Abstract
Purpose
Prostate cancer (PCa) is the second leading cause of cancer death among men in the USA. The emergence of resistance to androgen deprivation therapy gives rise to metastatic castration-resistant prostate cancer. Eprinomectin (EP) is a member of a family of drugs called avermectins with parasiticide and anticancer properties. The pupose of this study was to evaluate the anticancer effects of EP against metastatic PCa using cellular models.
Methods
In this study, we have investigated the effect of EP’s anticancer properties and delineated the underlying mechanisms in the DU145 cellular model using several assays such as cell viability assay, colony formation assay, wound-healing assay, immunofluorescence, apoptosis assay, cell cycle analysis, and immunoblotting.
Results
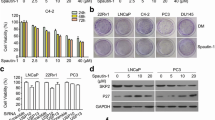
Our results indicate that EP significantly inhibits the cell viability, colony formation, and migration capacities of DU145 cells. EP induces cell cycle arrest at the G0/G1 phase, apoptosis via the activation of different caspases, and autophagy through the increase in the generation of reactive oxygen species and endoplasmic reticulum stress. In addition, EP downregulates the expression of cancer stem cell markers and mediates the translocation of β-catenin from the nucleus to the cytoplasm, indicating its role in inhibiting downstream target genes such as c-Myc and cyclin D1.
Conclusion
Our study shows that EP has tremendous potential to target metastatic PCa cells and provides new avenues for therapeutic approaches for advanced PCa.












Similar content being viewed by others
Data availability
The data represented here are available upon request from the corresponding author.
References
Bincoletto C, Bechara A, Pereira GJ, Santos CP, Antunes F, Peixoto da-Silva J, Muler M, Gigli RD, Monteforte PT, Hirata H, Jurkiewicz A, Smaili SS (2013) Interplay between apoptosis and autophagy, a challenging puzzle: new perspectives on antitumor chemotherapies. Chem Biol Interact 206:279–288
Boyer-Guittaut M, Poillet L, Liang Q, Bole-Richard E, Ouyang X, Benavides GA, Chakrama FZ, Fraichard A, Darley-Usmar VM, Despouy G, Jouvenot M, Delage-Mourroux R, Zhang J (2014) The role of GABARAPL1/GEC1 in autophagic flux and mitochondrial quality control in MDA-MB-436 breast cancer cells. Autophagy 10:986–1003
Chinnapaka S, Zheng G, Chen A, Munirathinam G (2019) Nitro aspirin (NCX4040) induces apoptosis in PC3 metastatic prostate cancer cells via hydrogen peroxide (H2O2)-mediated oxidative stress. Free Radic Biol Med 143:494–509
Dasari S, Alpa Samy A, Kajdacsy-Balla M, Bosland C, Munirathinam G (2018a) Vitamin K2, a menaquinone present in dairy products targets castration-resistant prostate cancer cell-line by activating apoptosis signaling. Food Chem Toxicol 115:218–227
Dasari S, Alpa Samy P, Narvekar VS, Dontaraju R, Dasari AK, Munirathinam G (2018b) Polygodial analog induces apoptosis in LNCaP prostate cancer cells. Eur J Pharmacol 828:154–162
Dasari S, Bakthavachalam V, Chinnapaka S, Venkatesan R, Samy A, Munirathinam G (2020) Neferine, an alkaloid from lotus seed embryo targets HeLa and SiHa cervical cancer cells via pro-oxidant anticancer mechanism. Phytother Res 34:2366–2384
Davies AH, Zoubeidi A (2021) Targeting androgen receptor signaling: a historical perspective. Endocr Relat Cancer 28:T11–T18
de Almagro MC, Vucic D (2012) The inhibitor of apoptosis (IAP) proteins are critical regulators of signaling pathways and targets for anti-cancer therapy. Exp Oncol 34:200–211
Derakhshan A, Chen Z, Van Waes C (2017) Therapeutic small molecules target inhibitor of apoptosis proteins in cancers with deregulation of extrinsic and intrinsic cell death pathways. Clin Cancer Res 23:1379–1387
