Abstract
Purpose
To review recent pharmacogenomics studies on breast cancer patients undergoing tamoxifen therapy, highlighting how our knowledge on cytochrome P450 2D6 (CYP2D6) can help to guide the development of adjuvant therapies for these patients.
Methods
A comprehensive literature search was conducted. Articles reporting findings pertaining to the effect of CYP2D6 on the therapeutic efficacy of tamoxifen, those reporting how targeting CYP2D6 could inform tamoxifen-based therapy development, and those on the tamoxifen effects on cell lines and animal models were included in the review.
Results
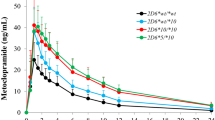
With CYP2D6 being the primary enzyme for tamoxifen metabolism, single-nucleotide polymorphisms (SNPs) in this gene were one of the determinants in the rate of tamoxifen metabolism, thereby potentially having an effect on the efficacy of tamoxifen-based therapies. Our review indicates the potential effectiveness of targeting these SNPs, including those for the CYP2D6*10 allele (c. 100C > T), in modifying the level of tamoxifen metabolism. These findings suggest the importance of pharmacogenomics research in our understanding of the efficacy of adjuvant therapies. However, the involvement of multiple enzymes in tamoxifen metabolism, dietary factors, ethnic differences in gene frequencies, and patients’ compliance to tamoxifen therapies in studies do present challenges in pharmacogenomics research.
Conclusions
Pharmacogenomics could play important roles in mediating the advancement of tamoxifen-based adjuvant therapies. Research efforts should be directed towards the exploration of further SNPs of CYP2D6 that affect tamoxifen metabolism, as well as epigenetic changes in CYP2D6, enabling the design of precision medicine and confirming clinical validity in the use of pharmacogenomics for tamoxifen.


Similar content being viewed by others
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Ahmed JH, Makonnen E, Fotoohi A, Aseffa A, Howe R, Aklillu E (2019) CYP2D6 genotype predicts plasma concentrations of tamoxifen metabolites in Ethiopian breast cancer patients. Cancers (Basel) 11:E1353. https://doi.org/10.3390/cancers11091353
American Cancer Society (2019) How common is breast cancer? https://www.cancer.org/cancer/breast-cancer/about/how-common-is-breast-cancer.html. Accessed 23 Mar 2020
Berthou F, Dreano Y, Belloc C, Kangas L, Gautier JC, Beaune P (1994) Involvement of cytochrome P450 3A enzyme family in the major metabolic pathways of toremifene in human liver microsomes. Biochem Pharmacol 47:1883–1895. https://doi.org/10.1016/0006-2952(94)90319-0
Binkhorst L, Mathijssen RH, Jager A, van Gelder T (2015) Individualization of tamoxifen therapy: much more than just CYP2D6 genotyping. Cancer Treat Rev 41:289–299. https://doi.org/10.1016/j.ctrv.2015.01.002
Bourel M, Ardaillou R (2006) Pharmacogenetics and pharmacogenomics. Bull Acad Natl Med 190:9–22
Brauch H, Schroth W, Mürdter T, Schwab M (2019) Tamoxifen pharmacogenetics and metabolism: the same is not the same. J Clin Oncol 37:1981–1982. https://doi.org/10.1200/JCO.19.00507
Chair SY, Waye MMY, Calzone K, Chan CWH (2019) Genomics education in nursing in Hong Kong, Taiwan and Mainland China. Int Nurs Rev 66:459–466. https://doi.org/10.1111/inr.12537
