Abstract
Background
The indications for liver transplantation (LT) in patients with hepatocellular carcinoma (HCC) continue to evolve. The aim of this study was to report outcomes in patients who underwent living donor liver transplantation (LDLT) for HCC outside traditional criteria including macrovascular invasion (MVI).
Methods
We reviewed outcomes in patients who met the University of California San Francisco (UCSF) criteria (n = 159) and our center-specific criteria (UCSF+) (largest tumor diameter ≤ 10 cm, any tumor number, AFP ≤ 1000 ng/ml) (n = 58). We also assessed outcomes in patients with MVI (n = 27).
Results
The median follow was 28 (10.6–42.7) months. The 5 year overall survival and risk of recurrence (RR) in the UCSF and UCSF + group was 71% vs 69% (P = 0.7) and 13% vs 36% (P = 0.1) respectively. When patients with AFP > 600 ng/ml were excluded from the UCSF + group, RR was 27% (P = 0.3). Among patients with MVI who had downstaging (DS), 4/5(80%) in low-risk group (good response and AFP ≤ 100 ng/ml) and 2/10 (20%) in the high-risk group (poor response or AFP > 100 ng/ml) were alive at the last follow-up. When DS was not feasible, 3/3 (100%) in the low-risk group (AFP ≤ 100 ng/ml + Vp1-2 MVI) and 1/9 (9.1%) in the high-risk group (AFP > 100 or Vp3 MVI) were alive. The 5 year OS in the low-risk MVI group was 85% (P = 0.003).
Conclusion
With inclusion of AFP, response to downstaging and degree of MVI, acceptable survival can be achieved with LDLT for HCC outside traditional criteria.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Liver transplantation (LT) is the most effective treatment for patients with cirrhosis and small hepatocellular carcinoma (HCC) (Costentin et al. 2019). Despite being restrictive, Milan criteria (single tumor ≤ 5 cm, upto 3 tumors ≤ 3 cm) remain the benchmark for LT in HCC. LT for tumors within Milan criteria is associated with low recurrence rate and a 5 year survival of 60–80% (Mazzaferro et al. 1996; Toso et al. 2011). The University of California San Francisco criteria (UCSF) (Single tumor ≤ 6.5 cm, up to 3 tumors ≤ 4.5 cm, total tumor diameter ≤ 8 cm), was proposed in the year 2001, and has outcomes comparable to Milan criteria (Yao et al. 2001, 2007). A number of other expanded criteria have been proposed to further increase transplant eligibility (Duvoux et al. 2012; Mazzaferro et al. 2018; Sapisochin et al. 2016; Lei et al. 2014). Living donor liver transplantation (LDLT) is a viable alternative to deceased donor liver transplantation (DDLT) for HCC. Worldwide, LDLT centers have expanded the cutoffs on tumor size and number to increase transplant eligible patients (Hong et al. 2016; Lee et al. 2016, 2008). While most of the expanded criteria in DDLT and LDLT allow modest expansion in transplant pool, the more liberal criteria often mandate a preoperative biopsy or PET scan for patient selection (Sapisochin et al. 2016; Lei et al. 2014; Lee et al. 2016). Although prognostically significant, widespread adoption of preoperative biopsy has been limited by presumed risks of tumor seeding, bleeding, and misdiagnosis (Sparchez et al. 2018). The National Cancer Center Korea (NCCK) criteria used PET scan to select patients with HCC ≤ 10 cm irrespective of tumor number (Lee et al. 2016). Factors such as low sensitivity, cost effectiveness, and variable SUV cutoffs limit wide spread utility of PET scans for selecting HCC patients for transplantation (Lu et al. 2019).
At our center, LDLT has been routinely performed since 2012. We developed our own center-specific selection criteria for LDLT in patients with HCC. Since 2012, patients within UCSF criteria and those outside UCSF criteria (maximum tumor size ≤ 10 cm, any tumor number) with AFP < 1000 ng/ml were offered LDLT. In addition, LDLT was also offered to selected patients with macrovascular invasion (MVI).
