Abstract
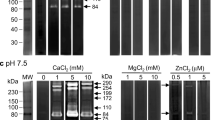
Ingestion of the larval nematode Angiostrongylus cantonensis can cause the human eosinophilic meningitis known as angiostrongyliasis. Analysis of the extracts and excretory-secretory (ES) products of A. cantonensis larvae and adult stages on gelatin substrate zymography demonstrated the presence of distinct gelatinolytic enzymes. In worm extracts, inhibitor studies showed that the metalloproteinases revealed in L1 (23 kDa), L3 (66, 42 and 30 kDa), young adult worm (72 and 94 kDa) and adult worm (72 and 94 kDa). In ES products, the L1 revealed one low (42 kDa) and two high (105 and 94 kDa) molecular weight proteolytic bands that degraded gelatin in substrate gels. The L3 revealed three low (66, 50, and 30 kDa) and one high (105 kDa) molecular weight proteolytic bands. Inhibitor studies confirmed that the 105 and 94 proteolytic bands of the L1, and the 50 and 30 kDa proteolytic bands of the L3 classification were metalloproteinases. These metalloproteinases secreted in the infective larvae may be associated with the parasite dissemination or pathogenesis.



Similar content being viewed by others
References
Alicata JE (1965) Biology and distribution of the rat lungworm, Angiostrongylus cantonensis, and its relationship to eosinophilic meningoencephalitis and other neurological disorders of man and animals. Adv Parasitol 3:223–248
Ash LR (1970) Diagnostic morphology of the third-stage larvae of Angiostrongylus cantonensis, Angiostrongylus vasorum, Aelurostrongylus abstrusus, and Anafilaroides rostratus (Nematoda: Metastrongyloidea). J Parasitol 56:249–253
Bessarab IN, Joshua GW (1997) Stage-specific gene expression in Angiostrongylus cantonensis: characterisation and expression of an adult-specific gene. Mol Biochem Parasitol 88:73–84
Harrop SA, Sawangjaroen N, Prociv P, Brindley PJ (1995) Characterization and localization of cathepsin B proteinases expressed by adult Ancylostoma caninum hookworms. Mol Biochem Parasitol 71:163–171
Hawdon JM, Jones BF, Perregaux MA, Hotez PJ (1995) Ancylostoma caninum: metalloprotease release coincides with activation of infective larvae in vitro. Exp Parasitol 80:205–211
Healer J, Ashall F, Maizels RM (1991) Characterization of proteolytic enzymes from larval and adult Nippostrongylus brasiliensis. Parasitology 103:305–314
Hill DE, Gamble HR, Rhoads ML, Fetterer RH, Urban JF Jr (1993) Trichuris suis: a zinc metalloprotease from culture fluids of adult parasites. Exp Parasitol 77:170–178
Hotez PJ, Trang NL, McKerrow JH, Cerami A (1985) Isolation and characterization of a proteolytic enzyme from the adult hookworm Ancylostoma caninum. J Biol Chem 260:7343–7348
Joshua GW, Perler FB, Wang CC (1995) Orphon spliced-leader sequences form part of a repetitive element in Angiostrongylus cantonensis. Nucl Acids Res 23:1030–1035
Kliks MM, Palumbo NE (1992) Eosinophilic meningitis beyond the Pacific Basin: the global dispersal of a peridomestic zoonosis caused by Angiostrongylus cantonensis, the nematode lungworm of rats. Soc Sci Med 34:199–212
Knox DP, Kennedy MW (1988) Proteinases released by the parasitic larval stages of Ascaris suum, and their inhibiton by antibody. Mol Biochem Parasitol 28:207–216
Lackey A, James ER, Sakanari JA, Resnick SD, Brown M, Bianco AE, McKerrow JH (1989) Extracellular proteases of Onchocerca. Exp Parasitol 68:176–185