Chan CWH, Law BMH, So WKW, Chow KM, Waye MMY (2017) Novel strategies on personalized medicine for breast cancer treatment: an update. Int J Mol Sci 18:E2423. https://doi.org/10.3390/ijms18112423
Chang JH, Chen J, Liu L, Messick K, Ly J (2016) Rifampin-mediated induction of tamoxifen metabolism in a humanized PXR-CAR-CYP3A4/3A7-CYP2D6 mouse model. Drug Metab Dispos 44:1736–1741. https://doi.org/10.1124/dmd.116.072132
Chao TC, Pan WC, Tsai YF, Chou YC, Liu YR, Wang SF, Chen YJ, Souček P, Ueng YF (2019) Plasma endoxifen and 4-hydroxytamoxifen levels in CYP2D6(C100T) carrying breast cancer patients and association with serum cholesterol. Toxicol Appl Pharmacol 378:114619. https://doi.org/10.1016/j.taap.2019.114619
Chye GLC, Rampal S, Yahaya H (2008) Cancer Incidence in Peninsular Malaysia, 2003–2005. https://www.researchgate.net/publication/301682407_Cancer_Incidence_in_Peninsular_Malaysia_2003-2005. Accessed 23 Mar 2020
Cronin-Fenton DP, Damkier P (2018) Tamoxifen and CYP2D6: A CONTROVERSY IN PHARMacogenetics. Adv Pharmacol 83:65–91. https://doi.org/10.1016/bs.apha.2018.03.001
Dalton R, Lee SB, Claw KG, Prasad B, Phillips BR, Shen DD, Wong LH, Fade M, McDonald MG, Dunham MJ, Fowler DM, Rettie AE, Schuetz E, Thornton TA, Nickerson DA, Gaedigk A, Thummel KE, Woodahl EL (2019) Interrogation of CYP2D6 structural variant alleles improves the correlation between CYP2D6 genotype and CYP2D6-mediated metabolic activity. Clin Transl Sci 13:147–156. https://doi.org/10.1111/cts.12695
De Ameida MM, De Vasconcelos-Valença RJ, Neto FM, Borges RS, Costa-Silva DR, Da Conceição B-O, Borges US, Alencar AP, Silva VC, Da Silva BB (2016) CYP2D6 gene polymorphisms in Brazilian patients with breast cancer treated with adjuvant tamoxifen and its association with disease recurrence. Biomed Rep 5:574–578. https://doi.org/10.3892/br.2016.771
Dean L (2012) Tamoxifen therapy and CYP2D6 genotype. In: Pratt V, McLeod H, Dean L, Malheiro A, Rubinstein W (eds) Medical genetics summaries. National Center for Biotechnology Information (US), Bethesda
Goetz MP, Kamal A, Ames MM (2008) Tamoxifen pharmacogenomics: the role of CYP2D6 as a predictor of drug response. Clin Pharmacol Ther 83:160–166. https://doi.org/10.1038/sj.clpt.6100367
Goetz MP, Sun JX, Suman VJ, Silva GO, Perou CM, Nakamura Y, Cox NJ, Stephens PJ, Miller VA, Ross JS, Chen D, Safgren SL, Kuffel MJ, Ames MM, Kalari KR, Gomez HL, Gonzalez-Angulo AM, Burgues O, Brauch HB, Ingle JN, Ratain MJ, Yelensky R (2014) Loss of heterozygosity at the CYP2D6 locus in breast cancer: implications for germline pharmacogenetic studies. J Natl Cancer Inst 107:401. https://doi.org/10.1093/jnci/dju401
Goetz MP, Sangkuhl K, Guchelaar HJ, Schwab M, Province M, Whirl-Carrillo M, Symmans WF, McLeod HL, Ratain MJ, Zembutsu H, Gaedigk A, van Schaik RH, Ingle JN, Caudle KE, Klein TE (2018) Clinical pharmacogenetics implementation consortium (CPIC) guideline for CYP2D6 and tamoxifen therapy. Clin Pharmacol Ther 103:770–777. https://doi.org/10.1002/cpt.1007
Hannouf MB, Zaric GS, Blanchette P, Brezden-Masley C, Paulden M, McCabe C, Raphael J, Brackstone M (2020) Cost-effectiveness analysis of multigene expression profiling assays to guide adjuvant therapy decisions in women with invasive early-stage breast cancer. Pharmacogenomics J 20:27–46. https://doi.org/10.1038/s41397-019-0089-x
