Abstract
For more than a century, the orbital angle has been studied by many authors to distinguish dog skulls from their progenitor, the wolf. In early studies, the angle was reported to be different between dogs (49°–55°) and wolves (39°–46°). This clear difference was, however, questioned in a more recent Scandinavian study that shows some overlap. It is clear that in all studies several methodological issues were unexplored or unclear and that group sizes and the variety of breeds and wolf subspecies were small. Archaeological dog skulls had also not been studied. Our goal was to test larger and more varied groups and add archaeological samples as they are an evolutionary stage between wolves and modern dogs. We also tested the influence of measuring methods, intra- and inter-reliability, angle symmetry, the influence of variations in skull position and the possibility of measuring and comparing this angle on 3D CT scan images. Our results indicate that there is about 50 % overlap between the angle range in wolves and modern dogs. However, skulls with a very narrow orbital angle were only found in wolves and those with a very wide angle only in dogs. Archaeological dogs have a mean angle very close to the one of the wolves. Symmetry is highest in wolves and lowest in archaeological dogs. The measuring method is very reliable, for both inter- and intra-reliability (0.99–0.97), and most skull position changes have no statistical influence on the angle measured. Three-dimensional CT scan images can be used to measure OA, but the angles differ from direct measuring and cannot be used for comparison. Evolutionary changes in dog skulls responsible for the wider OA compared to wolf skulls are mainly the lateralisation of the zygomatic process of the frontal bone. Our conclusion is that the orbital angle can be used as an additional morphological measuring method to discern wolves from recent and archaeological dogs. Angles above 60° are certainly from recent dogs. Angles under 35° are certainly of wolves.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The domestication of wolves into dogs is currently actively debated (Boudadi-Maligne and Escarguel 2014; Germonpré et al. 2012; Larson and Burger 2013; Larson et al. 2012; Morey 2014). Where and when dogs originated and how to distinguish dog remains from those of wolves have been investigated by both osteo-archaeologists (morphologists) (Aaris-Sørensen 1977, 2004; Benecke 1987, 1994; Boudadi-Maligne and Escarguel 2014; Huxley 1880; Iljin 1941; Köhler and Moyà-Solà 2004; Nehring 1888; Rütimeyer 1861, 1875; Stockhaus 1965; Studer 1901; Sumiński 1975; Von Den Driesch 1976) and geneticists (Boyko et al. 2010; Druzhkova et al. 2013; Freedman et al. 2014; Gundry et al. 2007; Leonard et al. 2002; Natanaelsson et al. 2006; Savolainen et al. 2002; Vonholdt et al. 2010). In essence three morphological methods have been used: classical morphology and thus observable differences in form (Olsen and Olsen 1977), classical morphometry: measuring sizes and ratios (Benecke 1994; Morey 1986, 1992; Wayne 1986; Wolfgram 1894) and more recently, geometric morphometrics (Pionnier-Capitan 2010). These methods describe objective differences between the two sub-species and help to determine whether archaeological skulls belonged to wolf or dog. As such they contribute significantly to the question of when and where dogs were domesticated.
The most important morphological and morphometric differences used to distinguish dogs from wolves and regarding dogs are: smaller stature, shorter and wider snouts, shorter carnassials, tooth crowding and wider orbital angles (Benecke 1987; Clutton-Brock 1962; Degerbøl 1961; Stockhaus 1965; Studer 1901; Wolfgram 1894). The orbital angle (OA) is a morphological ratio that depends on the width and height of specific skull landmarks. The method was developed by Studer and applied by observing the skull in rostral view (Fig. 1) (Studer 1901). Studer described the first leg of the angle as a horizontal line on top of the frontal bones. The second leg was defined by “placing a plane (Ebene) against the lateral side of the skull, in contact with two points of the orbita (Augenrand)”. Studer (1901) defined the upper contact point of the oblique leg as the most lateral point of the zygomatic process of the frontal bone (ZP), while the most ventral contact point was described as the most dorsolateral point of the frontal process of the zygomatic arch (FP) (Figs. 1, 2). The right figure in the original publication shows exactly the method as described in words (Fig. 1). The left figure, however, shows “another story” (Fig. 1). Here, the oblique leg is in contact with a different landmark at the ventral side: the dorsal rim of the zygomatic arch (ZA) (Figs. 1, 2) which is not part of the orbita and thus “sensu stricto” the use of this last point does not measure a “real” Orbital angle. While the difference between both ventral contact points may seem minimal seen in rostral view, there is a huge difference when seen from lateral (Fig. 3). Also, if ZA is used as the ventral contact point, the OA will be narrower compared to when FP would be used in the same skull. The reason for the two possible ventral contact points lies in the use of the “Ebene”: this plane touches the skull at the widest of the two anatomical structures. Studer does not report this difference.
