Abstract
Background
Despite mammography-based screening for breast cancer has been conducted in many countries, there are still little data on participation and diagnostic yield in population-based breast cancer screening in China.
Methods
We enrolled 151,973 eligible women from four cities in Hebei Province within the period 2013–2021 and followed up until December 31, 2021. Participants aged 40–74 who assessed as high risk were invited to undergo breast ultrasound and mammography examination. Overall and group-specific participation rates were calculated. Multivariable analyses were used to estimate the factors associated with participation rates. The diagnostic yield of both screening and no screening groups was calculated. We further analyzed the stage distribution and molecular subtype of breast cancer cases by different modes of cancer detection.
Results
A total of 42,547 participants were evaluated to be high risk of breast cancer. Among them, 23,009 subjects undertook screening services, with participation rate of 54.08%. Multivariable logistic regression model showed that aged 45–64, high education level, postmenopausal, current smoking, alcohol consumption, family history of breast cancer, and benign breast disease were associated with increased participation of screening. After median follow-up of 3.79 years, there were 456 breast cancer diagnoses of which 65 were screen-detected breast cancers (SBCs), 27 were interval breast cancers (IBCs), 68 were no screening cancers, and 296 were cancers detected outside the screening program. Among them, 92 participants in the screening group (0.40%) and 364 in the non-screening group (0.28%) had breast cancer detected, which resulted in an odds ratio of 1.42 (95% CI 1.13–1.78; P = 0.003). We observed a higher detection rate of breast cancer in the screening group, with ORs of 2.42 (95% CI 1.72–3.41) for early stage (stages 0–I) and 2.12 (95% CI 1.26–3.54) for luminal A subtype. SBCs had higher proportion of early stage (71.93%) and luminal A subtype (47.22%) than other groups.
Conclusions
The significant differences in breast cancer diagnosis between the screening and non-screening group imply an urgent need for increased breast cancer awareness and early detection in China.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer is the most common malignancy and primary cause of cancer mortality among women globally, with an estimated 2.26 million new cases and 685,000 new deaths in 2020 (Sung and Siegel 2021). It is estimated that 420,000 breast cancer new cases were diagnosed and 120,000 deaths in China in 2020, accounting for 18.4 and 17.1% of all the world cases (Lei et al. 2021). Our understanding of breast cancer etiology and prognosis has improved over time, and the treatment outcome and survivorship can be improved through earlier detection.
Mammography screening has been demonstrated to reduce breast cancer mortality (Canelo-Aybar et al. 2022; Xie et al. 2022; Moss et al. 2015). Nevertheless, the participation remains low in China due to health check-up hesitancy toward cancer screening and low cancer awareness to make informed choices in the target population (Cao et al. 2021). A population-based study in China enrolled 313,022 high-risk individuals of breast cancer during the period of 2013–2017, showing that a participation rate was 40.3% (Chen et al. 2020). Previous studies have shown that participation rates differ significantly between different populations (Mottram et al. 2021; Wu et al. 2019). Additionally, cultural factors, the health care system's infrastructure, and economic concerns also influence the effectiveness of organized population-based screening (Williams et al. 2015). Results from other countries may not apply to China because of different cultural background and delivery of screening. Therefore, it is urgent to explore the participation and possible determinants of cancer screening in China.
Stage distribution and molecular subtypes are one of the most important factors affecting prognosis. Studies showed that majority of screen-detected breast cancers (SBCs) were small in size, node-negative, of early stages and positive for estrogen receptor (ER) and progesterone receptor (PR) (Bellio et al. 2017; Johnson et al. 2019). Other studies observed no differences in molecular subtype between SBCs and interval cancers (IBCs), emphasizing the need for additional study (Irvin et al. 2020). A few studies examined breast cancer molecular subtypes according to cancer detection mode, and the relationship between them is not well understood (Cheasley et al. 2019; Farshid and Walters 2018). The distribution of breast cancer stage at diagnosis and molecular subtype has not been reported based on a large multi-center population-based screening program in China. Besides, Chinese women tend to be denser breasts (Sung et al. 2018), and have an average age at diagnosis of 45–55 years, much earlier than most western countries (Sun et al. 2018). Understanding the diagnostic yield of breast cancer screening in China may provide scientific evidence for disease prevention and early detection.