Hansten PD (2018) The underrated risks of tamoxifen drug interactions. Eur J Drug Metab Pharmacokinet 43:495–508. https://doi.org/10.1007/s13318-018-0475-9
Harris LN, Ismaila N, McShane LM, Andre F, Collyar DE, Gonzalez-Angulo AM, Hammond EH, Kuderer NM, Liu MC, Mennel RG, Van Poznak C, Bast RC, Hayes DF (2016) Use of biomarkers to guide decisions on adjuvant systemic therapy for women with early-stage invasive breast cancer: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol 34:1134–1150. https://doi.org/10.1200/JCO.2015.65.2289
Hennig EE, Piątkowska M, Goryca K, Pośpiech E, Paziewska A, Karczmarski J, Kluska A, Brewczyńska E, Ostrowski J (2019) Non-CYP2D6 variants selected by a GWAS improve the prediction of impaired tamoxifen metabolism in patients with breast cancer. J Clin Med 8:E1087. https://doi.org/10.3390/jcm8081087
Hong Kong Cancer Registry (2019) Female Breast Cancer in 2017. https://www3.ha.org.hk/cancereg/pdf/factsheet/2017/breast_2017.pdf. Accessed 23 Mar 2020
Hussaarts KGAM, Hurkmans DP, Oomen-de Hoop E, van Harten LJ, Berghuis S, van Alphen RJ, Spierings LEA, van Rossum-Schornagel QC, Vastbinder MB, van Schaik RHN, van Gelder T, Jager A, van Leeuwen RWF, Mathijssen RHJ (2019) Impact of curcumin (with or without Piperine) on the pharmacokinetics of tamoxifen. Cancers (Basel) 11:E403. https://doi.org/10.3390/cancers11030403
Hwang GS, Bhat R, Crutchley RD, Trivedi MV (2018) Impact of CYP2D6 polymorphisms on endoxifen concentrations and breast cancer outcomes. Pharmacogenomics J 18:201–208. https://doi.org/10.1038/tpj.2017.36
Ishiguro H, Ohno S, Yamamoto Y, Takao S, Sato N, Fujisawa T, Kadoya T, Kuroi K, Bando H, Teramura Y, Iwata H, Tanaka S, Toi M (2019) Pharmacogenomic-pharmacokinetic study of selective estrogen-receptor modulators with intra-patient dose escalation in breast cancer. Breast Cancer 26:535–543. https://doi.org/10.1007/s12282-019-00952-9
Jin Y, Desta Z, Stearns V, Ward B, Ho H, Lee KH, Skaar T, Storniolo AM, Li L, Araba A, Blanchard R, Nguyen A, Ullmer L, Hayden J, Lemler S, Weinshilboum RM, Rae JM, Hayes DF, Flockhart DA (2005) CYP2D6 genotype, antidepressant use, and tamoxifen metabolism during adjuvant breast cancer treatment. J Natl Cancer Inst 97:30–39. https://doi.org/10.1093/jnci/dji005
Joffe H, Deckersbach T, Lin NU, Makris N, Skaar TC, Rauch SL, Dougherty DD, Hall JE (2012) Metabolic activity in the insular cortex and hypothalamus predicts hot flashes: an FDG-PET study. J Clin Endocrinol Metab 97:3207–3215. https://doi.org/10.1210/jc.2012-1413
LLerena A, Naranjo ME, Rodrigues-Soares F, Penas-LLedó EM, Fariñas H, Tarazona-Santos E (2014) Interethnic variability of CYP2D6 alleles and of predicted and measured metabolic phenotypes across world populations. Expert Opin Drug Metab Toxicol 10:1569–1583. https://doi.org/10.1517/17425255.2014.964204
Lohmann DR, Gallie BL (2004) Retinoblastoma: revisiting the model prototype of inherited cancer. Am J Med Genet C Semin Med Genet 129C:23–28. https://doi.org/10.1002/ajmg.c.30024
López-Camarillo C, Gallardo-Rincón D, Álvarez-Sánchez ME, Marchat LA (2019) Pharmaco-epigenomics: On the Road of Translation Medicine. Adv Exp Med Biol 1168:31–42. https://doi.org/10.1007/978-3-030-24100-1_3