OA as depicted in the original Studer (1901) publication (Figure 1, p. 4). The skull is seen from rostral view. Left dog, right wolf skull. The OA is the dorsal angle between a horizontal leg on top of the frontal bones and tan oblique leg. The oblique leg can be drawn in two different ways: the dorsal contact point is identical in both (ZP), and the ventral contact point is the most lateral structure of two points to come in contact with “the measuring plane”. This ventral point can be the most dorsolateral point of the zygomatic arch (ZA) as in the dog skull, or the FP as shown in the wolf skull
Horizontal line on top of the frontal bones represents the first leg of the angle. The oblique leg of the angle can be drawn in two ways: the dorsal contact point is stable (ZP) and the ventral contact point is the most lateral structure to contact “the measuring plane”, this is either ZA (as in this skull) or FP
Studer (1901) measured 21 wolf (19 Eurasian) and 24 dog skulls (>20 breeds) with 1° precision. Several other authors measured the OA with 0.5° precision; they were: Bockelmann (1920) who measured four Eurasian wolves and three German shepherds, Iljin (1941) who measured four Eurasian wolves and four German shepherds, Aaris-Sørensen (1977) who measured 35 Eurasian wolves (of which some were sub-fossils) and 35 dogs from three different breeds (including nine German shepherds) and Sablin and Khlopachev (2002) who measured two prehistoric putative dogs from Eliseevichi (Aaris-Sørensen 1977; Bockelmann 1920; Iljin 1941; Sablin and Khlopachev 2002). None of the authors after Studer (1901) described their methodology in detail. As most of these studies used Eurasian wolves only and mainly German shepherds as dogs, the variability of groups was very narrow. Most studies found distinct OA differences between wolves (39.5°–46.5°) and dogs (49°–55°). Aaris-Sørensen (1977) did, however, describe wider ranges in dogs (44°–56°) and wolves (36°–49°). He also reported an overlap (44°–49°), contrary to his predecessors, making the method less reliable and more restricted in use. Many methodological facets were clearly unreported in the former studies: Was an OA measured once or more often? Were means reported? Were all measurements performed by only one researcher and was intra- and inter-reliability considered? What was the difference in degrees when using the two different oblique legs of the angle? And in what percentage does one measure the “sensu stricto” OA? Was the angle measured on one or both sides of the skull and was fluctuating asymmetry taken into account? Perfect symmetry (defined as stability) is considered to be perfection of development. Failure to achieve symmetry is called developmental instability. Symmetry informs on the potential of the organism (both individuals and groups) to cope and channel its development during growth in an imperfect environment (Van Dongen 2006) and thus on the amount of environmental stress, and on the (partially hereditary) fitness of the organism. A higher degree of symmetry is observed in more dominant (higher in hierarchy) and more sexually attractive individuals with more offspring (De Coster et al. 2013). Species that originated recently show higher degrees of asymmetry than long existing species that have had time to develop higher developmental stability (De Coster et al. 2013; Van Dongen 2006; Van Dongen et al. 2009). For this aspect it might be of interest to compare symmetry in modern dogs (a modern species), wolves (a long existing species) and archaeological dogs (an early domesticated species).
Our aim in the present study was to address the methodological questions listed above and also to examine whether measuring larger groups with more variability (breeds and wolf subspecies) would change the published results. Another objective was to examine OA in archaeological dog skulls and compare these results to modern dogs and to their progenitor, the wolf. We were also interested in testing whether OA could be measured on 3D-CT scan images to provide results consistent with normal measuring results. If so, this would allow future scientists to examine digital images instead of actual skulls that are valuable and fragile. Ultimately, we hoped to explain what anatomical landmarks are responsible for a narrower OA in wolves and to determine the possible value of this morphological method for further use.
Materials and methods
Materials
Modern dog skulls
A total of 384 dog skulls, belonging to 71 breeds and 5 crossbreed dogs, were measured. These belong to the collection of the Department of Anatomy, Faculty of Veterinary Medicine Ghent, University Ghent, Belgium (123 skulls) and the collection of the Natural History Museum Bern, Switzerland (NMBE) (261 skulls) (Table 1).
