Abstract
Purpose
Anlotinib is a novel multi-target tyrosine kinase inhibitor (TKI) for tumor angiogenesis and tumor cell proliferation. The efficacy of anlotinib as a third-line or beyond therapy for SCLC was confirmed in the ALTER1202 trial. For lung cancer patients treated with antiangiogenesis agents, the phenomenon of cavitation is commonly seen in the lung target lesions. The impact of tumor cavitation on survival in lung cancer patients treated with vascular-targeted therapy remains controversial. Our retrospective study was to investigate the prognostic value of tumor cavitation in extensive-stage SCLC patients treated with anlotinib.
Methods
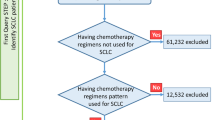
A total of 73 extensive-stage SCLC patients confirmed by histopathology from January 2018 to January 2019 were retrospectively analyzed. All patients received anlotinib therapy at Shanghai Chest Hospital. We defined tumor cavitation of the lung target lesions as that part of solid component was changed to air-filled area according to chest CT. Progression-free survival (PFS) was calculated from the beginning of anlotinib therapy to the disease progression or the last follow-up visit.
Results
Eleven (15.0%) patients had tumor cavitation during anlotinib therapy. The ORR of the 73 patients was 15.1%. Multivariate logistic regression analysis showed that tumor cavitation during anlotinib therapy was not associated with gender (P = 0.630), age (P = 0.190), smoking status (P = 0.165), anatomy type (P = 0.641), and the line of anlotinib therapy (P = 0.302). The median PFS of all patients was 2.6 months (95%CI 2.1–3.2). According to the univariate analysis, the median PFS in patients with and without tumor cavitation was 5.0 months and 2.2 months, respectively, and the difference was statistically significant (P = 0.041). According to the multivariate analysis, tumor cavitation was an independent factor for PFS after adjusting gender, age, smoking status, anatomy type, the line of anlotinib therapy, tumor cavitation, and response to anlotinib (adjusted HR 0.316, 95%CI 0.142–0.702; P = 0.005).
Conclusions
In 73 extensive-stage SCLC patients treated with anlotinib, no demographic/clinical characteristic was related to tumor cavitation, and tumor cavitation was an independent factor in predicting better PFS.


Similar content being viewed by others
Availability of data and materials
The data sets used during the present study are available from the corresponding author upon reasonable request.
References
Asai N, Ohkuni Y, Kaneko N, Yamaguchi E, Kubo A (2014) Relapsed small cell lung cancer: treatment options and latest developments. Ther Adv Med Oncol 6:69–82. https://doi.org/10.1177/1758834013517413
Byers LA, Rudin CM (2015) Small cell lung cancer: where do we go from here? Cancer 121:664–672. https://doi.org/10.1002/cncr.29098
Crabb SJ et al (2009) Tumor cavitation: impact on objective response evaluation in trials of angiogenesis inhibitors in non-small-cell lung cancer. J Clin Oncol 27:404–410. https://doi.org/10.1200/JCO.2008.16.2545
Gong J, Salgia R (2018) Managing patients with relapsed small-cell lung cancer. J Oncol Pract 14:359–366. https://doi.org/10.1200/JOP.18.00204
Han B et al (2018) Effect of anlotinib as a third-line or further treatment on overall survival of patients with advanced non-small cell lung cancer: the ALTER 0303 phase 3 randomized clinical trial. JAMA Oncol 4:1569–1575. https://doi.org/10.1001/jamaoncol.2018.3039
Huang C et al (2008) Analysis of cavitation of advanced NSCLC treated by rh-endostatin combined with NP chemotherapy. Zhonghua Zhong Liu Za Zhi 30:712–715
Huang C et al (2014) Incidence and clinical implication of tumor cavitation in patients with advanced non-small cell lung cancer induced by Endostar, an angiogenesis inhibitor. Thorac Cancer 5:438–446. https://doi.org/10.1111/1759-7714.12115
