Abstract
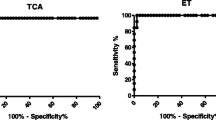
Total IgG ELISA was assessed comparing soluble adult worm (SWA), cercarial (SCA), and egg antigens (SEA) using sera of chronic schistosomiasis patients in two different concentrations (1/50 and 1/100). This response showed reactivity against all antigens. Concerning 1/100 serum concentration the SWA gave the best sensitivity (100 %) followed by SCA (86.6 %) and SEA (80 %). The best specificity was obtained from SCA (94.11 %) with an equivalent result from the SWA and SEA (76.47 %). Concerning the 1/50 serum concentration, the SWA gave the best sensitivity (100 %) with an equivalent result from the SCA and SEA (80 %). The best specificity was obtained from SEA (82.35 %) followed by SCA (76.47 %) then SWA (64.7 %). The best AUC value (0.992) was that for SWA indicating its high ability to predict the disease in comparison to (0.914) for SCA and 0.871 for SEA in serum dilution 1/100. The highest AUC value (0.955) was that for SWA in comparison to (0.914) for SEA and (0.88) for SCA in serum dilution 1/50. Comparison between the AUCs from the three mentioned antigens revealed highly significant differences when the data were analyzed by ROC. Each antigen showed a highly statistically significant correlation within its optical density values at both concentrations; 1/100 and 1/50. Comparing the two concentrations in each antigen revealed a non significant correlation obtained from the SWA which signifies a near outcome from both concentrations while the SCA and SEA showed a highly significant difference between the two concentrations. In conclusion, for the diagnosis of chronic schistosomiasis mansoni, total IgG reactivity revealed the best sensitivity by SWA. There was a difference in the type of antigen showing best specificity results between SCA and SEA according to serum concentration used.


Similar content being viewed by others
References
Abdel-Fattah MM, Al-Sherbiny M, Osman A, Charmy R, Tsang V (2011) Improving the detection limit of quantitative diagnosis of anti-S. haematobium antibodies using Falcon Assay Screening Test (FAST) ELISA by developing a new standard curve. Parasitol Res 108(6):1457–1463
Beltran S, Gourbal B, Boissier J, Duval D, Kieffer-Jaquinod S, Pierce RJ et al (2011) Vertebrate host protective immunity drives genetic diversity and antigenic polymorphism in Schistosoma mansoni. J Evol Biol 24:554–572
Bierman WF, Wetsteyn JC, van Gool T (2005) Presentation and diagnosis of imported schistosomiasis: relevance of eosinophilia, microscopy for ova, and serology. J Travel Med 12:9–13
Botelho MC, Crespo M, Almeida A, Vieira P, Delgado ML, Araujo L, Machado JC, Correia da Costa JM (2009) Schistosoma haematobium and schistosomiasis mansoni: production of an estradiol-related compound detected by ELISA. Exp Parasitol 122(3):250–253
Carod-Artal FJ (2008) Neurological complications of Schistosoma infection. Trans R Soc Trop Med Hyg 102:107–116
Cavalcanti MG, Silva LF, Peralta RH, Barreto MGM, Peralta JM (2013) Schistosomiasis in areas of low endemicity: a new era in diagnosis. Trends Parasitol 29:75–82
Cesari IM, Ballen DE, Mendoza L, Ferrer A, Pointier JP, Kombila M, Lenoble DR, Théron A (2010) Immunoblot analysis of membrane antigens of Schistosoma mansoni, Schistosoma intercalatum and Schistosoma haematobium against Schistosoma-infected patient sera. Parasitol Res 106(5):1225–1231
Chanda MA, Chiodinia PL, Doenhoff MJ (2010) Development of a new assay for the diagnosis of schistosomiasis, using cercarial antigens. Trans R Soc Trop Med Hyg 104:255–258