Dong L, Zieren RC, Xue W, de Reijke TM, Pienta KJ (2019) Metastatic prostate cancer remains incurable, why? Asian J Urol 6:26–41
Gao F, Sun Z, Kong F, Xiao J (2020) Artemisinin-derived hybrids and their anticancer activity. Eur J Med Chem 188:112044
Gheewala T, Skwor T, Munirathinam G (2018) Photodynamic therapy using pheophorbide and 670nm LEDs exhibits anti-cancer effects in-vitro in androgen dependent prostate cancer. Photodiagnosis Photodyn Ther 21:130–137
Glick D, Barth S, Macleod KF (2010) Autophagy: cellular and molecular mechanisms. J Pathol 221:3–12
Gupta PB, Onder TT, Jiang G, Tao K, Kuperwasser C, Weinberg RA, Lander ES (2009) Identification of selective inhibitors of cancer stem cells by high-throughput screening. Cell 138:645–659
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674
Heo JR, Kim SM, Hwang KA, Kang JH, Choi KC (2018) Resveratrol induced reactive oxygen species and endoplasmic reticulum stressmediated apoptosis, and cell cycle arrest in the A375SM malignant melanoma cell line. Int J Mol Med 42:1427–1435
Iwadate Y, Suganami A, Tamura Y, Matsutani T, Hirono S, Shinozaki N, Hiwasa T, Takiguchi M, Saeki N (2017) The pluripotent stem-cell marker alkaline phosphatase is highly expressed in refractory glioblastoma with DNA hypomethylation. Neurosurgery 80:248–256
Juarez M, Schcolnik-Cabrera A, Duenas-Gonzalez A (2018) The multitargeted drug ivermectin: from an antiparasitic agent to a repositioned cancer drug. Am J Cancer Res 8:317–331
Kaur J, Bachhawat AK (2009) A modified Western blot protocol for enhanced sensitivity in the detection of a membrane protein. Anal Biochem 384:348–349
Klose J, Kobalz U (1995) Two-dimensional electrophoresis of proteins: an updated protocol and implications for a functional analysis of the genome. Electrophoresis 16:1034–1059
Kodama M, Kodama T, Newberg JY, Katayama H, Kobayashi M, Hanash SM, Yoshihara K, Wei Z, Tien JC, Rangel R, Hashimoto K, Mabuchi S, Sawada K, Kimura T, Copeland NG, Jenkins NA (2017) In vivo loss-of-function screens identify KPNB1 as a new druggable oncogene in epithelial ovarian cancer. Proc Natl Acad Sci U S A 114:E7301–E7310
Kranz P, Neumann F, Wolf A, Classen F, Pompsch M, Ocklenburg T, Baumann J, Janke K, Baumann M, Goepelt K, Riffkin H, Metzen E, Brockmeier U (2017) PDI is an essential redox-sensitive activator of PERK during the unfolded protein response (UPR). Cell Death Dis 8:e2986
Kypta RM, Waxman J (2012) Wnt/beta-catenin signalling in prostate cancer. Nat Rev Urol 9:418–428
Levy JMM, Towers CG, Thorburn A (2017) Targeting autophagy in cancer. Nat Rev Cancer 17:528–542
Li D, Lv H, Hao X, Hu B, Song Y (2018) Prognostic value of serum alkaline phosphatase in the survival of prostate cancer: evidence from a meta-analysis. Cancer Manag Res 10:3125–3139
Li YQ, Zheng Z, Liu QX, Lu X, Zhou D, Zhang J, Zheng H, Dai JG (2021) Repositioning of antiparasitic drugs for tumor treatment. Front Oncol 11:670804
Melotti A, Mas C, Kuciak M, Lorente-Trigos A, Borges I, Ruiz i Altaba A (2014) The river blindness drug Ivermectin and related macrocyclic lactones inhibit WNT-TCF pathway responses in human cancer. EMBO Mol Med 6:1263–1278
Milkovic L, Cipak Gasparovic A, Cindric M, Mouthuy PA, Zarkovic N (2019) Short overview of ros as cell function regulators and their implications in therapy concepts. Cells 8:793
Molinari G, Kujawski M, Scuto A, Soloneski S, Larramendy ML (2013) DNA damage kinetics and apoptosis in ivermectin-treated Chinese hamster ovary cells. J Appl Toxicol 33:1260–1267
Mulcahy Levy JM, Thorburn A (2020) Autophagy in cancer: moving from understanding mechanism to improving therapy responses in patients. Cell Death Differ 27:843–857