The objective of this study was to share our experience with LDLT for HCC using expanded transplant criteria.
Materials and methods
Between April 2012 and September 2019, 874 patients underwent LDLT at our center. Among them, 244 patients with a preoperative diagnosis of HCC, were reviewed retrospectively.
Our patient and donor selection criteria and workup have been described elsewhere (Dar et al. 2015, 2018). The decision to proceed with LDLT was finalized in multi-disciplinary team meeting and liver transplant listing meeting. These meetings include team members from transplant surgery, hepatology, radiology, anesthesiology, and administrative committee. In addition, all potential donors were assessed independently by donor advocates.
Patient selection for LDLT
The radiological diagnosis of HCC was confirmed on dynamic imaging (CT scan or MRI) with an arterially enhancing lesion ≥ 1 cm demonstrating wash out on venous phase (Bruix and Sherman 2011). LDLT was only considered if there was no evidence of extra hepatic metastases and main portal vein tumor thrombosis (PVTT). All patients within UCSF criteria were offered upfront LDLT. In addition patients outside UCSF criteria (largest tumor size upto 10 cm, any tumor number and AFP < 1000 ng/ml) (UCSF +) were also considered for upfront LDLT. Patients with AFP > 1000 ng/ml and UCSF + tumors were considered for downstaging (DS) [(Transarterial chemo embolization (TACE), radio frequency ablation (RFA), microwave ablation (MWA), percutaneous ethanol ablation (PEA)]. In patients with an anticipated delay of > 3 months, locoregional therapy (LRT) was used as bridging therapy. For downstaging, TACE was performed routinely, while in selected patients (e.g. multi focal bilobar disease), it was combined with other ablative therapies. A drop in AFP to < 1000 ng/ml and radiological response based on modified response evaluation criteria in solid tumors (mRECIST) at 6–8 weeks was used to determine effectiveness of LRT and candidacy for LDLT (Lencioni et al. 2010). For bridging, RFA, MWA. PEA or TACE was considered based on the size and location of the tumors and underlying liver failure. In the post-transplant period, an annual surveillance CT scan was performed for the first two years with six monthly AFP levels. After two years, AFP and US was performed at 6 months interval. Sorafenib was used in patients with high-risk HCC (MVI, poor grade, tumors outside UCSF criteria) one month after LT. High-risk patients had their first surveillance CT scan 3 months after LT.
LDLT for macrovascular invasion
During the study period, patients with HCC and macrovascular invasion (MVI) also underwent LDLT. We reviewed the pre-transplant imaging and patients who fulfilled the A-VENA criteria for MVI were included (Sherman et al. 2019). With regards to PVTT, patients with tumor thrombosis in segmental branches (Vp1-2), and lobar branches (Vp3) were considered for LDLT (Kanehara 2003). All these patients were considered for downstaging with response evaluation and an observation period (4–6 months). Only those with stable disease (no interval progression to extra hepatic disease and substantial rise in AFP) were offered LDLT. In patients with decompensated liver disease precluding DS, LDLT was offered selectively. All these patients had informed discussions regarding the higher risk of post-transplant recurrence. This was subsequently documented in the patient files by the transplants surgeons and hepatologists.
Study design and statistical analysis
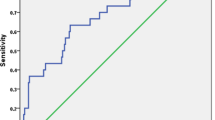
For this study, we compared demographics, etiology, tumor-related features, MELD score, and AFP for UCSF and UCSF + groups. Frequencies with percentage were reported for categorical data while medians with inter quartile range (IQR) were reported for interval data. For categorical variables, chi-squared and Fischer test were used while Mann–Whitney U test was used for interval variables. Overall survival (OS) was calculated by subtracting date of death from the date of transplantation. For survival analysis, Kaplan–Meier curves were used and Log rank test was used to determine significance. We also looked at the impact of AFP > 600 ng/ml on survival since it has been shown to be a strong predictor of outcomes (Bhatti et al. 2020a, b). On receiver operator curves (ROC) analysis, An AFP cutoff of 600 ng/ml (AUC = 0.77, P < 0.001) was a significant factor for recurrence. Among patients with MVI, we developed prognostic groups based on (1) response to DS, (2) AFP < or > 100 ng/ml, (3) Vp1-2 versus Vp3 PVTT as shown previously by other groups (Choi et al. 2017; Soin et al. 2020). Patients with complete or partial response were categorized as good responders while those with stable or progressive disease were considered as poor responders to LRT. A P value < 0.05 was considered statistically significant. The institutional review board and hospital ethics committee approved the study (IRB #433-1253-2020).