The main aim of this study was to understand the participation and diagnostic yield of breast cancer screening program in Hebei within the period 2013–2021. In addition, we analyzed the stage distribution and molecular subtype of breast cancer cases based on different cancer detection methods, so as to provide evidence support for the optimization of breast cancer screening strategies in China.
Methods
Study design and population
In 2012, China launched the Cancer Screening Program in Urban China (CanSPUC) (Wang et al. 2019). Hebei Province initiated and conducted the screening program for breast cancer as one of the first eight regions in China. This multi-center population-based study was under CanSPUC within the period 2013–2021, which covered four cities (Shijiazhuang, Tangshan, Xingtai, and Handan) in Hebei Province. In brief, researchers approached residents living in selected neighborhoods in participating cities by telephone and personal encounter. The target population was required to be women without a history of cancer who lived local for 3 years, aged 40–74. Qualified participants were assessed for cancer risk by a defined risk score system. Trained staff interviewed all eligible participants to determine their risk factors’ exposure. Only women identified as high risk for breast cancer were invited to receive breast ultrasound and mammography examination in a designated tertiary hospital. The Fourth Hospital of Hebei Medical University Ethics Board approved the study and written informed consent was obtained from all participants prior to implementation.
Risk assessment
High-risk individuals were identified using the Harvard Cancer Risk Index (Colditz et al. 2000). The risk scoring system for breast cancer included the following factors: age, body mass index (BMI), marital status, education, smoking history, alcohol consumption, breastfeeding, menopause status, family history of breast cancer, and benign breast disease. The expert panel assigned coefficient scores for each risk factor according to its correlation with breast cancer (He Yutong et al. 2021). The final individual relative risk is calculated by divided the cumulative risk score from population's mean risk score.
Clinical procedures
High-risk participants were referred for breast ultrasound and mammography in the tertiary-level hospital. All examinations were performed by radiologists who had at least 5 years of experience. Clinical information was recorded concerning mass characteristics, asymmetry density, calcification, and architectural distortion. The largest lesion was recorded if the subject has more than one lesion in the breast (multifocal). The reporting standard in both examinations was the Breast Imaging Reporting and Data System (BI-RADS) classification (Rao et al. 2016).
Annual capacity training was conducted across all study sites to ensure that radiologists are performing BI-RADS to the uniform standard. For each examination, experts from the National Cancer Center reviewed images of 1% of randomly selected negative results and all positive results (BI-RADS categories of 3, 4, and 5). Any differences from the original diagnosis were discussed until an agreement was reached.
Follow-up data
Women who screened with positive results were followed up by telephone or retrieval of medical record information to obtain the final diagnosis and outcome. The entire cohort population was passively followed up using the population-based Hebei Cancer Registry Database from October 1, 2013 until December 31, 2021. Tumor characteristics (including stage at diagnosis, molecular subtypes, and histological types) were collected from pathological reports by trained investigators. All breast cancer cases were reported according to the International Statistical Classification of Disease, Tenth Revision (codes D05.0-D05.9 and C50.0-C50.9).
Screen-detected breast cancer (SBC) referred to cancer that is identified within 0–6 months after a positive screening result. Interval breast cancer (IBC) was defined as cancer that was detected between 0 and 24 months after a negative screening result (Niraula et al. 2020). Women who were high risk for breast cancer but did not undergo any screening are classified as noncompliant breast cancers (NBCs). Breast cancer identified in women who had a low risk of developing the disease were labeled as cancers detected outside of the screening program.
The stage at diagnosis was categorized based on the American Joint Committee on Cancer Staging (AJCC) 7th edition (Edge and Compton 2010). We defined early stage using stages 0 to I. We extracted detailed information on status of ER, PR, human epidermal growth factor receptor 2 (HER 2), and Ki67 status from the pathological reports. Results of 1% or more tumor nuclear of positive staining were classed as positive ER (ER+) or PR (PR+) (Allison et al. 2020). Positive HER2 (HER2+) was defined as positive nuclear staining intensity in "2+" and "3+" of tumor cells (Wolff et al. 2018). The molecular subtype was classified according to the 2013 St. Gallen criteria (Zhang et al. 2019).
