 in dendritic cells. Proc Natl Acad Sci U S A 103:7777–7782. doi:10.1073/pnas.0508492103
Chen HT (1935) Un nouveau nématode pulmonaire, Pulmonema cantonensis n. g., n. sp., des rats de Canton. Ann Parasit Hum Comp 13:312–370
Chuang CC, Chen CW, Huang YT, Du WY (2016) Anti-ST2 monoclonal antibody inhibits eosinophil infiltration in Angiostrongylus cantonensis-infected mice. J Microbiol Immunol Infect 49:91–96. doi:10.1016/j.jmii.2014.01.009
Czeh M, Gressens P, Kaindl AM (2011) The yin and yang of microglia. Dev Neurosci 33:199–209. doi:10.1159/000328989
David S, Kroner A (2011) Repertoire of microglial and macrophage responses after spinal cord injury. Nat Rev Neurosci 12:388–399. doi:10.1038/nrn3053
Du WY, Chen CW, Lin FK, Chuang CC (2013) IL-33 mediates the expressions of IL-5 and IL-13 in Angiostrongylus cantonensis-infected mice. Exp Parasitol 135:587–594. doi:10.1016/j.exppara.2013.09.012
Eamsobhana P, Yoolek A, Punthuprapasa P, Yong HS (2009) Thai koi-hoi snail dish and angiostrongyliasis due to Angiostrongylus cantonensis: effects of food flavoring and alcoholic drink on the third-stage larvae in infected snail meat. Foodborne Pathog Dis 6:401–405. doi:10.1089/fpd.2008.0191
Ferrante CJ, Pinhal-Enfield G, Elson G, Cronstein BN, Hasko G, Outram S, Leibovich SJ (2013) The adenosine-dependent angiogenic switch of macrophages to an M2-like phenotype is independent of interleukin-4 receptor alpha (IL-4Ralpha) signaling. Inflammation 36:921–931. doi:10.1007/s10753-013-9621-3
Garcia-Palacios V, Chung HY, Choi SJ, Sarmasik A, Kurihara N, Lee JW, Galson DL, Collins R, Roodman GD (2007) Eosinophil chemotactic factor-L (ECF-L) enhances osteoclast formation by increasing in osteoclast precursors expression of LFA-1 and ICAM-1. Bone 40:316–322. doi:10.1016/j.bone.2006.08.004
Gordon S (2003) Alternative activation of macrophages. Nat Rev Immunol 3:23–35. doi:10.1038/nri978
Harbord M, Novelli M, Canas B, Power D, Davis C, Godovac-Zimmermann J, Roes J, Segal AW (2002) Ym1 is a neutrophil granule protein that crystallizes in p47phox-deficient mice. J Biol Chem 277:5468–5475. doi:10.1074/jbc.M110635200
Hung SI, Chang AC, Kato I, Chang NC (2002) Transient expression of Ym1, a heparin-binding lectin, during developmental hematopoiesis and inflammation. J Leukoc Biol 72:72–82
Iwashita H, Morita S, Sagiya Y, Nakanishi A (2006) Role of eosinophil chemotactic factor by T lymphocytes on airway hyperresponsiveness in a murine model of allergic asthma. Am J Respir Cell Mol Biol 35:103–109. doi:10.1165/rcmb.2005-0134OC
Lai CH, Yen CM, Chin C, Chung HC, Kuo HC, Lin HH (2007) Eosinophilic meningitis caused by Angiostrongylus cantonensis after ingestion of raw frogs. Am J Trop Med Hyg 76:399–402
Lee CG (2009) Chitin, chitinases and chitinase-like proteins in allergic inflammation and tissue remodeling. Yonsei Med J 50:22–30. doi:10.3349/ymj.2009.50.1.22
Loke P, Gallagher I, Nair MG, Zang X, Brombacher F, Mohrs M, Allison JP, Allen JE (2007) Alternative activation is an innate response to injury that requires CD4+ T cells to be sustained during chronic infection. J Immunol 179:3926–3936