Archaeological dog skulls
Forty-five skulls were measured. Forty-three were from the ZMK_ZMUC collection of the Centre for GeoGenetics, Natural History Museum of Denmark, University of Copenhagen, Denmark. Most have not been dated by AMS C14, but rather by stratigraphy and associated artefacts. Most are attributed to the Boreal and Atlantic (Mesolithic) phase, while a few are of Neolithic age. Additionally, one Mesolithic skull was from the collection of LVR-Landes Museum, Bonn, Germany, and one from the Antikvarie collection, Osteology, Lund Universitets Historiska Museum, Lund, Sweden (Table 2).
Recent wolf skulls
In total 55 skulls were measured. Thirty-eight (32 Canis lupus pallipes and 6 C.l. arabs) are from the collection of the George S. Wise Faculty of Life Sciences, Department of Zoology at Tel-Aviv University, Israel (ZMTAU) and seven (1 Canis lupus pallipes and 6 C.l. arabs) from the Natural History Museum, London, Great Britain (BMNH). Ten Eurasian skulls are from the Natural History Museum, Bern, Switzerland (NMBE) (Table 3).
Methods
Mandible-free skulls were positioned in a horizontal plane with the midline aligned on the centre of the photographic lens. The skull was positioned as symmetrically as possible, but minor asymmetries <1° could not be excluded and not all skulls were anatomically perfectly symmetrical. Digital pictures were taken with a Nikon 700 digital camera with a stable objective off 50 mm focal lenght. The proximal tip of the lens was positioned at a distance of 40–50 cm from the most rostral point of the skull. Images were saved in RAW format and imported to the Dicom viewer of OsiriX MD software to measure OA. Software measurements were 0.001° but rounded off to a full degree. First, a horizontal line was drawn on top of the frontal bones as the first leg of the angle (Fig. 2). The oblique leg was drawn between ZP dorsally and ventrally the most lateral of two points: ZP or ZA (as described above) (Figs. 2, 3). Measurements were performed bilaterally and differences between sides were used in the symmetry study. Measuring intra- and inter-reliability was performed on 50 dog skulls each (100 measures) selected randomly and blindly, without knowledge of the results of the first measurements. Intra-reliability was performed by the second author (IS) and all other measurements by the first (LJ).
We opted in our study to use the plane technique and realised that this meant that in some cases ZA was used while in others FP was used as the ventral contact point of the oblique leg of the angle.
We considered that using the two different measuring methods could create a problem that would bias the results and hamper comparisons between groups. To address this issue, the percentage in which the oblique angle leg was in contact with ZP or ZA was calculated using 75 randomly selected skulls (25 from each group) of wolves, recent and archaeological dogs. The OA was measured in the standard way on both sides, and the measurement was then repeated using the ZP-FP contact points. The difference in OA was calculated between methods. The influence of imperfect skull position was measured in 5 dog skulls chosen randomly. OA was measured with a horizontal and symmetrical skull, with a 5° tilt upwards or downwards of the nose and with a 5° rotational deviation (right side forward). Asymmetry was measured in each individual skull and per group.
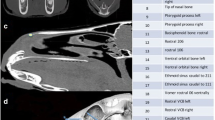
Twelve skulls of the ZMTAU collection were scanned with a single slice Picker CT scan. Transversal 1-mm-thick slices were recorded. The DICOM images were imported into Osirix MD to create a 3D reconstruction (Fig. 4). The printout of this image was used to measure the OA with an orthopaedic goniometer.
ANOVA models, with individual as random effect, were used to test for statistical differences between groups, where the denominator degrees of freedom of the F test were approximated using the Satterthwaite method. Significance level was set at 0.01. To test reliability, individual was modelled as a random effect, which allowed us to measure inter-individual variation and intra-observer reliability.
Results
The mean OA in modern dogs was 55° (SD 5.8°, minimum 41°, maximum 72°). In archaeological dogs it was 47° (SD 4.7°, minimum 35°, maximum 60°) and 42° in wolves (SD 5.3°; minimum 28°, maximum 52°) (Fig. 5) (Table 4). These values were significantly different between recent and archaeological dogs (p < 0.0001), modern dogs and wolves (p < 0.0001), and wolves and archaeological dogs (p = 0.01).