Statistical analysis
We presented the characteristics of the study population, and overall and group-specific participation by categorical variables. Chi-squared test was used to compare the association between candidate variables and participation. We further explored the potential factors associated with participation in breast cancer screening. Odds ratio (OR) and 95% confidence intervals (CIs) were estimated by logistic regression. Diagnostic yield among different groups, including detection rates of stage at diagnosis, histological type, and molecular subtype of breast cancer, was calculated. We further analyzed the stage distribution and molecular subtype of breast cancer cases by different cancer detection modes. We used R software (version 4.1.2) for all analyses, and considered P values of 0.05 or less to be statistically significant. All hypotheses were two-sided.
Results
Characteristics of the study population
Overall, 151,973 eligible participants were recruited from 2013 to 2021. After excluding participants assessed as low risk for breast cancer (N = 109,362), those with history of cancer (N = 54), and ineffective risk assessment results (N = 10), 42,547 participants were identified as high risk for breast cancer (Fig. 1).
Characteristics of high-risk women and who underwent screening are shown in Table 1. The majority (78.1%) of participants were 45–64 years, with the average age of 54.57 years (SD = 8.2 years). In addition, 59.1% of the high-risk group was overweight or obese, 52.6% had a family history, and 68.7% had one or more benign diseases. Among the 42,547 high-risk individuals, 23,009 participants underwent screening, with participation rate of 54.08%. In total, Handan had the highest participation rate (67.7%), compared with the lowest participation rate in Xingtai (50.5%).
Factors associated with screening participation
In univariate analyses, women aged 45–69 years; married; postmenopausal; current smoking; alcohol consumption; high level of education; with benign disease and family history were more likely to participate in the study. To explore the potential factors associated with participation rate, we also conducted multivariable logistic regression models (Table 2). We found that participation rate was associated with age, education level, current smoking, alcohol consumption, menopause status, benign breast disease, and family history of breast cancer. For instance, women with benign breast disease had 30% greater likelihood of undertaking screening than those without (OR = 1.30, 95% CI 1.24–1.37). The odds of participants with family history undergoing screening were 29% higher odds than participants without (OR = 1.29, 95% CI 1.23–1.35). While women aged 70–74 were less likely to undergo screening compared to those aged 40–45 (OR = 0.68, 95% CI 0.58–0.78). We additionally adjusted the study sites and recruitment year in the model II, and the odds ratios did not change significantly.
Follow-up results
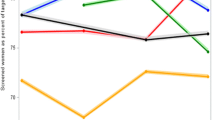
After a median time of 3.79 year follow-up, there are overall 456 breast cancer diagnoses of which 65 were screen-detected breast cancers (SBCs), 27 were interval breast cancers (IBCs), 68 were noncompliant breast cancers (NBCs), and 296 were cancers detected outside the screening program, yielding the detection rates for SBCs, IBCs, NBCs, and cancers detected outside the screening program at 0.28%, 0.12%, 0.35%, and 0.27%, respectively (Table 3). Of 321 patients with known stage, SBCs had the highest proportion of early stage (stages 0–I) (71.93%), followed by NBCs (56.25%), cancers detected outside the screening program (43.39%), and IBCs (22.22%) (Fig. 2A). Of 255 patients with known molecular subtype, the percentage of HER2-enriched and triple-negative subtype accounted for 50% of the IBCs, 19.38% of the cancers detected outside the screening program, 16.67% of the SBCs, and 12.2% of the NBCs (Fig. 2B).
The tumor characteristics of breast cancer stage, histologic type, and molecular subtype by screening status are summarized in Table 4. The detection rate of breast cancer was 0.40% (92/23009) in the screening women and 0.28% (364/128900) in the non-screening women, and the OR was 1.42 (95% CI 1.13–1.78; P = 0.003). For stage at diagnosis, we observed a higher detection rate of breast cancer in the screening group, with ORs of 2.42 (95% CI 1.72–3.41) for stage 0–I and 1.83 (95% CI 1.19–2.80) for stage II. For histological type and molecular subtype, we observed a higher detection rate of breast cancer in the screening group, with ORs of 1.71 (95% CI 1.28–2.28) for ductal type and 2.12 (95% CI 1.26–3.54) for luminal A subtype.