Lu WJ, Xu C, Pei Z, Mayhoub AS, Cushman M, Flockhart DA (2012) The tamoxifen metabolite norendoxifen is a potent and selective inhibitor of aromatase (CYP19) and a potential lead compound for novel therapeutic agents. Breast Cancer Res Treat 133:99–109. https://doi.org/10.1007/s10549-011-1699-4
Lu J, Li H, Guo P, Shen R, Luo Y, Ge Q, Shi W, Li Y, Zhu W (2017) The effect of CYP2D6 *10 polymorphism on adjuvant tamoxifen in Asian breast cancer patients: a meta-analysis. Onco Targets Ther 10:5429–5437. https://doi.org/10.2147/OTT.S149197
Luzum JA, Pakyz RE, Elsey AR et al (2017) The pharmacogenomics research network translational pharmacogenetics program: outcomes and metrics of pharmacogenetic implementations across diverse healthcare systems. Clin Pharmacol Ther 102:502–510. https://doi.org/10.1002/cpt.630
MacLeod AK, McLaughlin LA, Henderson CJ, Wolf CR (2017) Application of mice humanized for CYP2D6 to the study of tamoxifen metabolism and drug–drug interaction with antidepressants. Drug Metab Dispos 45:17–22. https://doi.org/10.1124/dmd.116.073437
Martins DM, Vidal FC, Souza RD, Brusaca SA, Brito LM (2014) Determination of CYP2D6 *3, *4, and *10 frequency in women with breast cancer in São Luís, Brazil, and its association with prognostic factors and disease-free survival. Braz J Med Biol Res 47:1008–1015. https://doi.org/10.1590/1414-431X20143761
Mayer SE, Weiss NS, Chubak J, Doody DR, Carlson CS, Makar KW, Wurscher MA, Malone KE (2019) CYP2D6-inhibiting medication use and inherited CYP2D6 variation in relation to adverse breast cancer outcomes after tamoxifen therapy. Cancer Causes Control 30:103–112. https://doi.org/10.1007/s10552-018-1117-x
Nardin JM, Schroth W, Almeida TA, Mürdter T, Picolotto S, Vendramini ECL, Hoppe R, Kogin JP, Miqueleto D, de Moraes SDR, Schwab M, Pecoits-Filho RF, Brauch H, Casali-da-Rocha JC (2019) The influences of adherence to tamoxifen and CYP2D6 pharmacogenetics on plasma concentrations of the active metabolite (Z)-endoxifen in breast cancer. Clin Transl Sci 13:284–292. https://doi.org/10.1111/cts.12707
Province MA, Goetz MP, Brauch H et al (2014) CYP2D6 genotype and adjuvant tamoxifen: meta-analysis of heterogeneous study populations. Clin Pharmacol Ther 95:216–227. https://doi.org/10.1038/clpt.2013.186
Qin T, Yuan ZY, Peng RJ, Zeng YD, Shi YX, Teng XY, Liu DG, Bai B, Wang SS (2013) Efficacy and tolerability of toremifene and tamoxifen therapy in premenopausal patients with operable breast cancer: a retrospective analysis. Curr Oncol 20:196–204. https://doi.org/10.3747/co.20.1231
Rae JM, Sikora MJ, Henry NL, Li L, Kim S, Oesterreich S, Skaar TC, Nguyen AT, Desta Z, Storniolo AM, Flockhart DA, Hayes DF, Stearns V, COBRA Investigators (2009) Cytochrome P450 2D6 activity predicts discontinuation of tamoxifen therapy in breast cancer patients. Pharmacogenomics J 9:258–264. https://doi.org/10.1038/tpj.2009.14
Raju KS, Taneja I, Valicherla GR, Challagundla MK, Rashid M, Syed AA, Gayen JR, Singh SP, Wahajuddin M (2015) No effect on pharmacokinetics of tamoxifen and 4-hydroxytamoxifen by multiple doses of red clover capsule in rats. Sci Rep 5:16126. https://doi.org/10.1038/srep16126
Reis SS, Carvalho AS, Fernandes R (2019) Pharmacogenomics, CYP2D6, and tamoxifen: a survey of the reasons sustaining European clinical practice paradigms. Medicina (Kaunas) 55:344. https://doi.org/10.3390/medicina55070344
