Abstract
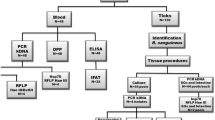
The occurrence of the insect vector (sand flies) with low rates of Leishmania infection, as well as autochthonous transmission in the absence of the natural vector in dogs, have been reported. These unexpected data suggest a hypothesis of other arthropods as a possible way of Leishmania transmission. The prevalence of Leishmania (Leishmania) infantum in fleas and ticks collected from dogs with canine visceral leishmaniasis (CVL), as well as parasite viability, were evaluated herein. The presence of L. (L.) infantum was assayed by PCR and ELISA in ectoparasites and biological samples from 73 dogs living in a Brazilian endemic area. As the occurrence of Leishmania DNA in ticks and fleas is expected given their blood-feeding habits, we next investigated whether parasites can remain viable inside ticks. PCR and ELISA confirmed that 83% of the dogs had CVL. Fleas and ticks (nymphs, male and female adults) were collected in 55% and 63% of the 73 dogs, respectively. Out of the 60 dogs with CVL, 80% harbored ectoparasites infected with L. (L.) infantum. The infection rates of the ectoparasites were 23% and 50% for fleas and ticks, respectively. The RNA analysis of the extract from ticks left in laboratory conditions during 7 to 10 days after removal from CVL dogs showed that parasites were alive. In addition, live parasites were also detected inside adult ticks recently molted in laboratory conditions. These findings indicate a higher infection rate of L. (L.) infantum in ticks and fleas, but they do not conclusively demonstrate whether these ticks can act as vectors of CVL, despite the fact that their rates were higher than those previously described in Lutzomyia longipalpis. The presence of viable L. (L.) infantum in ticks suggests the possible importance of dog ectoparasites in CVL dissemination.

Similar content being viewed by others
References
Aagaard-Hansen J, Nombela N, Alvar J (2010) Population movement: a key factor in the epidemiology of neglected tropical diseases. Trop Med Int Health 15:1281–1288
Aragão HB (1936) Ixodidas brasileiros e de alguns paizes limitrophes. Mem Inst Oswaldo Cruz 31:759–844
Armstrong TC, Patterson JL (1994) Cultivation of Leishmania braziliensis in an economical serum-free medium containing human urine. J Parasitol 80:1030–1032
Baneth G, Koutinas AF, Solano-Gallego L, Bourdeau P, Ferrer F (2008) Canine leishmaniasis—new concepts and insights on an expanding zoonosis: part one. Trends Parasitol 24:324–330
Blanc G, Caminopetros J (1930) La transmission du Kala—Azar méditerranéen par une tique: Rhipicephalus sanguineus. Compt Rendus Acad Sci 191:1162–1164
Camargo-Neves VLF (2004) A leishmaniose visceral americana no Estado de São Paulo. Estado atual. Bol Epid Paul 6. At: http://www.cve.saude.sp.gov.br
Costa CHN, Gomes ACG, Costa JML, Vieira JBF, Lima JWO, Dietz R (2001) Changes in the control program of visceral leishmaniasis in Brazil. Rev Soc Bras Med Trop 34:223–228
Coutinho MT, Linardi PM (2007) Can fleas from dogs infected with canine visceral leishmaniasis transfer the infection to other mammals? Vet Parasitol 147:320–325
Coutinho MT, Bueno LL, Sterzik A, Fujiwara RT, Botelho JR, Maria M, Genaro O, Linardi PM (2005) Participation of Rhipicephalus sanguineus (Acari: Ixodidae) in the epidemiology of canine visceral leishmaniasis. Vet Parasitol 128:149–155
CVE–Centro de Vigilância Epidemiológica do Estado de São Paulo (Surveillance Epidemiological Center of Sao Paulo State) (2010). Site in Portuguese. At: http://www.cve.saude.sp.gov.br
Dantas-Torres F (2006) Current epidemiological status of visceral leishmaniasis in Northeastern Brazil. Rev Saude Pub 40:537–541
