Abstract
Background
Synchronous multiple primary colorectal cancer (SMPCC) is a rare subtype of CRC, characterized by the presence of two or more primary CRC lesions simultaneously or within 6 months from the detection of the first lesion. We aim to develop a novel nomogram to predict OS and CSS for SMPCC patients using data from the SEER database.
Methods
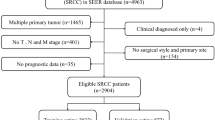
The clinical variables and survival data of SMPCC patients between 2004 and 2018 were retrieved from the Surveillance, Epidemiology, and End Results (SEER) database. Appropriate inclusion and exclusion criteria were established to screen the enrolled patients. Univariate and multivariate Cox regression analyses were used to identify the independent risk factors for OS and CSS. The performance of the nomogram was evaluated using the concordance index (C-index), calibration curves, and the area under the curve (AUC) of a receiver operating characteristics curve (ROC). A decision curve analysis (DCA) was generated to compare the net benefits of the nomogram with those of the TNM staging system.
Results
A total of 6772 SMPCC patients were enrolled in the study and randomly assigned to the training (n = 4670) and validation (n = 2002) cohorts. Multivariate Cox analysis confirmed that race, marital status, age, histology, tumor position, T stage, N stage, M stage, chemotherapy, and the number of dissected LNs were independent prognostic factors.The C-index values for OS and CSS prediction were 0.716 (95% CI 0.705–0.727) and 0.718 (95% CI 0.702–0.734) in the training cohort, and 0.760 (95% CI 0.747–0.773) and 0.749 (95% CI 0.728–0.769) in the validation cohort. The ROC and calibration curves indicated that the model had good stability and reliability. Decision curve analysis revealed that the nomograms provided a more significant clinical net benefit than the TNM staging system.
Conclusion
We developed a novel nomogram for clinicians to predict OS and CSS, which could be used to optimize the treatment in SMPCC patients.






Similar content being viewed by others
References
Arakawa K, Hata K, Nozawa H, Kawai K, Tanaka T, Nishikawa T et al (2018) Prognostic significance and clinicopathological features of synchronous colorectal cancer. Anticancer Res 38(10):5889–5895. https://doi.org/10.21873/anticanres.12932
Bos ACRK, Matthijsen RA, van Erning FN, van Oijen MGH, Rutten HJT, Lemmens VEPP (2018) Treatment and outcome of synchronous colorectal carcinomas: a nationwide study. Ann Surg Oncol 25(2):414–421. https://doi.org/10.1245/s10434-017-6255-y
Chen L,Yulin G, Guijin C,Yanbing J,Yu W,Zimeng L et al (2016) Clinical analysis of synchronous multiple primary colorectal carcinomas. Acad J Chin PLA Med Sch 37(07):735–738. https://kns.cnki.net/kcms/detail/11.3275.r.20160330.1041.004.html
Chin CC, Kuo YH, Chiang JM (2019) Synchronous colorectal carcinoma: predisposing factors and characteristics. Colorectal Dis 21(4):432–440. https://doi.org/10.1111/codi.14539
Dekker JW, Gooiker GA, Bastiaannet E, van den Broek CB, van der Geest LG, van de Velde CJ et al (2014) Cause of death the first year after curative colorectal cancer surgery; a prolonged impact of the surgery in elderly colorectal cancer patients. Eur J Surg Oncol 40(11):1481–1487. https://doi.org/10.1016/j.ejso.2014.05.010
Drew DA, Nishihara R, Lochhead P, Kuchiba A, Qian ZR, Mima K et al (2017) A prospective study of smoking and risk of synchronous colorectal cancers. Am J Gastroenterol 112(3):493–501. https://doi.org/10.1038/ajg.2016.589