Discussion
This study reported the results of 151,973 participates underwent breast cancer screening from 2013 to 2021 in China. Our results suggest that the detection rate and early diagnosis rates were higher in the screening group than in the non-screening group. In addition, interval cancers (IBCs) were more likely than screen-detected cancers (SBCs) to be of HER2-enriched and triple-negative subtype. To our knowledge, this is the first to report detection rate by molecular subtype of breast cancer screening program in a large multi-center population-based dataset in China. The results suggest that we need to further improve the diagnostic yield especially in interval cancers. Our study underscores the urgency to increase breast cancer awareness and early detection in China.
In our study, the overall participation rate among the high-risk women was different (58.19%) from other studies 47.27%-48.2% (Guo et al. 2021; Zhang et al. 2021), but higher than the overall participation in China (40.3%). Publicity and education, mobilization organizations, health awareness of residents, service capabilities of hospitals, and communities all contributed to different participation by regions. Smoking, alcohol consumption, a family history of breast cancer, and benign breast diseases have been confirmed for breast cancer risk factors (Sun et al. 2017). This research discovered that individuals with these characteristics were more likely to engage in breast cancer screening. Women with family history and benign diseases may have more health-oriented consciousness, and more likely to have routine health screening (Li et al. 2020). In addition, we found that participation rates were lower among women aged 70–74 years and with lower education. A lack of awareness and understanding regarding breast cancer screening may be a potential cause. In one meta-analysis from 29 studies, non-participation in screening was associated with low education (Ding et al. 2022). Therefore, targeted education interventions for awareness and cancer prevention are urgently needed in areas with lower screening rates, such as rural communities.
The overall breast cancer detection rates in the screening and non-screening group were at 0.40% and 0.28%, respectively. However, the detection rate was lower than other countries, for example, the United States (0.56%), the Netherlands (0.6%), and Japan (0.5%) (Barlow et al. 2020; Luiten et al. 2020; Ohuchi et al. 2016). It may be due to the low participation of screening and the insufficient follow-up time in our research. In screening group, one-third of breast cancers were IBCs. The incidence and proportion of IBCs may differ based on age and the length of screening interval (Houssami 2017). Our results were similar to the Flemish Breast Cancer Screening Program, with 67% of SBC and 33% of IBC (Timmermans et al. 2017). The results showed that the incidence of IBCs is significantly higher in women aged 50–54 as compared to older women, while the incidence of SBCs is significantly higher in women aged 60–64. A recent study observed that young age of diagnosis was associated with worse survival and more aggressive clinicopathologic features (Timmermans et al. 2017). Therefore, more attention should be paid to and strengthening preventive screening in young women less than 55 years.
The essence of cancer screening is early diagnosis and early treatment of cancer to reduce mortality. We observed a higher detection rate of early stage cases in screening group than in non-screening group, in accordance with the previous reports (Zhang et al. 2021; Huang et al. 2021). A randomized controlled trial in Japan showed that the screened population had 71.3% of cases in stage 0–I, while the non-screened population had 52.0% of cases in stage 0–I (Ohuchi et al. 2016). One population-based breast cancer study in Sweden indicated that screening reduced the risk of advanced breast cancer by 25% in screened population (Duffy et al. 2020). A cohort study for 6396 women aged 50–65 in New South Wales showed that SBCs were more likely to be diagnosed with localized disease (64.1% vs. 48.1%), compared with non-SBCs (Woods et al. 2016). Differences in stage distribution may partly be explained the better survival of SBCs.