Dantas-Torres F (2007) The role of dogs as reservoirs of Leishmania parasites, with emphasis on Leishmania (Leishmania) infantum and Leishmania (Viannia) braziliensis. Vet Parasitol 10:139–146
Dantas-Torres F, Faustino MAG, Lima OC, Acioli RV (2005) Epidemiologic surveillance of canine visceral leishmaniasis in the municipality of Recife, Pernambuco. Rev Soc Bras Med Trop 38:444–445
Dantas-Torres F, Lorusso V, Testini G, de Paiva-Cavalcanti M, Figueredo LA, Stanneck D, Mencke N, Brandão-Filho SP, Alves LC, Otranto D (2010a) Detection of Leishmania infantum in Rhipicephalus sanguineus ticks from Brazil and Italy. Parasitol Res 106:857–860
Dantas-Torres F, Martins TF, de Paiva-Cavalcanti M, Figueredo LA, Lima BS, Brandão-Filho SP (2010b) Transovarial passage of Leishmania infantum kDNA in artificially infected Rhipicephalus sanguineus. Exp Parasitol 125:184–185
Degrave W, Fernandes O, Campebell D, Bozza M, Lopes U (1994) Use of molecular probes and PCR for detection and typing of Leishmania—a mini review. Mem Inst Oswaldo Cruz 89:463–469
Díaz-Espińeira MM, Slappendel RJ (1997) A case of autochthonous canine leishmaniasis in the Netherlands. Vet Q 19:69–71
Duprey ZH, Steurer FJ, Rooney JA, Kirchhoff LV, Jackson JE, Rowton ED, Schantz PM (2006) Canine visceral leishmaniasis, United States and Canada, 2000-2003. Emerg Infect Dis 12:440–446
Ferreira MG, Fattori KR, Souza F, Lima VM (2009) Potential role for dog fleas in the cycle of Leishmania spp. Vet Parasitol 165:150–154
Freitas E, Melo MN, Costa-Val AP, Michalick MS (2006) Transmission of Leishmania infantum via blood transfusion in dogs: potential for infection and importance of clinical factors. Vet Parasitol 137:159–167
Gaskin AA, Schantz P, Jackson J, Birkenheuer A, Tomlinson L, Gramiccia M, Levy M, Steurer F, Kollmar E, Hegarty BC, Ahn A, Breitschwerdt EB (2002) Visceral leishmaniasis in a New York foxhound kennel. J Vet Intern Med 16:34–44
Giraud P, Ranque J, Cabassu H (1954) Epidemiologie de la leishmaniose viscérale humaine mediterranéenne, en particulier dans sés rapports avec la leishmaniose canine. Arch Fr Ped 11:337–353
Gomes AH, Ferreira IM, Lima ML, Cunha EA, Garcia AS, Araujo MF, Pereira-Chioccola VL (2007) PCR identification of Leishmania in diagnosis and control of canine leishmaniasis. Vet Parasitol 144:234–241
Grimaldi G, Tesh RB (1993) Leishmaniasis of the New World: current concepts and implications for the future research. Clin Microbiol Rev 6:230–250
Kullberg M, Nilsson MA, Arnason U, Harley EH, Janke A (2006) Housekeeping genes for phylogenetic analysis of eutherian relationships. Mol Biol Evol 23:1493–1503
Lachaud L, Marchergui-Hammami S, Chabbert E, Dereure J, Dedet JP, Bastien P (2002) Comparison of six PCR methods using peripheral blood for detection of canine visceral leishmaniasis. J Clin Microbiol 40:210–215
Lainson R, Rangel EF (2005) Lutzomyia longipalpis and the eco-epidemiology of American visceral leishmaniasis, with particular reference to Brazil–a review. Mem Inst Oswaldo Cruz 100:811–827
Lainson R, Shaw JJ (1998) New world leishmaniasis–the neotropical Leishmania species. In: Collier L, Balows A, Sussman M (eds) Topley & Wilson's microbiology and microbial infections, vol 5, 9th edn. Wiley, London
Le Fichoux Y, Quaranta JF, Aufeuvre JP, Lelievre A, Marty P, Suffia I, Rousseau D, Kubar J (1999) Occurrence of Leishmania infantum parasitemia in asymptomatic blood donors living in an area of endemicity in southern France. J Clin Microbiol 37:1953–1957
Linari PM, Guimarães LR (2000) Sifonápteros do Brasil. Ed. Museu de Zoologia USP/FAPESP. 291pp