The overlap between modern dogs and wolves ranged between 41° and 52°. In this range were 43 % of measurements of modern dogs and 61 % of wolves. The overlap zone of all dogs and wolves ranged between 35° and 60° and was present in 48 % of the dogs and 91 % of the wolves. The percentage of wolves that had an OA lower than 35° was 9 %. The percentage of recent dogs that had an OA larger than 52° was 56 % (Table 4).
Asymmetry in OA dog skulls was 1.7° (SD 1.8°) (p < 0.1), in wolves 1.2° (SD 1.3°) (p < 0.1) and in archaeological dog skulls 2.3° (SD 1.8°) (p < 0.1). Fluctuating asymmetry was significant between wolves and both dog groups (F 2,467 = 5.07, p = 0.007) but not between archaeological and modern dogs (p = 0.17). Asymmetry was larger in archaeological dogs (mean 2.29°, SD 0.27) compared to modern dogs (mean 1.69°, SD 0.08, p = 0.04) and wolves (mean 1.23°, SD 0.20, p = 0.005). The inter-observer reliability was 0.99. The mean difference between the two researchers was between 0° and 2°. The intra-observer reliability was 0.97. The mean difference between the two measuring sessions was 1.2° with a range of 0°–7°.
The oblique leg of the angle was in contact with ZA as the ventral point in 92/100 wolves, 90/100 archaeological dog and 64/100 modern dog measurements. The difference between wolves and recent dogs was statistically significant (χ 2 = 35.7, d.f. = 1, p < 0.0001), but no difference was observed between wolves and archaeological dogs (χ 2 = 0.06, d.f. = 1, p = 0.80). The OA, measured in the subgroups with ZA as the ventral contact point, was 42° in wolves, 47° in archaeological dogs and 53° in modern dogs. Re-measuring the same skulls with FP as the ventral contact point resulted in an OA of 47° in wolves (gain 5°), 50° in archaeological dogs (gain 3°) and 59° in modern dogs (gain 4°). This gain in OA was statistically significant in the three groups (F 1,25 = 220, p < 0.0001). The OA difference in the nose up position was 0.3° (p = 0.85) and nose down position 2.7° (p = 0.09). When rotating the skull position the OA decreased at the proximal side −1.1° (p = 0.56) and enlarged at the caudal side 7.3° (p = 0.0005).
The difference between measuring the same skulls with the standard method or in 3D CT scan images was 3° (0°–8°), a difference that was statistically significant (F 1,27 = 26.0, p < 0.0001). Nevertheless, there was a positive correlation between individuals measured with both methods (r = 0.67).
Asymmetry in OA dog skulls was 1.7° (SD 1.8°) (p < 0.1), in wolves 1.2° (SD 1.3°) (p < 0.1) and in archaeological dog skulls 2.3° (SD 1.8°) (p < 0.1). Fluctuating asymmetry was significant between wolves and both dog groups (F 2,467 = 5.07, p = 0.007) but not between archaeological and modern dogs (p = 0.17). Asymmetry was larger in archaeological dogs (mean 2.29°, SD 0.27) compared to modern dogs (mean 1.69°, SD 0.08, p = 0.04) and wolves (mean 1.23°, SD 0.20, p = 0.005). The inter-observer reliability was 0.99. The mean difference between the two researchers was between 0° and 2°. The intra-observer reliability was 0.97. The mean difference between the two measuring sessions was 1.2° with a range of 0°–7°.
The oblique leg of the angle was in contact with ZA as the ventral point in 92/100 wolves, 90/100 archaeological dog and 64/100 modern dog measurements. The difference between wolves and recent dogs was statistically significant (χ 2 = 35.7, d.f. = 1, p < 0.0001), but no difference was observed between wolves and archaeological dogs (χ 2 = 0.06, d.f. = 1, p = 0.80). The OA, measured in the subgroups with ZA as the ventral contact point, was 42° in wolves, 47° in archaeological dogs and 53° in modern dogs. Re-measuring the same skulls with FP as the ventral contact point resulted in an OA of 47° in wolves (gain 5°), 50° in archaeological dogs (gain 3°) and 59° in modern dogs (gain 4°). This gain in OA was statistically significant in the three groups (F 1,25 = 220, p < 0.0001). The OA difference in the nose up position was 0.3° (p = 0.85) and nose down position 2.7° (p = 0.09). When rotating the skull position the OA decreased at the proximal side −1.1° (p = 0.56) and enlarged at the caudal side 7.3° (p = 0.0005). The difference between measuring the same skulls with the standard method or in 3D CT scan images was 3° (0°–8°), a difference that was statistically significant (F 1,27 = 26.0, p < 0.0001). Nevertheless, there was a positive correlation between individuals measured with both methods (r = 0.67).