Our research supports prior studies which indicate that there was a noticeably higher proportion of luminal A subtype in SBCs (Sihto et al. 2008; Kobayashi et al. 2017), and IBCs were associated with poor tumor characteristics (O'Brien et al. 2018; Defossez et al. 2018). In this study, we found that luminal A subtype was more prevalent in screening women than non-screening ones. A study of 4559 patients in a Chilean cohort reported that the proportion of stage I and "luminal" subtype were significantly higher in SBCs than non-SBCs (Walbaum et al. 2021). One Canadian population-based screening program discovered that IBCs were more probable to present as ER negative compared to SBCs (OR, 2.88; 95% CI 2.01–4.13) (Niraula et al. 2020). The underrepresentation of triple-negative and HER2-enriched subtypes in SBCs is expected as these tumors grow rapidly and thus have shorter preclinical phases. As a result, they are more likely to become symptomatic between scheduled breast cancer screenings (Farshid and Walters 2018). Our study shows that conventional screening is more likely to detect indolent cancer types than fatally aggressive ones. Improvement of diagnostic yield of interval cancers requires personalized screening strategies based on baseline risks in breast cancer screening.
Our study has some limitations. First, the participants were recruited from four urban areas, where healthcare was fairly accessible. Therefore, this study population may not be represent the entire population of Hebei Province. Second, while detailed epidemiological information was collected in a standardized manner by trained study staff, smoking and drinking status were self-reported, which may have led to misclassification. Third, outcome information for breast cancer patients is still being obtained through ongoing follow-up work. Further studies are needed to evaluate the screening effect on breast cancer mortality.
Conclusions
In summary, in this large-scale screening program, breast cancer screening participation rates were affected by age, education level, postmenopausal status, smoking, drinking, benign breast disease, and family history of breast cancer. We illustrated higher detection rate for both early stage cases and luminal A subtype in screening group than non-screening group. Women who participated in population screening and had interval cancers had a worse subtype and stage distribution. Our results indicate that we need to improve the diagnostic yield, especially in interval cancer, in the future. These findings will provide data support for optimizing population-based breast cancer screening practices in China.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Allison K, Hammond M, Dowsett M, McKernin S, Carey L, Fitzgibbons P et al (2020) Estrogen and progesterone receptor testing in breast cancer: ASCO/CAP guideline update. J Clin Oncol 38(12):1346–1366
Barlow W, Beaber E, Geller B, Kamineni A, Zheng Y, Haas J et al (2020) Evaluating screening participation, follow-up, and outcomes for breast, cervical, and colorectal cancer in the PROSPR consortium. J Natl Cancer Inst 112(3):238–246
Bellio G, Marion R, Giudici F, Kus S, Tonutti M, Zanconati F et al (2017) Interval breast cancer versus screen-detected cancer: comparison of clinicopathologic characteristics in a single-center analysis. Clin Breast Cancer 17(7):564–571
Canelo-Aybar C, Posso M, Montero N, Solà I, Saz-Parkinson Z, Duffy S et al (2022) Benefits and harms of annual, biennial, or triennial breast cancer mammography screening for women at average risk of breast cancer: a systematic review for the European Commission Initiative on Breast Cancer (ECIBC). Br J Cancer 126(4):673–688
Cao M, Li H, Sun D, He S, Yu Y, Li J et al (2021) Cancer screening in China: the current status, challenges, and suggestions. Cancer Lett 506:120–127
Cheasley D, Li N, Rowley S, Elder K, Mann G, Loi S et al (2019) Molecular comparison of interval and screen-detected breast cancers. J Pathol 248(2):243–252
Chen WLN, Cao M, Ren J, Shi J, Chen H et al (2020) Preliminary analysis of cancer screening program in urban China from 2013 to 2017. China Cancer 29:1–6
Colditz G, Atwood K, Emmons K, Monson R, Willett W, Trichopoulos D et al (2000) Harvard report on cancer prevention volume 4: Harvard Cancer Risk Index. Risk Index Working Group, Harvard Center for Cancer Prevention. Cancer Causes Control 11(6):477–488