Chitsulo L, Engels D, Montresor A, Savioli L (2000) The global status of schistosomiasis and its control. Acta Trop 77(1):41–51
Corachán M (2002) Schistosomiasis and international travel. Clin Infect Dis 35:446–450
Cristhiany M, Pinheirol C, Carneiro TR, Hanemann A, de Oliveira SM, Schemelzer F, Bezerra M (2012) The combination of three faecal parasitological methods to improve the diagnosis of schistosomiasis mansoni in a low endemic setting in the state of Ceará, Brazil. Mem Inst Oswaldo Cruz, Rio de Janeiro 107(7):873–876
Crowther JR (2001) The ELISA guide book, 1st edn. Human Press, New Jersey
da Frota SM, Carneiro TR, Queiroz JAN, Alencar LM, Heukelbach J, Bezerra FSM (2011) Combination of Kato-Katz faecal examinations and ELISA to improve accuracy of diagnosis of intestinal schistosomiasis in a low-endemic setting in Brazil. Acta Trop 120(1):138–141
da Silva RM, Kanamura HY, Camargo ED, Chiodelli SG, Nakamura PM, Gargioni C, Vellosa SAG, Antunes JLF (1998) A comparative study on IgG-ELISA, IgM-IFT and Kato-Katz methods for epidemiological purposes in a low endemic area for schistosomiasis. Mem Inst Oswaldo Cruz, Rio de Janeiro 93(l):279–282
de Vlas SJ, Gryseels B (1992) Underestimation of Schistosoma mansoni prevalences. Parasitol Today 8:274–277
Doenhoff MJ, Chiodini PL, Hamilton JV (2004) Specific and sensitive diagnosis of Schistosoma infection: can it be done with antibodies. Trends Parasitol 20:35–39
El Ridi R, Ismail S, Gaafar T, El Demellawy M (1997) Differential responsiveness of humans with early-stage schistosomiasis haematobium to Schistosoma haematobium soluble adult-worm and egg antigens. Parasitol Res 83(5):471–477
Engels D, Sinzinkayo E, Gryseels B (1996) Day-to-day egg count fluctuation in Schistosoma mansoni and its operational implications. Am J Trop Med Hyg 54:319–324
Feldmeier H, Büttner DW (1983) Immunodiagnosis of schistosomiasis haematobium and schistosomiasis mansoni in man. Application of crude extracts from adult worms and cercariae in the IHA and the ELISA. Zentralbl Bakteriol Mikrobiol Hyg A 255:413–421
Ghandour AM, Tricker K, Doenhoff MJ, Al-Robai AA, Banaja AA (1997) An enzyme-linked immunosorbent assay using Schistosoma mansoni purified egg antigen for the diagnosis of schistosomiasis in Saudi Arabia. Trans R Soc Trop Med Hyg 91:287–289
Gryseels B, Polman K, Clerinx J, Kestens L (2006) Human schistosomiasis. Lancet 368:1106–1118
Hamilton JV, Klinkert M, Doenhoff MJ (1998) Diagnosis of schistosomiasis antibody detection, with notes on parasitological and antigen detection methods. Parasitology 117:41–57
Hotez PJ, Fenwick A (2009) Schistosomiasis in Africa: an emerging tragedy in our new global health decade. PLoS Negl Trop Dis vol 3(9):e485. doi:10.1371/journal.pntd.0000485
Hotez PJ, Savioli L, Fenwick A (2012) Neglected tropical diseases of the middle east and north africa: review of their prevalence, distribution, and opportunities for control. PLoS Negl Trop Dis 6(2):e1475. doi:10.1371/journal.pntd.0001475. Epub 2012 Feb 28
Jauréguiberry S, Paris L, Caumes E (2010) Acute schistosomiasis, a diagnostic and therapeutic challenge. Clin Microbiol Infect 16:225–231
Jelinek T, Nothdurft HD, Löscher T (1996) Schistosomiasis in travelers and expatriates. J Travel Med 3:160–164
Khalil HM, Makled MK, Elmissiry AG, Khalil NM, Sonbol SE (1989) The application of S. mansoni adult and soluble egg antigens for the serodiagnosis of schistosomiasis by CIEP, IHA and ELISA. J Egypt Soc Parasitol 19(2):827–843
Kinkel HF, Dittrich S, Baumer B, Weitzela T (2012) Evaluation of eight serological tests for diagnosis of imported schistosomiasis. Clin Vaccine Immunol 19(6):948–953