Duraker N, Civelek Çaynak Z, Hot S (2014) The prognostic value of the number of lymph nodes removed in patients with node-negative colorectal cancer. Int J Surg (Lond, Engl) 12(12):1324–1327. https://doi.org/10.1016/j.ijsu.2014.10.038
Easson AM, Cotterchio M, Crosby JA, Sutherland H, Dale D, Aronson M et al (2002) A population-based study of the extent of surgical resection of potentially curable colon cancer. Ann Surg Oncol 9(4):380–387. https://doi.org/10.1007/BF02573873
Flor N, Zanchetta E, Di Leo G, Mezzanzanica M, Greco M, Carrafiello G et al (2018) Synchronous colorectal cancer using CT colonography vs other means: a systematic review and meta-analysis. Abdom Radiol (n y) 43(12):3241–3249. https://doi.org/10.1007/s00261-018-1658-1
Flor N, Ceretti AP, Luigiano C, Brambillasca P, Savoldi AP, Verrusio C et al (2020) Performance of CT colonography in diagnosis of synchronous colonic lesions in patients with occlusive colorectal cancer. AJR Am J Roentgenol 214(2):348–354. https://doi.org/10.2214/AJR.19.21810
He W, Zheng C, Wang Y, Dan J, Zhu M, Wei M, Wang J, Wang Z (2019) Prognosis of synchronous colorectal carcinoma compared to solitary colorectal carcinoma: a matched pair analysis. Eur J Gastroenterol Hepatol 31(12):1489–1495. https://doi.org/10.1097/MEG.0000000000001487
Holubar SD, Wolff BG, Poola VP, Soop M (2010) Multiple synchronous colonic anastomoses: are they safe? Colorectal Dis 12(2):135–140. https://doi.org/10.1111/j.1463-1318.2009.01771.x
Hu H, Chang DT, Nikiforova MN, Kuan SF, Pai RK (2013) Clinicopathologic features of synchronous colorectal carcinoma: a distinct subset arising from multiple sessile serrated adenomas and associated with high levels of microsatellite instability and favorable prognosis. Am J Surg Pathol 37(11):1660–1670. https://doi.org/10.1097/PAS.0b013e31829623b8
Khattak MA, Townsend AR, Beeke C, Karapetis CS, Luke C, Padbury R (2012) Impact of age on choice of chemotherapy and outcome in advanced colorectal cancer. Eur J Cancer (Oxf, Engl, 1990) 48(9):1293–1298. https://doi.org/10.1016/j.ejca.2011.09.029
Kotake K, Honjo S, Sugihara K, Hashiguchi Y, Kato T, Kodaira S et al (2012) Number of lymph nodes retrieved is an important determinant of survival of patients with stage II and stage III colorectal cancer. Jpn J Clin Oncol 42(1):29–35. https://doi.org/10.1093/jjco/hyr164
Lam AK, Carmichael R, Gertraud Buettner P, Gopalan V, Ho YH, Siu S (2011) Clinicopathological significance of synchronous carcinoma in colorectal cancer. Am J Surg 202(1):39–44. https://doi.org/10.1016/j.amjsurg.2010.05.012
Lam AK, Chan SS, Leung M (2014) Synchronous colorectal cancer: clinical, pathological and molecular implications. World J Gastroenterol 20(22):6815–6820. https://doi.org/10.3748/wjg.v20.i22.6815
Lindberg LJ, Wegen-Haitsma W, Ladelund S, Smith-Hansen L, Therkildsen C, Bernstein I et al (2019) Risk of multiple colorectal cancer development depends on age and subgroup in individuals with hereditary predisposition. Fam Cancer 18(2):183–191. https://doi.org/10.1007/s10689-018-0109-z
Liu X, Goldblum JR, Zhao Z, Landau M, Heald B, Pai R et al (2012) Distinct clinicohistologic features of inflammatory bowel disease-associated colorectal adenocarcinoma: in comparison with sporadic microsatellite-stable and Lynch syndrome-related colorectal adenocarcinoma. Am J Surg Pathol 36(8):1228–1233. https://doi.org/10.1097/PAS.0b013e318253645a