Discussion
As the range in our study was 44°, measurements were rounded off to 1°, based on the statistical 30–300 rule (Heath 2002). Reliabilities were also very high with a 1°–2° difference in measuring, so there was no need to apply more precise measurement.
Our study demonstrates that archaeological dogs have OA values between those of dogs and wolves and are closer to wolves than modern dogs. In addition, the maximal OA in archaeological dogs lies closer to that of wolves: 50°–60°, not modern dogs (72°) (Table 4). Such OA values of archaeological dogs fit nicely into the evolutionary pathway, with wolves (mean OA 42°) as the progenitor to archaeological dogs (mean OA 47°) and the latter as the forefathers of modern breeds (mean OA 55°). However, things may be more complex. Indeed, most archaeological dog skulls were from a single geographical area (Scandinavia), and thus this group was probably geographically isolated for a long time and therefore closely related and morphologically similar. The OA of these archaeological dogs may thus not represent an evolutionary stage, but rather a genetically isolated population which had a coincidental OA close to wolves.
An interesting finding in this field comes from the study of Aaris-Sørensen (1977) who measured a statistically different OA in archaeological versus recent wolf skulls (resp. 44° and 41°) (t = 2.76, d.f. = 33, p = 0.01). This finding is contrary to the finding in dogs in our study, where more recent specimens have a higher OA. It might be that the difference in the study of Aaris-Sørensen (1977) is coincidental, but could, however, also be genetic or climate driven. If climate driven, it is difficult to explain why the OA in dogs and wolves would evolve following contrary paths. In our study, the OA dispersion is 28°–72° (44°) which is considerably more than the 16° scatter range published in earlier publications (Aaris-Sørensen 1977; Bockelmann 1920; Iljin 1941; Studer 1901). Further, the OA overlap between modern dogs and wolves is larger in our study than reported in the Aaris-Sørensen (1977) study (11° vs. 5°). This difference is probably caused by the larger group of skulls examined, as well as by the larger variety in wolf subspecies and dog breeds.
It is quite important to realise that if only recent wolf and dog skulls would have been studied, the overlap would be about half, and thus about 50 % of skulls could with certainty have been assigned to one of both groups: wolves to the group with OA between 28° and 42° and recent dogs to the group between 42° and 72°. Adding the archaeological dog group narrowed the percentage of wolf skulls that could be separated from archaeological and recent dog skulls to 9 % a small amount but still applicable.
It could be that the percentage to discern archaeological wolves from archaeological dogs by measuring OA is larger, given that archaeological wolves in the study of Aaris-Sørensen (1977) had a larger OA than recent wolves (44° vs. 41°). To be certain of this statement one needs, however, more archaeological wolf skulls to be studied and also more geographical areas. Such archaeological samples should ideally be obtained from archaeological sites from 18,000 to 16,000 calBP across Europe and the near East (thus the Magdalenian across its range in Europe as well as the Epipaleolithic and Natufian in Turkey and the near East) to complement the more recent specimens from Mesolithic and Neolithic contexts, which should be supplemented as well by specimens in different regions. It would be useful as well to have wolf specimens from non-archaeological contexts for this period to compare wolf with those specimens in direct contact with human populations.
Interestingly, the two putative archaeological dog skulls from Eliseevichi (Sablin and Khlopachev 2002) had an OA of 47°, which is in the non-diagnostic overlap zone, but the archaeological Predmosti 1, OK 1062 specimen (Germonpré et al. 2012) which could not be classified in the original study had an OA of 32° (Germonpré et al. 2012, Figure 6, p. 191), thus very close to the lowest OA of wolves. Here this additional measure (the OA) could convincingly place this last specimen in the wolf group.
The range of OA in modern dog breeds is the largest of the three groups (42°–72°). It is tempting to believe that the great anatomical variability of modern dog breeds is the cause of this OA range and these very high angles. However, the highest OA measured in our study was in a Border collie. This is at least puzzling as this is a mesaticephalic, small shepherd breed with a very “normal” morphology on view, much like a small German shepherd, the breed so favoured by previous authors examining the OA, as it was assumed to be a primitive type breed, still very like wolves.