Lambertucci JR (2010) Acute schistosomiasis mansoni: revisited and reconsidered. Mem Inst Oswaldo Cruz 105:422–435
Li YL, Idris MA, Corachan M, Han JJ, Kirschfink M, Ruppel A (1996) Circulating antigens in schistosomiasis: detection of 31/32-kDa proteins in sera from patients infected with Schistosoma japonicum, S. mansoni, S. haematobium, or S. intercalatum. Parasitol Res 82(1):14–18
Li YS, Ross AG, Sleigh AC, Li Y, Waine GJ, Williams GJ, Tanner M, McManus DP (1999) Antibody isotype responses, infection and re-infection for Schistosoma japonicum in a marshland area of China. Acta Trop 73:79–92
Lunde MN, Ottesen EA, Cheever AW (1979) Serological differences between acute and chronic schistosomiasis mansoni detected by ELISA. Am J Trop Med Hyg 28:87–91
Mafuyai HB, Uneke CJ, Njoku MO, Chuga G (2006) DOT-ELISA and parasitological examination for diagnosis of Schistosoma mansoni infection in Nigeria. Helminthologia 43(1):11–15
McLaren M, Draper CG, Roberts JM, Minter-Goedbloed E, Ligthart GS, Teesdale GH et al (1978) Studies on the enzyme linked immunosorbent assay test for Schistosoma mansoni. Ann Trop Med Parasitol 72:243–253
Negra˜o-Correˆa D, Fittipaldi JF, Lambertucci JR, Teixeira MM, Antunes CMF et al (2014) Association of Schistosoma mansoni-specific IgG and IgE antibody production and clinical schistosomiasis status in a rural area of Minas Gerais, Brazil. PLoS One 9(2):e88042. doi:10.1371/journal.pone.0088042
Nicolls DJ et al (2008) Characteristics of schistosomiasis in travelers reported to the GeoSentinel Surveillance Network 1997–2008. Am J Trop Med Hyg 79:729–734
Noya O, Fermin Z, Alarco´n De Noya B, Losada S, Colmenares C, Hermoso T (1995) Humoral immune response of children with schistosomiasis. Isotype recognition of adult worm antigens. Parasite Immunol 17:319–328
Noya O, Alarcon de Noya B, Losada S, Colmenares C, Guzman C, Lorenzo MA, Bermudez H (2002) Laboratory diagnosis of schistosomiasis in areas of low transmission: a review of a line of research. Mem Inst Oswaldo Cruz 97(1):167–169
Pardo J, Carranza C, Turrientes MC, Pe´rez Arellano JL, Lo´pez Ve´lez R, Ramajo V, Muro A (2004) Utility of Schistosoma bovis adult worm antigens for diagnosis of human schistosomiasis by enzyme linked immunosorbent assay and electroimmunotransfer blot techniques. Clin Diagn Lab Immunol 11(6):1165–1170
Peakman M, Vergani D (1997) Basic and clinical immunology first edition. Churchill Livingstone, United States
Ross AG, Vickers D, Olds GR, Shah SM, McManus DP (2007) Katayama syndrome. Lancet Infect Dis 7:218–224
Salah F, El Bassiouny A, Rabia I, Demerdash Z, Roshdy M, Shaker Z (2006) Human schistosomiasis haematobium: effective diagnosis of active infection using a pair of monoclonal antibodies against soluble egg antigen. Parasitol Res 99(5):528–533
Savioli L, Stansfield S, Bundy DA, Mitchell A, Bhatia R, Engels D et al (2002) Schistosomiasis and soil-transmitted helminth infections: forging control efforts. Trans R Soc Trop Med Hyg 96:577–579
Shane HL, Verani JR, Abudho B et al (2011) Evaluation of urine CCA assays for detection of Schistosoma mansoni infection in Western Kenya. PLoS Negl Trop Dis 5(1):e951. doi:10.1371/journal.pntd.0000951
Silva RM, Kanamura HY, Camargo ED, Chiodelli SG, Nakamura PM, Gargioni C, Vellosa SAG, Antunes JL (1998) A comparative study on IgG-ELISA, IgM-IFT and Kato-Katz methods for epidemiological purposes in a low endemic area for schistosomiasis. Mem Inst Oswaldo Cruz 93(1):279–282