Relling MV, Klein TE (2011) CPIC: clinical pharmacogenetics implementation consortium of the pharmacogenomics research network. Clin Pharmacol Ther 89:464–467. https://doi.org/10.1038/clpt.2010.279
Rodrigues-Soares F, Suarez-Kurtz G (2019) Pharmacogenomics research and clinical implementation in Brazil. Basic Clin Pharmacol Toxicol 124:538–549. https://doi.org/10.1111/bcpt.13196
Rodrigues-Soares F, Peñas-Lledó EM, Tarazona-Santos E, Sosa-Macías M, Terán E, López-López M, Rodeiro I, Moya GE, Calzadilla LR, Ramírez-Roa R, Grazina M, Estévez-Carrizo FE, Barrantes R, LLerena A, RIBEF Ibero-American Network of Pharmacogenetics, and Pharmacogenomics (2020) Genomic ancestry, CYP2D6, CYP2C9, and CYP2C19 among Latin Americans. Clin Pharmacol Ther 107:257–268. https://doi.org/10.1002/cpt.1598
Sanchez-Spitman AB, Swen JJ, Dezentje VO, Moes DJAR, Gelderblom H, Guchelaar HJ (2019a) Clinical pharmacokinetics and pharmacogenetics of tamoxifen and endoxifen. Expert Rev Clin Pharmacol 12:523–536. https://doi.org/10.1080/17512433.2019.1610390
Sanchez-Spitman A, Dezentjé V, Swen J, Moes DJAR, Böhringer S, Batman E, van Druten E, Smorenburg C, van Bochove A, Zeillemaker A, Jongen L, Los M, Neven P, Gelderblom H, Guchelaar HJ (2019b) Tamoxifen pharmacogenetics and metabolism: results from the prospective CYPTAM study. J Clin Oncol 37:636–646. https://doi.org/10.1200/JCO.18.00307
Schroth W, Antoniadou L, Fritz P, Schwab M, Muerdter T, Zanger UM, Simon W, Eichelbaum M, Brauch H (2007) Breast cancer treatment outcome with adjuvant tamoxifen relative to patient CYP2D6 and CYP2C19 genotypes. J Clin Oncol 25:5187–5193. https://doi.org/10.1200/JCO.2007.12.2705
Schroth W, Goetz MP, Hamann U, Fasching PA, Schmidt M, Winter S, Fritz P, Simon W, Suman VJ, Ames MM, Safgren SL, Kuffel MJ, Ulmer HU, Boländer J, Strick R, Beckmann MW, Koelbl H, Weinshilboum RM, Ingle JN, Eichelbaum M, Schwab M, Brauch H (2009) Association between CYP2D6 polymorphisms and outcomes among women with early stage breast cancer treated with tamoxifen. JAMA 302:1429–1436. https://doi.org/10.1001/jama.2009.1420
Thompson AM, Johnson A, Quinlan P, Hillman G, Fontecha M, Bray SE, Purdie CA, Jordan LB, Ferraldeschi R, Latif A, Hadfield KD, Clarke RB, Ashcroft L, Evans DG, Howell A, Nikoloff M, Lawrence J, Newman WG (2011) Comprehensive CYP2D6 genotype and adherence affect outcome in breast cancer patients treated with tamoxifen monotherapy. Breast Cancer Res Treat 125:279–287. https://doi.org/10.1007/s10549-010-1139-x
Wang G, Qin S, Zayas J, Ingle JN, Liu M, Weinshilboum RM, Shen K, Wang L (2019) 4-Hydroxytamoxifen enhances sensitivity of estrogen receptor α-positive breast cancer to docetaxel in an estrogen and ZNF423 SNP-dependent fashion. Breast Cancer Res Treat 175:567–578. https://doi.org/10.1007/s10549-019-05194-z
Warchal SJ, Dawson JC, Shepherd E, Munro AF, Hughes RE, Makda A, Carragher NO (2020) High content phenotypic screening identifies serotonin receptor modulators with selective activity upon breast cancer cell cycle and cytokine signaling pathways. Bioorg Med Chem 28:115209. https://doi.org/10.1016/j.bmc.2019.115209
Wei X, Cai J, Zhuang J, Zheng B, Sui Y, Zhang G, Lin Y, Sun H (2020) CYP2D6*10 pharmacogenetic-guided SERM could be a cost-effective strategy in Chinese patients with hormone receptor-positive breast cancer. Pharmacogenomics 21:43–53. https://doi.org/10.2217/pgs-2019-0073