The highest amount of asymmetry was seen in archaeological dogs followed by modern dogs. Wolves were the most symmetrical. These findings fit nicely in the evolutionary development of the three groups with wolves being a long existing species and dogs having split from wolves about 18,000 years ago (Thalmann et al. 2013). Wolves should thus logically have the most symmetrical OA and archaeological dogs the least as this was “the new species” close in time to the source of its origin. As our archaeological dog skulls were estimated to be on average 6–7000 years old, they had split from wolves for about 11–12,000 years, while the modern dogs we examined had genetically split off from wolves 18,000 years ago, and thus had over a longer period of time to develop into a new species, and thus develop more symmetry. An argument to support this view is that the oldest specimen in our series, the Bedburg dog, had a very high asymmetry (4°), more than the mean of archaeological dogs (mean 2.29°, SD 0.27) (Table 2).
The discovery that Studer (1901) presented two different methods to measure the OA was unexpected. Re-measuring the skulls with the FP contact point resulted in a higher OA in the three groups (wolves +5°, archaeological dogs +3°, recent dogs +4°), and thus there was hardly any difference in discriminating the group means. This difference is not statistical (F 3,46 = 0.90, p = 0.45), and thus both groups can be separated as well with the corrected method.
Positional differences had little influence on OA outcome. Only rotational asymmetry created a statistically different angle and only at the caudally translated side. These rotations were, however, extreme and incomparable with minimal asymmetries of some skulls examined in our series.
Why the OA widens can be explained mathematically by a lateral and/or downward shift of ZA (or FP) or an upward and/or inward shift of ZP (Fig. 2). One of these shifts or combinations could be possible. Studer (1901) carefully studied the question of wider OA in dogs and found that in equally large skulls of dog and wolf, with a 12° difference in OA, the total width of skulls measured at the widest point of the zygomatic arch (zygon–zygon) (measure 30) was identical (Von Den Driesch 1976). Therefore the lateral shift of ZA (or FP) could be excluded as the reason of a change in OA. Studer (1901) then realised that dog skulls had a wider ectorbitale–ectorbitale width (measure 32) (Von Den Driesch 1976) and thus a lateralisation of FP. The same conclusion, regarding the difference between wolves and modern dogs, came from the study of Aaris-Sørensen (1977). Moreover, he measured a 10 % increase in measure 32 in archaeological wolves when compared to recent wolves. That explained the larger OA he measured in these archaeological wolves (as described above).
Three recent studies report on anatomical changes between skulls of wolves and dogs. One study describes the enlarged orbital region (and thus FP lateralisation) in dogs when compared to wolves (Schmitt and Wallace 2012). A geometric morphometrics study in mesaticephalic dogs describes maxillar skull widening at the M1 level, which is at the rostral region of the orbital region (and thus FP lateralisation). Finally, a new study compared skulls of recent and archaeological dogs and wolves (Drake et al. 2015). In dogs, changes were shortening and angulation of the snout, rostral and upward movement of frontals (stop formation) and widening of the orbital region (and thus FP lateralisation). These recent studies confirm Studer’s (1901) conclusions from more than 100 years ago: the frontal process (FP) shifts to lateral and thus the OA widens.
To conclude, this paper examined five main aspects for the use of OA as a morphological method to distinguish between wolves and dogs: (1) methodological issues, (2) increased sample size, (3) use of 3D CT scan images, (4) identification of the anatomical landmarks responsible for a narrower OA in wolves and (5) evaluation of the potential value of this method. First, the use of FP instead of ZA as the ventral contact point results in statistically significant differences, with a gain in degrees in all three groups, the method is very reliable with respect to intra- and inter-reliability, symmetry is highest in wolves and lowest in dogs and skull position changes have no significant effect on the angle measured. Second, increased sample size showed a broader OA dispersion and a greater overlap between modern dogs and wolves than reported in other studies. Third, OA can be measured on 3D CT scans but cannot be compared with direct measuring results on actual skulls. Fourth, the wider OA in dogs is due to lateralisation of the zygomatic process of the frontal bone. Fifth, our conclusion is that the OA can be used as an additional morphological measuring method. Although its value to separate dog and wolf skulls is restricted to extreme values, a considerable amount of skulls fall into these categories.
In the future a wider geographic range of archaeological (pre- and post-LGM) and recent dogs ought to be measured, to confirm that archaeological dogs elsewhere are intermediate between wolves (ancient and recent) and modern dog breeds. This might be one aspect that helps to resolve recent claims that Gravettian canids are semi-domesticated “wolf-dogs”.
