Abstract
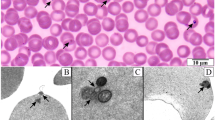
The aim of this study was to perform in vitro cultivation of Babesia canis protozoa isolated from dogs with clinical babesiosis. A primary culture initiated in RPMI-1640 medium supplemented with 40% canine serum supported parasite growth in vitro in 5% carbon dioxide in air atmosphere. Subsequent subcultures into HL-1 medium with 40% dog serum or EMEM with 40% foetal bovine serum also supported parasite propagation. The parasites have been continuously cultured through six passages, although the parasitemias are low, ranging from 0.56% to 0.59%. The partial small subunit ribosomal rRNA gene sequence was identical in blood-derived and culture-derived Babesia. The parasites from 17 cultures were classified as EU622792, and from 13 cultures as EU622793. These data show that an efficient in vitro cultivation of B. canis could serve as a starting point to obtain a protozoan antigen used for immunisation of the dogs against piroplasmosis


Similar content being viewed by others
References
Adaszek Ł, Winiarczyk S (2008) Molecular characterization of Babesia canis canis isolates from naturally infected dogs in Poland. Vet Parasitol 152:235–241
Adaszek Ł, Winiarczyk S (2010) Application of the SYBR Green real-time HRM PCR technique in the differentiation of the Babesia canis canis protozoa isolated in the areas of eastern Poland. Parasitol Res 106:1253–1256
Adaszek Ł, Winiarczyk S, Skrzypczak M (2009a) The clinical course of babesiosis in 76 dogs infected with protozoa parasites Babesia canis canis. Pol J Vet Sci 12:81–87
Adaszek Ł, Winiarczyk S, Ziętek J (2009b) In vitro cultivation of Babesia organisms isolated from animals [in Polish]. Med Wet 65:12–14
Adaszek Ł, Winiarczyk S, Górna M (2010) From piroplasmosis to babesiosis—problems with classification of Babesia protozoa isolated from dogs. Wiad Parazytol 56:111–115
Brkljacić M, Matijatko V, Kis I, Kucer N, Forsek J, Rafaj RB, Grden D, Torti M, Mayer I, Mrljak V (2010) Molecular evidence of natural infection with Babesia canis canis in Croatia. Acta Vet Hung 58:39–46
Camacho AT, Pallas E, Gestal JJ, Guitiàn FJ, Olmeda AS, Goethert HK, Telford SR (2001) Infection of dogs in north–west Spain with a Babesia microti-like agent. Vet Rec 149:552–555
Carret C, Walas F, Carcy B, Grande N, Précigout É, Moubri K, Schetters T, Gorenflot A (1999) Babesia canis canis, Babesia canis vogeli, Babesia canis rossi: differentiation of three subspecies by restriction fragment length polymorphism analysis on amplified small subunit ribosomal RNA genes. J Eukaryot Microbiol 46:298–303
Conrad P, Thomford J, Yamane I, Whiting J, Bosma L, Uno T, Holshuh HJ, Shelley S (1991) Hemolytic anemia caused by Babesia gibsoni infections in dogs. J Am Vet Med Assoc 199:601–605
Holman PJ, Chieves L, Frerichs WM, Olson D, Wagner GG (1994) Babesia equi erythrocytic stage continuously cultured in an enriched medium. J Parasitol 80:232–236
Holman PJ, Becu T, Bakos E, Polledo G, Cruz D, Wagner GG (1998) Babesia equi field isolates cultured from horse blood using a microcentrifuge method. J Parasitol 84:696–699
Holman PJ, Spencer AM, Droleskey RE, Goethert HK, Telford SR 3rd (2005) In vitro cultivation of a zoonotic Babesia sp. isolated from eastern cottontail rabbits (Sylvilagus floridanus) on Nantucket Island, Massachusetts. J Clin Microbiol 43:3995–4001
Jackson LA, Waldron SJ, Weier HM, Nicoll CL, Cooke BM (2001) Babesia bovis: culture of laboratory-adapted parasite lines and clinical isolates in a chemically defined medium. Exp Parasitol 99:168–174
Kjemtrup AM, Wainwright K, Miller M, Penzhorn BL, Carreno RA (2006) Babesia conradae, sp. nov., a small canine Babesia identified in California. Vet Parasitol 138:103–111
Lehtinen LE, Birkenheuer AJ, Droleskey RE, Holman PJ (2008) In vitro cultivation of a newly recognized Babesia sp. in dogs in North Carolina. Vet Parasitol 151:150–151
Levy MG, Ristic M (1980) Babesia bovis: continuous cultivation in a microaerophilus stationary phase culture. Science 207:218–222
Neves L, Cross HF, Loureiro L, Akça A, Hommel M, Trees AJ (2001) Addition of hypoxanthine to culture media allows in vitro cultivation of Babesia bovis and B. bigemina at reduced serum concentrations. Parasitology 123:357–363
Schetters TP, Moubri K, Précigout E, Kleuskens J, Scholtes NC, Gorenflot A (1997) Different Babesia canis isolates, different diseases. Parasitology 115:485–493
Schetters TP, Kleuskens JA, Scholtes NC, Gorenflot A, Moubri K, Vermeulen AN (2001) Vaccination of dogs against heterologous Babesia canis infection using antigens from culture supernatants. Vet Parasitol 100:75–86
Schuster FL (2002) Cultivation of Babesia and Babesia-like blood parasites: agents of an emerging zoonotic disease. Clin Microbiol Rev 15:365–373
Spencer AM, Goethert HK, Telford SR 3rd, Holman PJ (2006) In vitro host erythrocyte specificity and differential morphology of Babesia divergens and a zoonotic Babesia sp. from eastern cottontail rabbits (Sylvilagus floridanus). J Parasitol 92:333–340
Zahler M, Rinder H, Schein E, Gothe R (2000) Detection of a new pathogenic Babesia microt i-like species in dogs. Vet Parasitol 89:241–248
Zintl A, Skerrett HE, Gray JS, Brophy PO, Mulcahy G (2004) Babesia divergens (Phylum Apicomplexa) in vitro growth in the presence of calf serum. Vet Parasitol 122:127–130
Zweygarth E, Van Niekerk C, Just MC, De Waal DT (1995) In vitro cultivation of a Babesia sp. from cattle in South Africa. Onderstepoort J Vet Res 62:139–142
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Adaszek, Ł., Winiarczyk, S. In vitro cultivation of Babesia canis canis parasites isolated from dogs in Poland. Parasitol Res 108, 1303–1307 (2011). https://doi.org/10.1007/s00436-010-2181-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-010-2181-7