Defossez G, Quillet A, Ingrand P (2018) Aggressive primary treatments with favourable 5-year survival for screen-interval breast cancers. BMC Cancer 18(1):393
Ding L, Wang J, Greuter M, Goossens M, Van Hal G, de Bock G (2022) Determinants of Non-participation in population-based breast cancer screening: a systematic review and meta-analysis. Front Oncol 12:817222
Duffy S, Tabár L, Yen A, Dean P, Smith R, Jonsson H et al (2020) Mammography screening reduces rates of advanced and fatal breast cancers: results in 549,091 women. Cancer 126(13):2971–2979
Edge SB, Compton CC (2010) The American Joint Committee on Cancer: the 7thedit ion of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol 17(6):1471–1474
Farshid G, Walters D (2018) Molecular subtypes of screen-detected breast cancer. Breast Cancer Res Treat 172(1):191–199
Guo L, Zhang S, Liu S (2021) Analysis of compliance with breast cancer screening and its influencing factors in urban areas of Henan province. J Pract Oncol 35(4):307–313
He Yutong LY, Wei G, Jianjun H, Xinyan M, Changjin X, Shuohua C, Jing W, Di L, Jin S, Baoen S (2021) Analysis for the breast cancer screening among urban population in Hebei province, 2018–2019. Chin J Prev Med 55(4):89–92
Houssami NHK (2017) The epidemiology, radiology and biological characteristics of interval breast cancers in population mammography screening. NPJ Breast Cancer 13(3):12
Huang Y, Wang H, Lyu Z, Dai H, Liu P, Zhu Y et al (2021) Development and evaluation of the screening performance of a low-cost high-risk screening strategy for breast cancer. Cancer Biol Med 19(9):1375–1384
Irvin VLZZ, Simon MS, Chlebowski RT, Luoh SW, Shadyab AH, Krok-Schoen JL, Tabung FK, Qi L, Stefanick ML, Schedin P, Jindal S (2020) Comparison of mortality among participants of women’s health initiative trials with screening-detected breast cancers vs interval breast cancers. JAMA Netw Open 3(6):e207227
Johnson K, Zackrisson S, Rosso A, Sartor H, Saal L, Andersson I et al (2019) Tumor characteristics and molecular subtypes in breast cancer screening with digital breast tomosynthesis: the Malmö Breast tomosynthesis screening trial. Radiology 293(2):273–281
Kobayashi N, Hikichi M, Ushimado K, Sugioka A, Kiriyama Y, Kuroda M et al (2017) Differences in subtype distribution between screen-detected and symptomatic invasive breast cancer and their impact on survival. Clin Transl Oncol 19(10):1232–1240
Lei S, Zheng R, Zhang S et al (2021) Global patterns of breast cancer incidence and mortality: a population-based cancer registry data analysis from 2000 to 2020. Cancer Commun (london, England) 41(11):1183–1194
Li H, Zeng H, Zheng R et al (2020) Association of cancer awareness levels with the risk of cancer in rural China: a population-based cohort study. Cancer 126(20):4563–4571
Luiten J, Voogd A, Luiten E, Broeders M, Roes K, Tjan-Heijnen V et al (2020) Recall and outcome of screen-detected microcalcifications during 2 decades of mammography screening in the Netherlands National Breast Screening Program. Radiology 294(3):528–537
Moss S, Wale C, Smith R, Evans A, Cuckle H, Duffy S (2015) Effect of mammographic screening from age 40 years on breast cancer mortality in the UK Age trial at 17 years’ follow-up: a randomised controlled trial. Lancet Oncol 16(9):1123–1132
Mottram R, Knerr W, Gallacher D, Fraser H, Al-Khudairy L, Ayorinde A et al (2021) Factors associated with attendance at screening for breast cancer: a systematic review and meta-analysis. BMJ Open 11(11):e046660
Niraula S, Biswanger N, Hu P, Lambert P, Decker K (2020) Incidence, characteristics, and outcomes of interval breast cancers compared with screening-detected breast cancers. JAMA Netw Open 3(9):e2018179
O’Brien K, Mooney T, Fitzpatrick P, Sharp L (2018) Screening status, tumour subtype, and breast cancer survival: a national population-based analysis. Breast Cancer Res Treat 172(1):133–142
Ohuchi N, Suzuki A, Sobue T, Kawai M, Yamamoto S, Zheng Y et al (2016) Sensitivity and specificity of mammography and adjunctive ultrasonography to screen for breast cancer in the Japan Strategic Anti-cancer Randomized Trial (J-START): a randomised controlled trial. Lancet (london, England) 387(10016):341–348