Michalsky EM, Fortes-Dias CL, França-Silva JC, Rocha MF, Barata RA, Dias ES (2009) Association of Lutzomyia longipalpis (Diptera: Psychodidae) population density with climate variables in Montes Claros, an area of American visceral leishmaniasis transmission in the state of Minas Gerais, Brazil. Mem Inst Oswaldo Cruz 104:1191–1193
Missawa NA, Dias ES (2007) Phlebotomine sand flies (Diptera: Psychodidae) in the municipality of Várzea Grande: an area of transmission of visceral leishmaniasis in the state of Mato Grosso, Brazil. Mem Inst Oswaldo Cruz 102:913–918
MSB–Ministerio da Saude do Brasil (Health Ministry of Brazil) (2010) Manual de vigilância e controle da leishmaniose visceral (visceral leishmaniasis: surveillance and control. Technical manual) (in Portuguese). At http://portal.saude.gov.br/portal/svs
Odorizzi RM, Galati EA (2007) Sand flies in the Aguapeí river floodplain, northwest area of State of São Paulo, Brazil. Rev Saude Publ 41:645–652
Paz GF, Ribeiro MF, Michalsky EM, da Rocha Lima AC, França-Silva JC, Barata RA, Fortes-Dias CL, Dias ES (2010) Evaluation of the vectorial capacity of Rhipicephalus sanguineus (Acari: Ixodidae) in the transmission of canine visceral leishmaniasis. Parasitol Res 6:523–528
Pereira-Chioccola VL (2009) Molecular diagnosis of leishmaniasis: contribution to the American visceral leishmaniasis surveillance program in Sao Paulo state. Bol Epidemiol Paul 6:4–13. doi:http://www.cve.saude.sp.gov.br
Ravel S, Cuny G, Reynes J, Veas F (1995) A highly sensitive and rapid procedure for direct PCR detection of Leishmania infantum within human peripheral blood mononuclear cells. Acta Trop 59:187–196
Rosypal AC, Troy GC, Zajac AM, Frank G, Lindsay DS (2005) Transplacental transmission of a North American isolate of Leishmania infantum in an experimentally infected beagle. J Parasitol 91:970–972
Savani ES, Nunes VL, Galati EA, Castilho TM, Zampieri RA, Floeter-Winter LM (2009) The finding of Lutzomyia almerioi and Lutzomyia longipalpis naturally infected by Leishmania spp. in a cutaneous and canine visceral leishmaniases focus in Serra da Bodoquena, Brazil. Vet Parasitol 160:18–24
Schantz PM, Steurer FJ, Duprey ZH, Kurpel KP, Barr SC, Jackson JE, Breitschwerdt EB, Levy MG, Fox JC (2005) Autochthonous visceral leishmaniasis in dogs in North America. J Am Vet Med Assoc 226:1316–1322
Sherlock IA (1964) Nota sobre a transmissão da leishmaniose visceral no Brasil. Rev Bras Malariol Doenças Trop 16:19–26
Silva FL, Oliveira RG, Silva TM, Xavier MN, Nascimento EF, Santos RL (2009a) Venereal transmission of canine visceral leishmaniasis. Vet Parasitol 160:55–59
Silva SM, Ribeiro VM, Ribeiro RR, Tafuri WL, Melo MN, Michalick MSM (2009b) First report of vertical transmission of Leishmania (Leishmania) infantum in a naturally infected bitch from Brazil. Vet Parasitol 3:159–162
Stuart K, Brun R, Croft S, Fairlamb A, Gurtler RE, McKerrow J, Reed S, Tarleton R (2008) Kinetoplastids: related protozoan pathogens, different diseases. J Clin Invest 118:1301–1310
Troughton DR, Levin ML (2007) Life cycles of seven ixodid tick species (Acari: Ixodidae) under standardized laboratory conditions. J Med Entomol 44:732–740
WHO–World Health Organization (2010) At: http://gamapserver.who.int/mapLibrary/Files/Documents/DD_visceral.pdf
Acknowledgments
This study was supported by grants from Fundaçao de Amparo à Pesquisa do Estado de Sao Paulo, Brazil (FAPESP) (proc 08/00520-6). FAC was supported by fellowships from FAPESP (proc-08/57245-7).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Colombo, F.A., Odorizzi, R.M.F.N., Laurenti, M.D. et al. Detection of Leishmania (Leishmania) infantum RNA in fleas and ticks collected from naturally infected dogs. Parasitol Res 109, 267–274 (2011). https://doi.org/10.1007/s00436-010-2247-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-010-2247-6