Mulder SA, Kranse R, Damhuis RA, de Wilt JH, Ouwendijk RJ, Kuipers EJ et al (2011) Prevalence and prognosis of synchronous colorectal cancer: a Dutch population-based study. Cancer Epidemiol 35(5):442–447. https://doi.org/10.1016/j.canep.2010.12.007
Ochiai K, Kawai K, Nozawa H, Sasaki K, Kaneko M, Murono K et al (2021) Prognostic impact and clinicopathological features of multiple colorectal cancers and extracolorectal malignancies: a nationwide retrospective study. Digestion 102(6):911–920. https://doi.org/10.1159/000517271
Oya M, Takahashi S, Okuyama T, Yamaguchi M, Ueda Y (2003) Synchronous colorectal carcinoma: clinico-pathological features and prognosis. Jpn J Clin Oncol 33(1):38–43. https://doi.org/10.1093/jjco/hyg010
Park SH, Lee JH, Lee SS, Kim JC, Yu CS, Kim HC et al (2012) CT colonography for detection and characterisation of synchronous proximal colonic lesions in patients with stenosing colorectal cancer. Gut 61(12):1716–1722. https://doi.org/10.1136/gutjnl-2011-301135
Renfro LA, Grothey A, Xue Y, Saltz LB, André T, Twelves C et al (2014) ACCENT- based web calculators to predict recurrence and overall survival in stage III colon cancer. J Natl Cancer Inst 106(12):dju333. https://doi.org/10.1093/jnci/dju333
Riegler G, Bossa F, Caserta L, Pera A, Tonelli F, Sturniolo GC et al (2003) Colorectal cancer and high grade dysplasia complicating ulcerative colitis in Italy. A retrospective co-operative IG-IBD study. Dig Liver Dis 35(9):628–634. https://doi.org/10.1016/s1590-8658(03)00380-3
Siegel RL, Miller KD, Fuchs HE, Jemal A (2022) Cancer statistics, 2022. CA Cancer J Clinicians 72(1):7–33. https://doi.org/10.3322/caac.21708
Van Leersum NJ, Aalbers AG, Snijders HS, Henneman D, Wouters MW, Tollenaar RA et al (2014) Synchronous colorectal carcinoma: a risk factor in colorectal cancer surgery. Dis Colon Rectum 57(4):460–466. https://doi.org/10.1097/DCR.0000000000000068
Weiser MR, Gönen M, Chou JF, Kattan MW, Schrag D (2011) Predicting survival after curative colectomy for cancer: individualizing colon cancer staging. J Clin Oncol 29(36):4796–4802. https://doi.org/10.1200/JCO.2011.36.5080
Wu A, He S, Li J, Liu L, Liu C, Wang Q, Peng X et al (2017) Colorectal cancer in cases of multiple primary cancers: clinical features of 59 cases and point mutation analyses. Oncol Lett 13(6):4720–4726. https://doi.org/10.3892/ol.2017.6097
Yang J, Peng JY, Chen W (2011) Synchronous colorectal cancers: a review of clinical features, diagnosis, treatment, and prognosis. Dig Surg 28(5–6):379–385. https://doi.org/10.1159/000334073
Yoon JW, Lee SH, Ahn BK, Baek SU (2008) Clinical characteristics of multiple primary colorectal cancers. Cancer Res Treat 40(2):71–74. https://doi.org/10.4143/crt.2008.40.2.71
Funding
No funding support.
Author information
Authors and Affiliations
Contributions
Conceptualization: XZ, BM. Data curation: XZ, CL, YH. Formal analysis: YH, JZ, WR. Writing—original draft: XZ, BM, KD. Writing—review and editing: KD, WR, BM.
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflicts of interest.
Data availability
The data that support the findings of this study are available from the Surveillance, Epidemiology, and End Results (SEER) database at http://www.seer.cancer.gov.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, X., Hu, Y., Deng, K. et al. Developing prognostic nomograms to predict overall survival and cancer-specific survival in synchronous multiple primary colorectal cancer based on the SEER database. J Cancer Res Clin Oncol 149, 14057–14070 (2023). https://doi.org/10.1007/s00432-023-05221-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-05221-z