Results
The median follow-up from transplantation was 28 (10.6–42.7) months. Median AFP was 16.2 (5.3–81) (range = 0.7–5129) ng/ml. The 5 year OS for the entire cohort was 71%. The 5 year OS with an AFP < or > 600 ng/ml was 74% and 27% (P = 0.01) (not shown). Table 1 shows the operative details in our cohort. There was no donor mortality.
UCSF and UCSF + groups
We looked at various patient and tumor-related features in the UCSF (n = 159) and UCSF+ (n = 58) group (Table 2). Other than tumor size and number, there was a significant difference in median AFP level (P = 0.01) and microvascular invasion (P = 0.001). There was no significant difference with regards to HCC with AFP > 600 ng/ml and poor differentiation.
Outcomes with LDLT
The actual survival rate in the UCSF and UCSF+ group was 117/159 (73.5%) and 44/58 (75.8%) (P = 0.7). The estimated 5 year OS was 72% and 69% for UCSF and UCSF+ group (P = 0.7) (Fig. 1A). The 5 year OS was 79% for patients who met Toronto criteria and Hangzhou criteria, and was comparable to patients within UCSF criteria (Fig. 1B, C). Among patients in the UCSF+ group, 16/58 (27.5%) would have been excluded under Toronto criteria and 18/58 (31%) under Hangzhou criteria. The recurrence risk (RR) for the UCSF and UCSF+ group was 13 and 36% (P = 0.1) (Fig. 1D). The RR was 26% and 24% for patients who met Toronto and Hangzhou criteria (Fig. 1E, F). When patients with AFP > 600 ng/ml (n = 4) were excluded from the UCSF+ group, RR was 27% (P = 0.3) (Fig. 2a). The RR was > 50% in patients with AFP > 600 ng/ml both in UCSF and UCSF + groups (Fig. 2b).
LDLT for macrovascular invasion
Median AFP in patients with MVI was 155 (16–702) ng/ml and AFP was < 100 ng/ml in 11 (40.8%) patients as shown in Table 3. The actual number of deaths in patients with an AFP ≤ or > 100 ng/ml was 4 (36.3%) and 13 (86.7%) (P = 0.04), respectively. On explant, the median largest tumor diameter was 5.5 (2.5–6) cm and tumor number was 2 (1–4). Figure 3 shows the survival in patients with MVI based on various prognostic groups. The estimated 5 year OS was 85% in the low-risk group and was not reached in the high-risk group (P = 0.003) (Fig. 4).
Management of recurrence
There were 27/217 (12.5%) recurrences in patients with HCC without MVI (supplementary table). Out of these, two patients had solitary hepatic recurrence. One of these patients was managed with TACE and sorafenib. The other patient underwent MWA and later required surgical excision of an isolated diaphragmatic recurrence three years after transplant. All other patients had recurrence involving multiple sites and were offered palliation with sorafenib. Three patients with painful bone metastases received palliative radiation. Table 3 shows the site of metastases in patients who underwent LDLT for HCC.
There were 5/27 (18.5%) recurrences in patients with HCC and MVI. All patients had recurrence involving multiple sites. One patient received palliative radiation while sorafenib was considered in all patients.