Kahnert K, Kauffmann-Guerrero D, Huber RM (2016) SCLC—state of the art and what does the future have in store? Clin Lung Cancer 17:325–333. https://doi.org/10.1016/j.cllc.2016.05.014
Kalemkerian GP et al (2013) Small cell lung cancer. J Natl Compr Canc Netw 11:78–98
Kelly RJ, Hnatiuk O, Giaccone G (2009) Antiangiogenesis induced tumor cavitation in lung cancer. J Thorac Oncol 4:1573–1575. https://doi.org/10.1097/JTO.0b013e3181c09fc2
Lee HY et al (2010) Molecularly targeted therapy using bevacizumab for non-small cell lung cancer: a pilot study for the new CT response criteria Korean. J Radiol 11:618–626. https://doi.org/10.3348/kjr.2010.11.6.618
Marom EM et al (2008) Tumor cavitation during therapy with antiangiogenesis agents in patients with lung cancer. J Thorac Oncol 3:351–357. https://doi.org/10.1097/JTO.0b013e318168c7e9
Nishino M et al (2012) Tumoral cavitation in patients with non-small-cell lung cancer treated with antiangiogenic therapy using bevacizumab. Cancer Imaging 12:225–235. https://doi.org/10.1102/1470-7330.2012.0027
Pietanza MC et al (2012) Phase II trial of temozolomide in patients with relapsed sensitive or refractory small cell lung cancer, with assessment of methylguanine-DNA methyltransferase as a potential biomarker. Clin Cancer Res 18:1138–1145. https://doi.org/10.1158/1078-0432.CCR-11-2059
Pietanza MC, Byers LA, Minna JD, Rudin CM (2015) Small cell lung cancer: will recent progress lead to improved outcomes? Clin Cancer Res 21:2244–2255. https://doi.org/10.1158/1078-0432.CCR-14-2958
Shen G et al (2018) Anlotinib: a novel multi-targeting tyrosine kinase inhibitor in clinical development. J Hematol Oncol 11:120. https://doi.org/10.1186/s13045-018-0664-7
Simos D et al (2014) Third-line chemotherapy in small-cell lung cancer: an international analysis. Clin Lung Cancer 15:110–118. https://doi.org/10.1016/j.cllc.2013.11.003
Smit EF, Fokkema E, Biesma B, Groen HJ, Snoek W, Postmus PE (1998) A phase II study of paclitaxel in heavily pretreated patients with small-cell lung cancer. Br J Cancer 77:347–351. https://doi.org/10.1038/bjc.1998.54
van der Lee I, Smit EF, van Putten JW, Groen HJ, Schlosser NJ, Postmus PE, Schramel FM (2001) Single-agent gemcitabine in patients with resistant small-cell lung cancer. Ann Oncol 12:557–561. https://doi.org/10.1023/a:1011104509759
Yang S, Zhang Z, Wang Q (2019) Emerging therapies for small cell lung cancer. J Hematol Oncol 12:47. https://doi.org/10.1186/s13045-019-0736-3
Acknowledgements
We would like to thank all of the investigators for their involvement in this study.
Funding
This work was supported by the Natural Science Foundation of Shanghai (19ZR1449700) and Western Medicine Guide Project of Shanghai Committee of Science and Technology (16411964700).
Author information
Authors and Affiliations
Contributions
All authors designed the study. Dongfang Chen collected the data. Jianlin Xu analyzed the data. Yizhuo Zhao and Tianqing Chu interpreted the results. Dongfang Chen drafted the manuscript with critical revisions from Hua Zhong, Baohui Han, Runbo Zhong. All authors approved the final version.
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare that there is no conflict of interest.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Ethical approval
The study protocol was approved by the Ethics Committee of Shanghai Chest Hospital and was conducted in accordance with the Helsinki Declaration of 1964 (revised 2008).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Dongfang Chen and Jianlin Xu are first and co-first authors.
Rights and permissions
About this article
Cite this article
Chen, D., Xu, J., Zhao, Y. et al. Prognostic value of tumor cavitation in extensive-stage small-cell lung cancer patients treated with anlotinib. J Cancer Res Clin Oncol 146, 401–406 (2020). https://doi.org/10.1007/s00432-019-03064-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-019-03064-1