Wei X, Sun H, Zhuang J, Weng X, Zheng B, Lin Q, Zhang G, Cai J (2019) Cost-effectiveness analysis of CYP2D6*10 pharmacogenetic testing to guide the adjuvant endocrine therapy for postmenopausal women with estrogen receptor positive early breast cancer in China. Clin Drug Investig 40:25–32. https://doi.org/10.1007/s40261-019-00842-0
Wellmann R, Borden BA, Danahey K, Nanda R, Polite BN, Stadler WM, Ratain MJ, O'Donnell PH (2018) Analyzing the clinical actionability of germline pharmacogenomic findings in oncology. Cancer 124:3052–3065. https://doi.org/10.1002/cncr.31382
Wheeler SB, Spencer J, Pinheiro LC, Murphy CC, Earp JA, Carey L, Olshan A, Tse CK, Bell ME, Weinberger M, Reeder-Hayes KE (2019) Endocrine therapy nonadherence and discontinuation in black and white women. J Natl Cancer Inst 111:498–508. https://doi.org/10.1093/jnci/djy136
Yenny PSS, Purwanto DJ, Hidayat A, Louisa M, Andalusia R, Setiabudy R (2019) Association of CYP2D6*10 (c. 100 C%3eT) Genotype with Z-END Concentration in Patients with Breast Cancer Receiving Tamoxifen Therapy in Indonesian Population. Endocr Metab Immune Disord Drug Targets 19:1198–1206. https://doi.org/10.2174/1871530319666190306094617
Yin SJ, Ni YB, Wang SM, Wang X, Lou YQ, Zhang GL (2012) Differences in genotype and allele frequency distributions of polymorphic drug metabolizing enzymes CYP2C19 and CYP2D6 in mainland Chinese Mongolian, Hui and Han populations. J Clin Pharm Ther 37:364–369. https://doi.org/10.1111/j.1365-2710.2011.01298.x
Yokoyama Y, Sasaki Y, Terasaki N, Kawataki T, Takekawa K, Iwase Y, Shimizu T, Sanoh S, Ohta S (2018) Comparison of drug metabolism and its related hepatotoxic effects in HepaRG, cryopreserved human hepatocytes, and HepG2 cell cultures. Biol Pharm Bull 41:722–732. https://doi.org/10.1248/bpb.b17-00913
Yu J, Zayas J, Qin B, Wang L (2019) Targeting DNA methylation for treating triple-negative breast cancer. Pharmacogenomics 20:1151–1157. https://doi.org/10.2217/pgs-2019-0078
Zembutsu H, Sasa M, Kiyotani K, Mushiroda T, Nakamura Y (2011) Should CYP2D6 inhibitors be administered in conjunction with tamoxifen? Expert Rev Anticancer Ther 11:185–193. https://doi.org/10.1586/era.10.228
Zhang C, Zhong Q, Zhang Q, Zheng S, Miele L, Wang G (2015) Boronic prodrug of endoxifen as an effective hormone therapy for breast cancer. Breast Cancer Res Treat 152:283–291. https://doi.org/10.1007/s10549-015-3461-9
Zhong Q, Zhang C, Zhang Q, Miele L, Zheng S, Wang G (2015) Boronic prodrug of 4-hydroxytamoxifen is more efficacious than tamoxifen with enhanced bioavailability independent of CYP2D6 status. BMC Cancer 15:625. https://doi.org/10.1186/s12885-015-1621-2
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
CWHC and MMYW set the aim and focus of the review. MMYW drafted the manuscript. CWHC, BMHL, WKWS, and KMC critically reviewed and revised the manuscript. All authors had read and approved the final version of the manuscript for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that no conflicts of interest exist.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chan, C.W.H., Law, B.M.H., So, W.K.W. et al. Pharmacogenomics of breast cancer: highlighting CYP2D6 and tamoxifen. J Cancer Res Clin Oncol 146, 1395–1404 (2020). https://doi.org/10.1007/s00432-020-03206-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-020-03206-w