Discussion
A number of expanded transplant criteria have been proposed for HCC in the DDLT and LDLT setting (Duvoux et al. 2012; Mazzaferro et al. 2018; Sapisochin et al. 2016, Lee et al. 2008, Choi et al. 2017). Most of these lead to a modest expansion in transplant pool. A pre-transplant biopsy appears to be a pre-requisite for more liberal expansion on tumor size and number and excludes patients with aggressive biology (Sapisochin et al. 2016; Lei et al. 2014). Although with this approach, survival comparable to Milan and UCSF criteria might be achieved, the use of pre-transplant biopsy remains debatable in clinical practice. We used AFP < 1000 ng/ml instead of biopsy, in patients outside UCSF criteria for patient selection. AFP > 1000 ng/ml is associated with increased risk of post-transplant recurrence even with HCC fulfilling UCSF criteria and recently has been incorporated into United Network for Organ Sharing (UNOS) HCC staging for patient listing (DuBay et al. 2011; Hameed et al. 2014; Bhatti et al. 2020). The OS achieved with tumor size cutoff of 10 cm and AFP < 1000 ng/ml was comparable to UCSF, Toronto, and Hangzhou criteria. Consistent with our previous experience as well as that from other centers, AFP > 600 ng/ml was a poor prognostic factor for patients in the UCSF and UCSF+ group with > 50% RR (Wong et al. 2019; Bhatti et al. 2020). Although, RR was not significantly different in the UCSF and UCSF+ groups, it is likely that statistical significance was not reached due to relatively lower patient number in the UCSF+ group. Nevertheless, 5 year RR of 36% in the UCSF+ group is acceptable and could be further reduced with an AFP cutoff of 600 ng/ml. Late recurrences were more common in the UCSF+ group and might have contributed to similar OS in the two groups.
Tumor size > 10 cm is associated with high risk of MVI, poor differentiation, and metastases (Wu et al. 2018). In the context of LDLT, tumor size cutoff of 10 cm has been used in combination with PET scan for patient selection by NCCK (Lee et al. 2016). The sensitivity of PET scan for HCC varies with tumor grade and location of metastases and optimal SUV cutoffs to predict recurrence are yet to be established. Moreover, it needs technical skill and experience for interpretation, and remains a costly investigation (Lu et al. 2019). Nevertheless, PET scan has become an invaluable clinical investigation in the pre-transplant workup of HCC patients. The clinical utility of pre-transplant AFP in patient selection for transplantation is already well established (Wu et al. 2018; Halazun et al. 2017). AFP is easily available, reproducible, and cost-effective investigation. The dynamic nature of AFP enables effective decision-making in patients receiving various locoregional treatments in the pre-transplant setting (Bhatti et al. 2020). In patients with advanced HCC, AFP in combination with other biomarkers like AFP L-3 and PIVKAII might improve patient selection for curative treatments. An AFP L-3 < 35% and PIVKAII < 400 mAU/ml allows safe expansion on tumor size and number while HCC exceeding these cutoff might yield unacceptable results even in patients fulfilling traditional criteria (Chaiteerakij et al. 2015; Kaido et al. 2013).
Traditionally, MVI has been a relative contraindication to LDLT. However, in carefully selected patients with other positive prognostic features, LT can achieve acceptable long-term survival. Some of these factors include low AFP (10–100 ng/ml), good response to DS and Vp1-2 MVI (Assalino et al. 2020; Bhatti et al. 2020; Mehta et al. 2017; Lee et al. 2017). Our results are consistent with these reports and patient with favorable prognostic factors had acceptable outcomes. At our center, all patients with MVI undergo DS if feasible with an observation period of 4–6 months. We have shown that acceptable post-transplant survival is possible in a small group of carefully selected patients with Vp3 PVTT (Fig. 5). These patients have good response to DS based on mRECIST and an AFP < 100 ng/ml at the time of transplant. Patients with partial response, stable or progressive disease do poorly after LDLT. When DS is not feasible, patients with Vp1-2 MVI can be considered for upfront LDLT provided AFP at the time of transplant is < 100 ng/ml. Otherwise, outcome remains dismal and LT should be discouraged in patients with MVI.