References
Aaris-Sørensen K (1977) The subfossil wolf, Canis lupus L. in Demark. Vidensk Meddr dabsk naturhistorisch Forensen 29:129–146
Aaris-Sørensen K (2004) Med Hunden i Fokus en metod att identifiera hundars användningsområde utifrån det postkraniala skelettet. Vidensk Medd fra Dansk naturh Foren Lund Institutionen för arkeologi och antikens historia 140:129–146
Benecke N (1987) Studies on early dog remains from Northern Europe. J Archaeol Sci 14:31–49
Benecke N (1994) Archäozoologische Studien zur Entwicklung der Haustierhaltung in Mitteleuropa und S¨dskandinavien von den Anfängen bis zum ausgehenden Mittelalter. Schriften zur Ur- und Frühgeschichte Bd 46 Akademie-Verlag Berlin
Bockelmann H (1920) Untersuchungen an Wolfsbastarden nach Ziichtungsversuchen im Haustiergarten zu Halle. Dissertation, Universit Halle
Boudadi-Maligne M, Escarguel G (2014) A biometric re-evaluation of recent claims for Early Upper Palaeolithic wolf domestication in Eurasia. J Archaeol Sci 45:80–89
Boyko AR et al (2010) A simple genetic architecture underlies morphological variation in dogs. PLoS Biol 8:e1000451
Clutton-Brock J (1962) Near Eastern canids and the affinities of the Natufian dogs. Zeitschrift für Tierzüchtung und Züchtungsbiologie 76:326–333
De Coster G, Van Dongen S, Malaki P, Muchane M, Alcántara-Exposito A, Matheve H, Lens L (2013) Fluctuating asymmetry and environmental stress: understanding the role of trait history. PLoS One 8:e57966
Degerbøl M (1961) Der Hund, das älteste Haustier Dänemarks. Zeitschrift für Tierzüchtung und Züchtungsbiologie 76:334–341
Drake AG, Coquerelle M, Colombeau G (2015) 3D morphometric analysis of fossil canid skulls contradicts the suggested domestication of dogs during the late Paleolithic. Sci Rep 5. doi:10.1038/srep08299
Druzhkova AS et al (2013) Ancient DNA analysis affirms the canid from Altai as a primitive dog. PLoS One 8:e57754
Freedman AH et al (2014) Genome sequencing highlights the dynamic early history of dogs. PLoS Genet 10:e1004016
Germonpré M, Lázničková-Galetová M, Sablin MV (2012) Palaeolithic dog skulls at the Gravettian Předmostí site, the Czech Republic. J Archaeol Sci 39:184–202
Gundry RL, Allard MW, Moretti TR, Honeycutt RL, Wilson MR, Monson KL, Foran DR (2007) Mitochondrial DNA analysis of the domestic dog: control region variation within and among breeds. J Forensic Sci 52:562–572
Heath D (2002) An introduction to experimental design and statistics for biology. CRC Press, Boca Raton
Huxley TH (1880) On the cranial and dental characters of the Canidæ. In: Proceedings of the zoological society of London. Wiley Online Library, pp 238–288
Iljin NA (1941) Wolf-dog genetics. J Genet 42:359–414
Köhler M, Moyà-Solà S (2004) Reduction of brain and sense organs in the fossil insular bovid Myotragus. Brain Behav Evol 63:125–140
Larson G, Burger J (2013) A population genetics view of animal domestication. Trends Genet 29:197–205
Larson G et al (2012) Rethinking dog domestication by integrating genetics, archeology, and biogeography. Proc Natl Acad Sci 109:8878–8883
Leonard JA, Wayne RK, Wheeler J, Valadez R, Guillén S, Vila C (2002) Ancient DNA evidence for Old World origin of New World dogs. Science 298:1613–1616
Morey DF (1986) Studies on Amerindian dogs: taxonomic analysis of canid crania from the Northern Plains. J Archaeol Sci 13:119–145
Morey DF (1992) Size, shape and development in the evolution of the domestic dog. J Archaeol Sci 19:181–204
Morey DF (2014) In search of paleolithic dogs: a quest with mixed results. J Archaeol Sci 52:300–307
Natanaelsson C, Oskarsson MC, Angleby H, Lundeberg J, Kirkness E, Savolainen P (2006) Dog Y chromosomal DNA sequence: identification, sequencing and SNP discovery. BMC Genet 7:45
Nehring A (1888) Zur Abstammung der Hunde-Rassen. Zoologische Jahrbücher Abtheilung Systematik, Geographie und Biologie 3:51–58