Mao Q, Xie Z, Wang X, Chen W, Ren M, Shang M, Lei H, Tian Y, Li S, Liang P, Chen T, Liang C, Xu J, Li X, Huang Y, Yu X (2015) Clonorchis sinensis ferritin heavy chain triggers free radicals and mediates inflammation signaling in human hepatic stellate cells. Parasitol Res 114:659–670. doi:10.1007/s00436-014-4230-0
Miron VE, Boyd A, Zhao JW, Yuen TJ, Ruckh JM, Shadrach JL, van Wijngaarden P, Wagers AJ, Williams A, Franklin RJ, ffrench-Constant C (2013) M2 microglia and macrophages drive oligodendrocyte differentiation during CNS remyelination. Nat Neurosci 16:1211–1218. doi:10.1038/nn.3469
Nair MG, Gallagher IJ, Taylor MD, Loke P, Coulson PS, Wilson RA, Maizels RM, Allen JE (2005) Chitinase and Fizz family members are a generalized feature of nematode infection with selective upregulation of Ym1 and Fizz1 by antigen-presenting cells. Infect Immun 73:385–394. doi:10.1128/IAI.73.1.385-394.2005
Oba Y, Chung HY, Choi SJ, Roodman GD (2003) Eosinophil chemotactic factor-L (ECF-L): a novel osteoclast stimulating factor. J Bone Miner Res 18:1332–1341. doi:10.1359/jbmr.2003.18.7.1332
Owhashi M, Arita H, Hayai N (2000) Identification of a novel eosinophil chemotactic cytokine (ECF-L) as a chitinase family protein. J Biol Chem 275:1279–1286
Owhashi M, Nawa Y (1987) Eosinophil chemotactic lymphokine produced by spleen cells of Schistosoma japonicum-infected mice. III. Isolation and characterization of two distinctive eosinophil chemotactic lymphokines directed against different maturation stages of eosinophils. Int Arch Allergy Appl Immunol 84:185–189
Peng H, Sun R, Zhang Q, Zhao J, Wei J, Zeng X, Zheng H, Wu Z (2013) Interleukin 33 mediates type 2 immunity and inflammation in the central nervous system of mice infected with Angiostrongylus cantonensis. J Infect Dis 207:860–869. doi:10.1093/infdis/jis682
Pittman K, Kubes P (2013) Damage-associated molecular patterns control neutrophil recruitment. J Innate Immun 5:315–323. doi:10.1159/000347132
Raes G, De Baetselier P, Noel W, Beschin A, Brombacher F, Hassanzadeh Gh G (2002) Differential expression of Fizz1 and Ym1 in alternatively versus classically activated macrophages. J Leukoc Biol 71:597–602
Reece JJ, Siracusa MC, Scott AL (2006) Innate immune responses to lung-stage helminth infection induce alternatively activated alveolar macrophages. Infect Immun 74:4970–4981. doi:10.1128/IAI.00687-06
Rosen L, Laigret J, Boils PL (1961) Observation on an outbreak of eosinophilic meningitis on Tahiti. French Polynesia Am J Trop Med Hyg 12:613–615
Sinawat S, Sanguansak T, Angkawinijwong T, Ratanapakorn T, Intapan PM, Sinawat S, Yospaiboon Y (2008) Ocular angiostrongyliasis: clinical study of three cases. Eye (Lond) 22:1446–1448. doi:10.1038/eye.2008.135
Sutherland TE, Logan N, Ruckerl D, Humbles AA, Allan SM, Papayannopoulos V, Stockinger B, Maizels RM, Allen JE (2014) Chitinase-like proteins promote IL-17-mediated neutrophilia in a tradeoff between nematode killing and host damage. Nat Immunol 15:1116–1125. doi:10.1038/ni.3023
Sutherland TE, Maizels RM, Allen JE (2009) Chitinases and chitinase-like proteins: potential therapeutic targets for the treatment of T-helper type 2 allergies. Clin Exp Allergy 39:943–955. doi:10.1111/j.1365-2222.2009.03243.x