Long-term survival (60 months) after LDLT with prior downstaging for macrovascular invasion + HCC. A and B Liver dynamic CT scan in a patient with multi focal HCC, largest tumor size 10 cm and right portal vein tumor thrombus (red arrows). C Post TACE × 4 scan 12 months later, showed significant response to treatment in segment 5 (green arrow) and reduction in portal vein expansion. D Re canalization of right portal vein with significant resolution of portal vein tumor thrombus (green arrow), histopathology confirmed a poorly differentiated necrotic tumor with foci of residual HCC
Our findings have prompted certain modifications in our protocol for LDLT in HCC. We routinely check PIVKAII levels in all patients with HCC. Patients with AFP > 1000 ng/ml, both in UCSF and UCSF+ groups undergo downstaging. All patients with MVI are evaluated with a staging PET scan. These patients are considered for downstaging and undergo LDLT only if good radiological response is documented or AFP drops below < 100 ng/ml. If not eligible for DS, patients with Vp1-2 MVI and low AFP are still considered for upfront LDLT.
The relatively small number of patients in the high-risk category (AFP > 1000 ng/ml or MVI) can be considered a limitation of the current study. We think it’s a substantial number as very few patients outside traditional transplant criteria, undergo LT with such tumor-related features (HCC 6.5–10 cm, any tumor number, MVI). The results of the current study are based exclusively upon LDLT experience and their application to DDLT is limited considering varying dynamics of prioritization and listing. Due to relatively small prognostic groups, a multivariate analysis to determine independent predictors of OS was not possible. Rates of LRT failure in our patients could not be reported since some patients were lost due to complex factors besides progressive liver failure and tumor burden. This, however, is not likely to have impacted outcomes in patients who eventually had LDLT for HCC.
The current study demonstrates acceptable post-transplant survival with more liberal expansion of cutoffs on tumor size and number with incorporation of AFP in the selection protocol. This approach makes it possible to consider LDLT even in selected patients with MVI. In the future, transplant criteria based on radiological and AFP response to LRT, and inclusion of biomarkers like AFP L-3 and PIVKAII might improve patient selection for LT.
Availability of data and material
The data are available from authors upon reasonable request.
Abbreviations
- AFP:
-
Alpha fetoprotein
- LDLT:
-
Living donor liver transplantation
- LRT:
-
Locoregional therapy
- mRECIST:
-
Modified response evaluation criteria in solid tumors
- MVI:
-
Macrovascular invasion
- MWA:
-
Microwave ablation
- RFA:
-
Radio frequency ablation
- PEI:
-
Percutaneous ethanol injection
- PIVKAII:
-
Prothrombin induced Vitamin K II antagonist
- PVTT:
-
Portal vein tumor thrombosis
- DDLT:
-
Deceased donor liver transplantation
- RR:
-
Recurrence risk
- TACE:
-
Trans arterial chemo embolization
- UCSF:
-
University of California San Francisco
References
Assalino M et al (2020) Liver transplantation for hepatocellular carcinoma after successful treatment of macrovascular invasion—a multi-center retrospective cohort study. Transpl Int 33(5):567–575. https://doi.org/10.1111/tri.13586
Bhatti ABH et al (2020) When to call it off: defining transplant candidacy limits in liver donor liver transplantation for hepatocellular carcinoma. BMC Cancer 20(1):754. https://doi.org/10.1186/s12885-020-07238-w
Bhatti ABH, Hassan M, Rana A, Khan NY, Khan ZA, Zia HH (2020a) Risk score model for liver transplant outcomes after preoperative locoregional therapy for hepatocellular carcinoma. J Gastrointest Cancer. https://doi.org/10.1007/s12029-020-00554-1
Bhatti ABH, Waheed A, Khan NA (2020b) Living donor liver transplantation for hepatocellular carcinoma: appraisal of the united network for organ sharing modified TNM staging. Front Surg. https://doi.org/10.3389/fsurg.2020.62217