Lai SC, Twu JJ, Jiang ST, Hsu JD, Chen KM, Chiaing HC, Wang CJ, Tseng CK, Shyu LY, Lee HH (2004) Induction of matrix metalloproteinase-9 in murine eosinophilic meningitis caused by Angiostrongylus cantonensis. Ann Trop Med Parasitol 98:715–724
Lee HH, Chou HL, Chen KM, Lai SC (2004) Association of matrix-metalloproteinase-9 in eosinophilic meningitis of BALB/c mice caused by Angiostrongylus cantonensis. Parasitol Res 94:321–328
Lindo JF, Waugh C, Hall J, Cunningham-Myrie C, Ashley D, Eberhard ML, Sullivan JJ, Bishop HS, Robinson DG, Holtz T, Robinson RD (2002) Enzootic Angiostrongylus cantonensis in rats and snails after an outbreak of human eosinophilic meningitis, Jamaica. Emerg Infect Dis 8:324–326
Loukas A, Selzer PM, Maizels RM (1998) Characterisation of Tc-cp1–1, a cathepsin L-like cysteine protease from Toxocara canis infective larvae. Mol Biochem Parasitol 92:275–289
Matrisian LM (1992) The matrix-degrading metalloproteinases. Bioessays 14:455–463
Matthews BE (1982) Behaviour and enzyme release by Anisakis sp larvae (Nematoda: Ascaridida). J Helminthol 56:177–183
McKerrow JH (1989) Parasite proteases. Exp Parasitol 68:111–115
McKerrow JH, Doenhoff MJ (1988) Schistosoma proteases. Parasitol Today 12:334–339
McKerrow JH, Brindley P, Brown M, Gam AA, Staunton C, Neva FA (1990) Strongyloides stercoralis: identification of a protease that facilitates penetration of skin by the infective larvae. Exp Parasitol 70:134–143
Morris SR, Sakanari JA (1994) Characterization of the serine protease and serine protease inhibitor from the tissue-penetrating nematode Anisakis simplex. J Biol Chem 269:27650–27656
Parsons JC, Grieve RB (1990) Effect of egg dosage and host genotype on liver trapping in murine larval toxocariasis. J Parasitol 76:53–58
Petralanda I, Yarzabal L, Piessens WF (1986) Studies on a filarial antigen with collagenase activity. Mol Biochem Parasitol 19:51–59
Richer JK, Sakanari JA, Frank GR, Grieve RB (1992) Dirofilaria immitis: proteases produced by third- and fourth-stage larvae. Exp Parasitol 75:213–222
Robertson BD, Bianco AT, McKerrow JH, Maizels RM (1989) Toxocara canis: proteolytic enzymes secreted by the infective larvae in vitro. Exp Parasitol 69:30–36
Sato H, Seiki M (1996) Membrane-type matrix metalloproteinases (MT-MMPs) in tumor metastasis. J Biochem 119:209–215
Stetler-Stevenson WG, Aznavoorian S, Liotta LA (1993) Tumor cell interactions with the extracellular matrix during invasion and metastasis. Annu Rev Cell Biol 9:541–573
Tort J, Brindley PJ, Knox D, Wolfe KH, Dalton JP (1999) Proteinases and associated genes of parasitic helminths. Adv Parasitol 43:161–266
Uparanukraw P, Morakote N, Harnnoi T, Dantrakool A (2001) Molecular cloning of a gene encoding matrix metalloproteinase-like protein from Gnathostoma spinigerum. Parasitol Res 87:751–757
Wada K, Sato H, Kinoh H, Kajita M, Yamamoto H, Seiki M (1998) Cloning of three Caenorhabditis elegans genes potentially encoding novel matrix metalloproteinases. Gene 211:57–62
Wang LC, Chao D, Chen ER (1991) Experimental infection routes of Angiostrongylus cantonensis in mice. J Helminthol 65:296–300
Acknowledgement
This study was supported by a grant (NSC-90–2320–B040–021) from the National Science Council, Republic of China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lai, S.C., Jiang, S.T., Chen, K.M. et al. Matrix metalloproteinases activity demonstrated in the infective stage of the nematodes, Angiostrongylus cantonensis. Parasitol Res 97, 466–471 (2005). https://doi.org/10.1007/s00436-005-1484-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-005-1484-6