Rao A, Feneis J, Lalonde C, Ojeda-Fournier H (2016) A pictorial review of changes in the BI-RADS fifth edition. Radiographics 36(3):623–639
Sihto H, Lundin J, Lehtimäki T, Sarlomo-Rikala M, Bützow R, Holli K et al (2008) Molecular subtypes of breast cancers detected in mammography screening and outside of screening. Clin Cancer Res 14(13):4103–4110
Sun Y, Zhao Z, Yang Z, Xu F, Lu H, Zhu Z et al (2017) Risk factors and preventions of breast cancer. Int J Biol Sci 13(11):1387–1397
Sun KZR, Gu X, Zhang S, Zeng H, Zou X et al (2018) Incidence trend and change in the age distribution of female breast cancer in cancer registration areas of China from 2000 to 2014. Chin J Prev Med 52(6):567–571
Sung H, Ren J, Li J, Pfeiffer R, Wang Y, Guida J et al (2018) Breast cancer risk factors and mammographic density among high-risk women in urban China. NPJ Breast Cancer 4:3
Sung HFJ, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249
Timmermans LBL, Bacher K, Van Herck K, Lemmens K, Van Ongeval C, Van Steen A, Martens P, De Brabander I, Goossens M, Thierens H (2017) Screen-detected versus interval cancers: effect of imaging modality and breast density in the Flemish Breast Cancer Screening Programme. Eur Radiol 27(9):3810–3819
Walbaum BPK, Medina L, Merino T, Camus M, Razmilic D, Navarro ME, Dominguez F, Cordova-Delgado M, Pinto MP, Acevedo F, Sánchez C (2021) Screen-detected breast cancer is associated with better prognosis and survival compared to self-detected/symptomatic cases in a Chilean cohort of female patients. Breast Cancer Res Treat 189(2):561–569
Wang Y, Chen H, Li N, Ren J, Zhang K, Dai M et al (2019) Ultrasound for breast cancer screening in high-risk women: results from a population-based cancer screening program in China. Front Oncol 9:286
Williams J, Garvican L, Tosteson A, Goodman D, Onega T (2015) Breast cancer screening in England and the United States: a comparison of provision and utilisation. Int J Public Health 60(8):881–890
Wolff A, Hammond M, Allison K, Harvey B, Mangu P, Bartlett J et al (2018) Human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline focused update. J Clin Oncol 36(20):2105–2122
Woods LMRB, O’Connell DL, Lawrence G, Coleman MP (2016) Are international differences in breast cancer survival between Australia and the UK present amongst both screen-detected women and non-screen-detected women? survival estimates for women diagnosed in West Midlands and New South Wales 1997–2006. Int J Cancer 138(10):2404–2414
Wu Z, Liu Y, Li X, Song B, Ni C, Lin F (2019) Factors associated with breast cancer screening participation among women in mainland China: a systematic review. BMJ Open 9(8):e028705
Xie ZXW, Liang Y, Lin H, Wu J, Cui Y, Su X, Zeng D (2022) Associations of obesity, physical activity, and screening with state-level trends and racial and ethnic disparities of breast cancer incidence and mortality in the US. JAMA Netw Open 5(6):e2216958
Zhang L, Huang Y, Feng Z, Wang X, Li H, Song F et al (2019) Comparison of breast cancer risk factors among molecular subtypes: a case-only study. Cancer Med 8(4):1882–1892
Zhang X, Yang L, Liu S, Li H, Li Q, Cheng Y et al (2021) Evaluation of different breast cancer screening strategies for high-risk women in Beijing, China: a real-world population-based study. Front Oncol 11:776848
Acknowledgements
Our sincere thanks go out to all the hospitals and communities involved in cancer screening in Hebei Province for their cooperation. Participants were sincerely thanked for taking part in this program.
Funding
The authors declare that no funds, grants, or other supports were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
The study was designed by YH. SW drafted the manuscript and performed the data analysis. DL and JS contributed to data collection and quality control. DL and YL are accountable for the administration and supervision. YH, SM, and XD revised the paper. This version of article was approved by all authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wu, S., Liang, D., Shi, J. et al. Evaluation of a population-based breast cancer screening in North China. J Cancer Res Clin Oncol 149, 10119–10130 (2023). https://doi.org/10.1007/s00432-023-04905-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-04905-w