Bruix J, Sherman M (2011) American Association for the study of liver diseases. management of hepatocellular carcinoma: an update. Hepatology 53:1020–1022
Chaiteerakij R et al (2015) Combinations of biomarkers and Milan criteria for predicting hepatocellular carcinoma recurrence after liver transplantation. Liver Transpl 21(5):599–606. https://doi.org/10.1002/lt.24117
Choi HJ et al (2017) The clinical outcomes of patients with portal vein tumor thrombi after living donor liver transplantation. Liver Transpl 23(8):1023–1031. https://doi.org/10.1002/lt.24782
Costentin CE, Bababekov YJ, Zhu AX, Yeh H (2019) Is It Time to Reconsider the Milan Criteria for selecting patients with hepatocellular carcinoma for deceased-donor liver transplantation? Hepatology 69(3):1324–1336. https://doi.org/10.1002/hep.30278
Dar FS et al (2015) The travails of setting up a living donor liver transplant program: experience from Pakistan and lessons learned. Liver Transpl 21(7):982–990. https://doi.org/10.1002/lt.24151
Dar FS et al (2018) Living donor liver transplantation in South Asia: single center experience on intermediate-term outcomes. World J Surg 42(4):1111–1119. https://doi.org/10.1007/s00268-017-4259-1
DuBay D et al (2011) Liver transplantation for advanced hepatocellular carcinoma using poor tumor differentiation on biopsy as an exclusion criterion. Ann Surg 253(1):166–172. https://doi.org/10.1097/sla.0b013e31820508f1
Duvoux C et al (2012) Liver Transplantation French Study Group. Liver transplantation for hepatocellular carcinoma: a model including α-fetoprotein improves the performance of Milan criteria. Gastroenterology 143(4):986–94.e3. https://doi.org/10.1053/j.gastro.2012.05.052 (quiz e14–5)
Halazun KJ et al (2017) Recurrence after liver transplantation for hepatocellular carcinoma: a new MORAL to the story. Ann Surg 265(3):557–564. https://doi.org/10.1097/SLA.0000000000001966
Hameed B, Mehta N, Sapisochin G, Roberts JP, Yao FY (2014) Alpha-fetoprotein level > 1000 ng/ml as an exclusion criterion for liver transplantation in patients with hepatocellular carcinoma meeting the Milan criteria. Liver Transpl 20(8):945–951. https://doi.org/10.1002/lt.23904
Hong SK, Lee KW, Kim HS, Yoon KC, Yi NJ, Suh KS (2016) Living donor liver transplantation for hepatocellular carcinoma in Seoul National University. Hepatobiliary Surg Nutr 5(6):453–460. https://doi.org/10.21037/hbsn.2016.08.07
Kaido T et al (2013) Usefulness of the Kyoto criteria as expanded selection criteria for liver transplantation for hepatocellular carcinoma. Surgery 154(5):1053–1060. https://doi.org/10.1016/j.surg.2013.04.056
Kanehara & Co Ltd (2003) The general rules for the clinical and pathological study of primary liver cancer, 2nd edn. Kanehara, Tokyo
Lee SG et al (2008) Expanded indication criteria of living donor liver transplantation for hepatocellular carcinoma at one large-volume center. Liver Transpl 14:935–945
Lee SD et al (2016) Proposal of new expanded selection criteria using total tumor size and (18)F-fluorodeoxyglucose—positron emission tomography/computed tomography for living donor liver transplantation in patients with hepatocellular carcinoma: the National Cancer Center Korea criteria. World J Transplant 6(2):411–422. https://doi.org/10.5500/wjt.v6.i2.411
Lee KW et al (2017) Macrovascular invasion is not an absolute contraindication for living donor liver transplantation. Liver Transpl 23(1):19–27. https://doi.org/10.1002/lt.24610
Lei JY, Wang WT, Yan LN (2014) Hangzhou criteria for liver transplantation in hepatocellular carcinoma: a single-center experience. Eur J Gastroenterol Hepatol 26(2):200–204. https://doi.org/10.1097/MEG.0b013e3283652b66
Lencioni R, Llovet JM (2010) Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis 30(1):52–60. https://doi.org/10.1055/s-0030-1247132