Murillo-Garzon V, Kypta R (2017) WNT signalling in prostate cancer. Nat Rev Urol 14:683–696
Nikoletopoulou V, Markaki M, Palikaras K, Tavernarakis N (2013) Crosstalk between apoptosis, necrosis and autophagy. Biochim Biophys Acta 1833:3448–3459
Rashid HO, Yadav RK, Kim HR, Chae HJ (2015) ER stress: autophagy induction, inhibition and selection. Autophagy 11:1956–1977
Rawla P (2019) Epidemiology of prostate cancer. World J Oncol 10:63–89
Samy A, Bakthavachalam V, Vudutha M, Vinjamuri S, Chinnapaka S, Munirathinam G (2020) Eprinomectin, a novel semi-synthetic macrocylic lactone is cytotoxic to PC3 metastatic prostate cancer cells via inducing apoptosis. Toxicol Appl Pharmacol 401:115071
Santer FR, Erb HH, Oh SJ, Handle F, Feiersinger GE, Luef B, Bu H, Schafer G, Ploner C, Egger M, Rane JK, Maitland NJ, Klocker H, Eder IE, Culig Z (2015) Mechanistic rationale for MCL1 inhibition during androgen deprivation therapy. Oncotarget 6:6105–6122
Shang S, Hua F, Hu ZW (2017) The regulation of beta-catenin activity and function in cancer: therapeutic opportunities. Oncotarget 8:33972–33989
Shoop WL, DeMontigny P, Fink DW, Williams JB, Egerton JR, Mrozik H, Fisher MH, Skelly BJ, Turner MJ (1996) Efficacy in sheep and pharmacokinetics in cattle that led to the selection of eprinomectin as a topical endectocide for cattle. Int J Parasitol 26:1227–1235
Siegel RL, Miller KD, Fuchs HE, Jemal A (2022) Cancer statistics, 2022. CA Cancer J Clin 72:7–33
Stoddart MJ (2011) Cell viability assays: introduction. Methods Mol Biol 740:1–6
Teo MY, Rathkopf DE, Kantoff P (2019) Treatment of advanced prostate cancer. Annu Rev Med 70:479–499
Valenta T, Hausmann G, Basler K (2012) The many faces and functions of beta-catenin. EMBO J 31:2714–2736
Wallace KL, Landsteiner A, Bunner SH, Engel-Nitz NM, Luckenbaugh AN (2021) Increasing prevalence of metastatic castration-resistant prostate cancer in a managed care population in the United States. Cancer Causes Control 32:1365–1374
Wong YK, Xu C, Kalesh KA, He Y, Lin Q, Wong WSF, Shen HM, Wang J (2017) Artemisinin as an anticancer drug: recent advances in target profiling and mechanisms of action. Med Res Rev 37:1492–1517
Yu Z, Pestell TG, Lisanti MP, Pestell RG (2012) Cancer stem cells. Int J Biochem Cell Biol 44:2144–2151
Zhang Y, Wang X (2020) Targeting the Wnt/beta-catenin signaling pathway in cancer. J Hematol Oncol 13:165
Zhang S, Wang Y, Chen Z, Kim S, Iqbal S, Chi A, Ritenour C, Wang YA, Kucuk O, Wu D (2013) Genistein enhances the efficacy of cabazitaxel chemotherapy in metastatic castration-resistant prostate cancer cells. Prostate 73:1681–1689
Zhang X, Zhang G, Zhai W, Zhao Z, Wang S, Yi J (2020) Inhibition of TMEM16A Ca(2+)-activated Cl(−) channels by avermectins is essential for their anticancer effects. Pharmacol Res 156:104763
Acknowledgements
This study is partly supported by the Master in Medical Biotechnology Program, Department of Biomedical Science, UICOM, Rockford, IL.
Funding
This study is partly supported by the Master’s in Medical Biotechnology Program, Department of Biomedical Science, UICOM, Rockford, IL.
Author information
Authors and Affiliations
Contributions
Conceptualization, A.S., M.H., and G.M.; methodology, A.S., M.H., and G.M.; writing original draft, A.S., M.H., writing, review, and editing A.S., M.H., and G.M.; supervision, G.M. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Samy, A., Hussein, M.A. & Munirathinam, G. Eprinomectin: a derivative of ivermectin suppresses growth and metastatic phenotypes of prostate cancer cells by targeting the β-catenin signaling pathway. J Cancer Res Clin Oncol 149, 9085–9104 (2023). https://doi.org/10.1007/s00432-023-04829-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-04829-5