Siqueira LMV, Coelho PMZ, de Oliveira AA, Massara CL, Carneiro NFF, Lima ACL, Enk MJ (2011) Evaluation of two coproscopic techniques for the diagnosis of schistosomiasis in a low-transmission area in the state of Minas Gerais, Brazil. Mem Inst Oswaldo Cruz 106:844–850
Steinmann P, Keiser J, Bos R, Tanner M, Utzinger (2006) Schistosomiasis and water resources development: systematic review, meta-analysis, and estimates of people at risk. Lancet Infect Dis 6(7):411–425
Tarp B, Black FT, Petersen E (2000) The immunofluorescence antibody test (IFAT) for the diagnosis of schistosomiasis used in a non-endemic area. Trop Med Int Health 5:185–191
Tchuem Tchuente LA, Behnke JM, Gilbert FS, Southgate VR, Vercruysse J (2003) Polyparasitism with Schistosoma haematobium and soil transmitted helminth infections among school children in Loum. Cameroon Trop Med Int Health 8:975–986
Turner P, Lalloo K, Bligh J, Armstrong M, Whitty CJM, Doenhoff MJ, Chiodini PL (2004) Serological speciation of human Schistosoma infections by ELISA with a panel of three antigens. J Clin Pathol 57(11):1193–1196
Valli LC et al (1997) Efficacy of an enzyme-linked immunosorbent assay in the diagnosis of and serologic distinction between acute and chronic Schistosoma mansoni infection. Am J Trop Med Hyg 57:358–362
Van Gool T et al (2002) Serodiagnosis of imported schistosomiasis by a combination of a commercial indirect hemagglutination test with Schistosoma mansoni adult worm antigens and an enzyme-linked immunosorbent assay with S. mansoni egg antigens. J Clin Microbiol 40(9):3432–3437
Visser LG, Polderman AM, Stuiver PC (1995) Outbreak of schistosomiasis among travelers returning from Mali, West Africa. Clin Infect Dis 20:280–285
Whitty CJ, Carroll B, Armstrong M et al. (2000) Utility of history, examination and laboratory tests in screening those returning to Europe from the tropics for parasitic infection. Trop Med Int Health 5:818–823
World Health Organization Expert Committee (2002) Prevention and control of schistosomiasis and soil transmitted helminthiasis. World Health Organ Tech Rep Ser 912: 1–57
Wout AB V 't, De Jonge N, Wood SM, Van Lieshout L, Mitchell GF, Deelder AM (1995) Serum levels of circulating anodic antigen and circulating cathodic antigen detected in mice infected with Schistosoma japonicum or S. mansoni. Parasitol Res 81(5):434–437
Wout L V 't, Polderman AM, Deelder AM (2000) Immunodiagnosis of schistosomiasis by determination of the circulating antigens CAA and CCA, in particular in individuals with recent or light infections. Acta Trop 77:69–80
Xue CG, Taylor MG, Bickle QD, Savioli LE, Renganathan A (1993) Diagnosis of Schistosoma haematobium infection: evaluation of ELISA using keyhole limpet haemocyanin or soluble egg antigen in comparison with detection of eggs or haematuria. Trans Roy Soc Trop Med Hyg 87(6):654–658
Zhou X, Obuchowski N, McClish D (2002) Statistical methods in diagnostic medicine. Wiley, New York, pp 373–384
Zhu YC (2005) Immunodiagnosis and its role in schistosomiasis control in China: a review. Acta Trop 96:130–136
Zhu H, Yu C, Xia X, Dong G, Tang J, Fang L, Du Y (2010) Assessing the diagnostic accuracy of immunodiagnostic techniques in the diagnosis of schistosomiasis japonica: a meta-analysis. Parasitol Res 107(5):1067–1073
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sarhan, R.M., Aminou, H.A., Saad, G.A.R. et al. Comparative analysis of the diagnostic performance of adult, cercarial and egg antigens assessed by ELISA, in the diagnosis of chronic human Schistosoma mansoni infection. Parasitol Res 113, 3467–3476 (2014). https://doi.org/10.1007/s00436-014-4017-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-014-4017-3