Lu RC et al (2019) Positron-emission tomography for hepatocellular carcinoma: Current status and future prospects. World J Gastroenterol 25(32):4682–4695. https://doi.org/10.3748/wjg.v25.i32.4682
Mazzaferro V et al (1996) Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med 334(11):693–699. https://doi.org/10.1056/NEJM199603143341104
Mazzaferro V et al (2018) Metroticket 2.0 model for analysis of competing risks of death after liver transplantation for hepatocellular carcinoma. Gastroenterology 154(1):128–139. https://doi.org/10.1053/j.gastro.2017.09.025
Mehta N et al (2017) Validation of a risk estimation of tumor recurrence after transplant (RETREAT) score for hepatocellular carcinoma recurrence after liver transplant. JAMA Oncol 3(4):493–500. https://doi.org/10.1001/jamaoncol.2016.5116
Sapisochin G et al (2016) The extended Toronto criteria for liver transplantation in patients with hepatocellular carcinoma: a prospective validation study. Hepatology 64(6):2077–2088. https://doi.org/10.1002/hep.28643
Sherman CB, Behr S, Dodge JL, Roberts JP, Yao FY, Mehta N (2019) Distinguishing tumor from bland portal vein thrombus in liver transplant candidates with hepatocellular carcinoma: the A-VENA criteria. Liver Transpl 25(2):207–216. https://doi.org/10.1002/lt.25345
Soin AS et al (2020) Experience with LDLT in patients with hepatocellular carcinoma and portal vein tumor thrombosis post downstaging. Transplantation 104(11):2334–2345. https://doi.org/10.1097/TP.0000000000003162
Sparchez Z, Mocan T (2018) Contemporary role of liver biopsy in hepatocellular carcinoma. World J Hepatol 10(7):452–461. https://doi.org/10.4254/wjh.v10.i7.452
Toso C, Mentha G, Majno P (2011) Liver transplantation for hepatocellular carcinoma: five steps to prevent recurrence. Am J Transplant 11(10):2031–2035. https://doi.org/10.1111/j.1600-6143.2011.03689.x
Wong TCL et al (2019) Long-term survival outcome between living donor and deceased donor liver transplant for hepatocellular carcinoma: intention-to-treat and propensity score matching analyses. Ann Surg Oncol 26(5):1454–1462. https://doi.org/10.1245/s10434-019-07206-0
Wu G, Wu J, Wang B, Zhu X, Shi X, Ding Y (2018) Importance of tumor size at diagnosis as a prognostic factor for hepatocellular carcinoma survival: a population-based study. Cancer Manag Res 10(10):4401–4410. https://doi.org/10.2147/CMAR.S177663
Yao FY et al (2001) Liver transplantation for hepatocellular carcinoma: expansion of the tumor size limits does not adversely impact survival. Hepatology 33(6):1394–1403. https://doi.org/10.1053/jhep.2001.24563
Yao FY, Xiao L, Bass NM, Kerlan R, Ascher NL, Roberts JP (2007) Liver transplantation for hepatocellular carcinoma: validation of the UCSF-expanded criteria based on preoperative imaging. Am J Transplant 7(11):2587–2596. https://doi.org/10.1111/j.1600-6143.2007.01965.x
Funding
No funding was received for this research. Aster life sciences agreed to fund the open access publication fee.
Author information
Authors and Affiliations
Contributions
AHB contributed to concept, design, analysis, writing and review of the manuscript. WN contributed to data collection, analysis, and writing. NYK, HHZ, FSD contributed to writing and critical review of the manuscript. ZAK, AR contributed to design and critical review of the manuscript. All authors agree to be accountable for the content of the work.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interests.
Ethics approval
The institutional review board and hospital ethics committee of Shifa International Hospital approved the study (IRB #433-1253-2020). The hospital ethics committee waived the requirement for written informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bhatti, A.B.H., Naqvi, W., Khan, N.Y. et al. Living donor liver transplantation for advanced hepatocellular carcinoma including macrovascular invasion. J Cancer Res Clin Oncol 148, 245–253 (2022). https://doi.org/10.1007/s00432-021-03665-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-021-03665-9