Tsai ML, Liaw SH, Chang NC (2004) The crystal structure of Ym1 at 1.31 Å resolution. J Struct Biol 148:290–296. doi:10.1016/j.jsb.2004.07.002
Vainchtein ID, Vinet J, Brouwer N, Brendecke S, Biagini G, Biber K, Boddeke HW, Eggen BJ (2014) In acute experimental autoimmune encephalomyelitis, infiltrating macrophages are immune activated, whereas microglia remain immune suppressed. Glia 62:1724–1735. doi:10.1002/glia.22711
Wallace GD, Rosen L (1966) Studies on eosinophilic meningitis. 2. Experimental infection of shrimp and crabs with Angiostrongylus cantonensis. Am J Epidemiol 84:120–131
Wang QP, Lai DH, Zhu XQ, Chen XG, Lun ZR (2008) Human angiostrongyliasis. Lancet Infect Dis 8:621–630. doi:10.1016/S1473-3099(08)70229-9
Wang QP, Wu ZD, Wei J, Owen RL, Lun ZR (2012) Human Angiostrongylus cantonensis: an update. Eur J Clin Microbiol Infect Dis 31:389–395. doi:10.1007/s10096-011-1328-5
Wei J, Wu F, He A, Zeng X, Ouyang LS, Liu MS, Zheng HQ, Lei WL, Wu ZD, Lv ZY (2015) Microglia activation: one of the checkpoints in the CNS inflammation caused by Angiostrongylus cantonensis infection in rodent model. Parasitol Res 114:3247–3254. doi:10.1007/s00436-015-4541-9
Wei J, Wu F, Sun X, Zeng X, Liang JY, Zheng HQ, Yu XB, Zhang KX, Wu ZD (2013) Differences in microglia activation between rats-derived cell and mice-derived cell after stimulating by soluble antigen of IV larva from Angiostrongylus cantonensis in vitro. Parasitol Res 112:207–214. doi:10.1007/s00436-012-3127-z
Welch JS, Escoubet-Lozach L, Sykes DB, Liddiard K, Greaves DR, Glass CK (2002) TH2 cytokines and allergic challenge induce Ym1 expression in macrophages by a STAT6-dependent mechanism. J Biol Chem 277:42821–42829. doi:10.1074/jbc.M205873200
Yu L, Wu X, Wei J, Liao Q, Xu L, Luo S, Zeng X, Zhao Y, Lv Z, Wu Z (2015) Preliminary expression profile of cytokines in brain tissue of BALB/c mice with Angiostrongylus cantonensis infection. Parasit Vectors 8:328. doi:10.1186/s13071-015-0939-6
Zhao J, Lv Z, Wang F, Wei J, Zhang Q, Li S, Yang F, Zeng X, Wu X, Wu Z (2013) Ym1, an eosinophilic chemotactic factor, participates in the brain inflammation induced by Angiostrongylus cantonensis in mice. Parasitol Res 112:2689–2695. doi:10.1007/s00436-013-3436-x
Acknowledgments
This work was supported by National Natural Science Foundation of China (NSFC; Nos. 81271855 and 81261160324) to Z-D Wu, NSFC (No. 81572023) to Z-Y Lv, and NSFC (No. 81401686) to J Wei.
Authors’ contributions
FW, JW, XZ, and ZL carried out the experiments and performed the statistical analyses. FW and ZDW drafted the manuscript. ZYL and XZ helped to do the transwell assay section of the study. ZDW and XS conceived the study and coordinated the project. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Rights and permissions
About this article
Cite this article
Wu, F., Wei, J., Liu, Z. et al. Soluble antigen derived from IV larva of Angiostrongylus cantonensis promotes chitinase-like protein 3 (Chil3) expression induced by interleukin-13. Parasitol Res 115, 3737–3746 (2016). https://doi.org/10.1007/s00436-016-5135-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-016-5135-x