Olsen SJ, Olsen JW (1977) The Chinese wolf, ancestor of New World dogs. Science 197:533–535
Pionnier-Capitan M (2010) La domestication du chien en Eurasie: étude de la diversité passée, approches ostéoarchéologiques, morphométriques et paléogénétiques. Lyon, Disseration
Rütimeyer L (1861) Die Fauna der Pfahlbauten der Schweiz. Geschichte der Wilden und der Haus-Saugetiere. Neue Denkschrift der Algemeinne Schweizerische Geselschaft der ges. Naturwissenschaft, 19, Basel, Switzerland
Rütimeyer L (1875) Die Knochenhöhle von Thayingen bei Schaffhausen. F. Vieweg & Sohn, Braunschweig
Sablin M, Khlopachev G (2002) The earliest ice age dogs: evidence from Eliseevichi 11. Curr Anthropol 43:795–799
Savolainen P, Y-p Zhang, Luo J, Lundeberg J, Leitner T (2002) Genetic evidence for an East Asian origin of domestic dogs. Science 298:1610–1613
Schmitt E, Wallace S (2012) Shape change and variation in the cranial morphology of wild canids (Canis lupus, Canis latrans, Canis rufus) compared to domestic dogs (Canis familiaris) using geometric morphometrics. Int J Osteoarchaeol 24:42–50. doi:10.1002/oa.1306
Stockhaus K (1965) Metrische Untersuchungen an Schädeln von Wölfen und Hunden. J Zool Syst Evol Res 3:157–258
Studer T (1901) Die prähistorischen Hunde in ihrer Beziehung zu den gegenwärtig lebenden Rassen. Zurcher und Furrer, Zurich, pp 1–154
Sumiński P (1975) Morphologische Unterscheidungsmerkmale zwischen Wolfs-(Canis lupus L.) und Hundeschädel (Canis familiaris L.). Zeitschrift für Jagdwissenschaft 21:227–232
Thalmann O et al (2013) Complete mitochondrial genomes of ancient canids suggest a European origin of domestic dogs. Science 342:871–874
Van Dongen S (2006) Fluctuating asymmetry and developmental instability in evolutionary biology: past, present and future. J Evol Biol 19:1727–1743
Van Dongen S, Lens L, Pape E, Volckaert F, Raeymaekers J (2009) Evolutionary history shapes the association between developmental instability and population-level genetic variation in three-spined sticklebacks. J Evol Biol 22:1695–1707
Von Den Driesch A (1976) A guide to the measurement of animal bones from archaeological sites: as developed by the Institut für Palaeoanatomie, Domestikationsforschung und Geschichte der Tiermedizin of the University of Munich. Cambridge, Peabody Museum Press
Vonholdt B et al (2010) Genome-wide SNP and haplotype analyses reveal a rich history underlying dog domestication. Nature 464:898–902
Wayne RK (1986) Cranial morphology of domestic and wild canids: the influence of development on morphological change. Evolution 40:243–261
Wolfgram A (1894) Die Einwerkung der Gefangenschaft auf die Gestaltung des Wolfschädels. Zoologisches Jahrbuch (Abteilung fur Systematik) 7:773–822
Acknowledgments
For help with the skull collections and permission to study these we thank Daniel Berkowic for the Israel wolf collection, Roberto Portela Miguez from the Natural History Museum in London, Paul Schmid and Marc Nussbaumer from Bern, Kristian Murphy Gregerson from Copenhagen and Paul Simoens from Ghent. Ron Shahar, from the Koret School of Veterinary Medicine, The Robert H. Smith Faculty of Agriculture, Food and Environment, The Hebrew University of Jerusalem, Rehovot, Israel, is also thanked for allowing us to perform the wolf CT scans. Erika Rosengren from Lund University and Ralf Schmitz from Bonn are also thanked for allowing us to examine specimen. Peter Vandekerckhove is thanked for help with the Osirix program, Christien de Paepe for layout and the reviewers for advice.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by A. Schmidt-Rhaesa.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Janssens, L., Spanoghe, I., Miller, R. et al. Can orbital angle morphology distinguish dogs from wolves?. Zoomorphology 135, 149–158 (2016). https://doi.org/10.1007/s00435-015-0294-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00435-015-0294-3