Abstract
Purpose
Nodal positive lung adenocarcinoma includes wide range of survival. Several methods for the classification of nodal-positive lung cancer have been proposed. However, classification considering the impact of targetable genetic variants are lacking. The possibility of genetic variants for the better stratification of nodal positive lung adenocarcinoma was estimated.
Methods
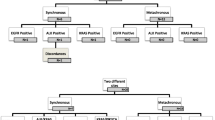
Mutations of 36 genes between primary sites and metastatic lymph nodes (LNs) were compared using next-generation sequencing. Subsequently, mutations in EGFR and BRAF, rearrangements in ALK and ROS1 were evaluated in 69 resected pN1–2M0 adenocarcinoma cases. Recurrence-free survival (RFS), post-recurrence survival (PRS), and overall survival (OS) were evaluated with respect to targetable variants and tyrosine kinase inhibitor (TKI) therapy after recurrence.
Results
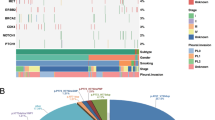
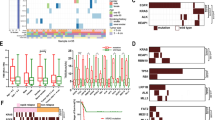
About 90% of variants were shared and allele frequencies were similar between primary and metastatic sites. In 69 pN1–2M0 cases, EGFR/ALK were positive in primary sites of 39 cases and same EGFR/ALK variants were confirmed in metastatic LNs of 96.7% tissue-available cases. Multivariate analyses indicated positive EGFR/ALK status was associated with worse RFS (HR 2.366; 95% CI 1.244–4.500; P = 0.009), and PRS was prolonged in cases receiving TKI therapy (no post-recurrence TKI therapies, HR 3.740; 95% CI 1.449–9.650; P = 0.006). OS did not differ with respect to targetable variants or TKI therapy.
Conclusions
Cases harbouring targetable genetic variants had a higher risk of recurrence, but PRS was prolonged by TKI therapy. Classification according to the targetable genetic status provides a basis for predicting recurrence and determining treatment strategies after recurrence.



Similar content being viewed by others
References
Camidge DR, Kim HR, Ahn MJ, Yang JC, Han JY, Lee JS et al (2018) Brigatinib versus crizotinib in ALK-positive non-small-cell lung cancer. N Engl J Med 379:2027–2039. https://doi.org/10.1056/NEJMoa1810171
Chaft JE, Dagogo-Jack I, Santini FC, Eng J, Yeap BY, Izar B et al (2018) Clinical outcomes of patients with resected, early-stage ALK-positive lung cancer. Lung Cancer 122:67–71. https://doi.org/10.1016/j.lungcan.2018.05.020
Cortot AB, Italiano A, Burel-Vandenbos F, Martel-Planche G, Hainaut P (2010) KRAS mutation status in primary nonsmall cell lung cancer and matched metastases. Cancer 116:2682–2687. https://doi.org/10.1002/cncr.25014
Gao Q, Li P, Jiang X, Zhan Z, Yan Q, Zhang B et al (2017) Worse disease-free, tumor-specific, and overall survival in surgically-resected lung adenocarcinoma patients with ALK rearrangement. Oncotarget 8:86066–86081. https://doi.org/10.18632/oncotarget.20973
Ito M, Yamashita Y, Tsutani Y, Misumi K, Harada H, Miyata Y et al (2013) Classifications of n2 non-small-cell lung cancer based on the number and rate of metastatic mediastinal lymph nodes. Clin Lung Cancer 14:651–657. https://doi.org/10.1016/j.cllc.2013.04.012
Ito M, Miyata Y, Hirano S, Kimura S, Irisuna F, Ikeda K et al (2017) Therapeutic strategies and genetic profile comparisons in small cell carcinoma and large cell neuroendocrine carcinoma of the lung using next-generation sequencing. Oncotarget 8:108936–108945. https://doi.org/10.18632/oncotarget.22426
Ito M, Miyata Y, Kushitani K, Yoshiya T, Kai Y, Tsutani Y et al (2018) Increased risk of recurrence in resected EGFR-positive pN0M0 invasive lung adenocarcinoma. Thorac Cancer 9:1594–1602. https://doi.org/10.1111/1759-7714.12866
Jeon JH, Kang CH, Kim HS, Seong YW, Park IK, Kim YT (2015) Prognostic and predictive role of epidermal growth factor receptor mutation in recurrent pulmonary adenocarcinoma after curative resection. Eur J Cardiothorac Surg 47:556–562. https://doi.org/10.1093/ejcts/ezu177
Jonnalagadda S, Smith C, Mhango G, Wisnivesky JP (2011) The number of lymph node metastases as a prognostic factor in patients with N1 non-small cell lung cancer. Chest 140:433–440. https://doi.org/10.1378/chest.10-2885
Kalemkerian GP, Narula N, Kennedy EB, Biermann WA, Donington J, Leighl NB et al (2018) Molecular testing guideline for the selection of patients with lung cancer for treatment with targeted tyrosine kinase inhibitors: American Society of Clinical Oncology endorsement of the College of American Pathologists/International Association for the Study of Lung Cancer/Association for Molecular Pathology Clinical Practice Guideline Update. J Clin Oncol 36:911–919. https://doi.org/10.1200/JCO.2017.76.7293
Kalikaki A, Koutsopoulos A, Trypaki M, Souglakos J, Stathopoulos E, Georgoulias V et al (2008) Comparison of EGFR and K-RAS gene status between primary tumours and corresponding metastases in NSCLC. Br J Cancer 99:923–929. https://doi.org/10.1038/sj.bjc.6604629
Lee GD, Kim DK, Moon DH, Joo S, Hwang SK, Choi SH et al (2016) A comparison of the proposed classifications for the revision of N descriptors for non-small-cell lung cancer. Eur J Cardiothorac Surg 49:580–588. https://doi.org/10.1093/ejcts/ezv134
Matsumoto S, Takahashi K, Iwakawa R, Matsuno Y, Nakanishi Y, Kohno T et al (2006) Frequent EGFR mutations in brain metastases of lung adenocarcinoma. Int J Cancer 119:1491–1494. https://doi.org/10.1002/ijc.21940
Nagai Y, Miyazawa H, Huqun Tanaka T, Udagawa K, Kato M et al (2005) Genetic heterogeneity of the epidermal growth factor receptor in non-small cell lung cancer cell lines revealed by a rapid and sensitive detection system, the peptide nucleic acid-locked nucleic acid PCR clamp. Cancer Res 65:7276–7282. https://doi.org/10.1158/0008-5472.CAN-05-0331
Park S, Holmes-Tisch AJ, Cho EY, Shim YM, Kim J, Kim HS et al (2009) Discordance of molecular biomarkers associated with epidermal growth factor receptor pathway between primary tumors and lymph node metastasis in non-small cell lung cancer. J Thorac Oncol 4:809–815. https://doi.org/10.1097/JTO.0b013e3181a94af4
Rami-Porta R, Call S, Dooms C, Obiols C, Sánchez M, Travis WD et al (2018) