Abstract
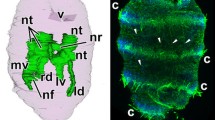
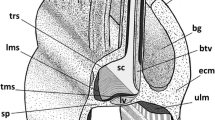
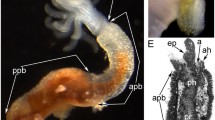
Results provided by modern TEM methods indicate the existence of the lophophoral and trunk coelomes but not of the preoral coelom in Phoronida. In the present work, the epistome in Phoronopsis harmeri was studied by histological and ultrastructural methods. Two kinds of cells were found in the frontal epidermis: supporting and glandular. The coelomic compartment is shown to be inside the epistome. This compartment has a complex shape, consists of a central part and two lateral branches, and contacts the lophophoral coelom, forming two complete dissepiments on the lateral sides and a partition with many holes in the center. TEM reveals that some portions of the incomplete partition are organized like a mesentery, with the two layers of cells separated by ECM. The myoepithelial cells of the coelomic lining form the circular and radial musculature of the epistome. Numerous amoebocytes occur in the coelom lumen. The tip of the epistome and its dorso-lateral parts lack a coelomic cavity and are occupied by ECM and muscle cells. The fine structure of the T-shaped vessel is described, and its localization inside lophophoral coelom is demonstrated. We assert that the cavity inside the epistome is the preoral coelom corresponding to the true preoral coelom of the larva of this species. Proving this assertion will require additional study of metamorphosis in this species. To clarify the patterns of coelom organization in phoronids, we discuss the bipartite coelomic system in Phoronis and the tripartite coelomic system in Phoronopsis.











Similar content being viewed by others
References
Bartolomaeus T (2001) Ultrastructure and formation of the body cavity lining in Phoronis muelleri (Phoronida, Lophophorata). Zoomorph 120(3):135–148
Benito J, Pardos F (1997) Hemichordata. In: Harrison FW, Ruppert EE (eds) Microscopic anatomy of Invertebrates. Vol. 15. Hemichordata, chaetognata, and the invertebrate chordates. Wiley, New York, pp 15–102
Chipman AD (2010) Parallel evolution of segmentation by co-option of ancestral gene regulatory networks. BioEssays 32:60–70. doi:10.1002/bies.200900130
Couso J (2009) Segmentation, metamerism and the Cambrian explosion. Int J Dev Biol 53:1305–1316. doi:10.1387/ijdb.072425jc
Cowles RP (1904) Origin and fate of the body-cavities and the nephridia of the Actinotrocha. Ann Mag Nat Hist 14:69–78
Dunn CW, Hejnol A, Matus DQ, Pang K, Browne WE, Smith SA, Seaver E, Rouse GW, Obst M, Edgecombe GD, Sørensen MV, Haddock SHD, Schmidt-Rhaesa A, Okusu A, Kristensen RM, Wheeler WC, Martindale MQ, Giribet G (2008) Broad phylogenomic sampling improves resolution of the animal tree of life. Nature 452:745–749. doi:10.1038/nature06614
Emig CC (1977) Sur la structure des parois de l’appareil circulatore de Phoronis psammophila Cori (Phoronida, Lophophorata). Zoomorphol 87:147–153
Emig CC, Siewing R (1975) The epistome of Phoronis psammophila (Phoronida). Zool Anz 194(1/2):47–54
Grobe P (2008) http://www.diss.fu-berlin.de/2008/81/chapter2.pdf. Accepted 30may 2010
Gruhl A, Grobe P, Bartolomaeus T (2005) Fine structure of the epistome in Phoronis ovalis: significance for the coelomic organization in Phoronida. Invert Biol 124(4):332–343
Gruhl A, Wegener I, Bartolomaeus T (2009) Ultrastructure of the body cavities in Phylactolaemata (Bryozoa). J Morphol 270(3):306–318
Halanych KM, Bacheller JD, Aguinaldo AM, Liva SM, Hillis DM (1995) Evidence from 18S ribosomal DNA that the lophophorates are protostome animals. Science 267:1641–1643
Hejnol A, Obst M, Stamatakis A, Ott M, Rouse GW, Edgecombe GD, Martinez P, Baguñà J, Bailly X, Jondelius U, Wiens M, Müller WEG, Seaver E, Wheeler WC, Martindale MQ, Giribet G, Dunn CW (2009) Assessing the root of bilaterian animals with scalable phylogenomic methods. Proc Biol Sci 276:4261–4270
Herrmann K (1980) The regionation of Phoronis muelleri (Tentaculata). Zool Jahrb Anat 103(2):234–249
Herrmann K (1986) Ontogenesis of Phoronis muelleri (Tentaculata) with a special sight for differentiation of mesoderm and phylogenesis of coelom. Zool Jahrb Anat 114(4):441–463
Hyman LH (1959) Phoronida. In: Boell EJ (ed) The invertebrates. V. 5. Smaller coelomate groups. McGraw-Hill, New York, pp 228–274
Menon KR (1902) Notes of Actinotrocha. Q J Microsc Sci 45:473–484
Paps J, Baguñà J, Riutort M (2010) Lophotrochozoa internal phylogeny: new insights from an up-to-date analysis of nuclear ribosomal genes. Proc R Soc 276:1245–1254. doi:10.1098/rspb.2008.1574
Peterson KJ, Eernisse DJ (2001) Animal phylogeny and the ancestry of bilaterians: inferences from morphology and 18S rDNA gene sequences. Evol Dev 3:170–205
Pross A (1974) Untersuchugen uber die Muskulatur des Epistoms der Phoroniden (Phoronis ijimai Oka [Phoronidea]). Zool Jahrb Anat 92:391–403
Pross A (1978) Motion of epistom in Phoronis ijimai (Phoronida). Zool Jahrb Anat 99:54–58
Selys-Longchamps M (1907) Phoronis. Fauna und Flora des Golfes von Neapel. Monogr 30:280
Siewing R (1974) Morphologische Untersuchungen zum Archicoelomatenproblem. The body segmentation in Phoronis muelleri de Selys-Longchamps (Phoronidea) ontogenese—larve—metamorphose—adultus. Zool Jahrb Anat 92(2):275–318
Siewing R (1980) Das Archicoelomatenkonzept. Zool Jahrb Anat Ont 103:439–482
Temereva EN (2006) Trimeric organization of the larval coelom in phoronids. Abst VIIth Int Larv Biol Symp, p 42
Temereva EN (2009) New data on distribution, morphology and taxonomy of phoronid larvae (Phoronida, Lophophorata). Invert Zool 6(1):1–25
Temereva EN (2010) The digestive tract of actinotroch larvae (Lophotrochozoa, Phoronida): anatomy, ultrastructure, innervations, and some observations of metamorphosis. Can J Zool 88(12):1149–1168
Temereva EN, Malakhov VV (2004) Ultrastructure of the blood system in Phoronid Phoronopsis harmeri Pixell, 1912: II. Main vessels. Russ J Mar Biol 30(2):101–112
Temereva EN, Malakhov VV (2006) The answer to Thomas Bartolomaeus: “Larva of phoronid Phoronopsis harmeri Pixell, 1912 has trimeric coelom organization”. Invert Zool 2(2):394–402
Temereva EN, Malakhov VV (2007) Embryogenesis and larval development of phoronid Phoronopsis harmeri Pixell, 1912: dual origin of the coelomic mesoderm. Invert Reprod Dev 50(2):57–66
Temereva EN, Malakhov VV (2010) Method of ciliary filter feeding in Phoronopsis harmeri (Phoronida, Lophophorata). Russ J Mar Biol 36(2):109–116
Vandermeulen JH (1970) Functional morphology of the digestive tract epithelium in Phoronis vancouverensis Pixell: an ultrastructural and histological study. J Morphol 130:271–286
Vandermeulen JH, Reid RG (1969) Digestive tract enzymes in Phoronida. Comput Biochem Phys 28:443–448
Zimmer RL (1978) The comparative structure of the preoral hood coelom in Phoronida and the fate of this cavity during and metamorphosis. In: Chia FS, Rice ME (eds) Settlement and metamorphosis of marine invertebrate larvae. Elsevier, New York, pp 23–40
Zimmer RL (1980) Mesoderm proliferation and formation of the protocoel and metacoel in early embryos of Phoronis vancouverensis (Phoronida). Zool Jahrb Anat 103(2):219–233
Acknowledgments
ENT is grateful to Svetlana Maslakova for hosting her at the Oregon Institute of Marine Biology (Coos Bay) in 2010. We appreciate an anonymous reviewer for his criticisms and improving of manuscript. We are most grateful to Dr. Mikhailov, Dr. Jaffee for the help with manuscript preparation. The project was funded by the Russian Foundation for Basic Research (project 11-04-00690), Russian Ministry of Education and Science and Grant Council of the President of Russia (#4456.2010.4, #02.740.11.0280, #P727, # MD-2892.2011.4).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by T. Bartolomaeus.
Rights and permissions
About this article
Cite this article
Temereva, E.N., Malakhov, V.V. Organization of the epistome in Phoronopsis harmeri (Phoronida) and consideration of the coelomic organization in Phoronida. Zoomorphology 130, 121–134 (2011). https://doi.org/10.1007/s00435-011-0126-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00435-011-0126-z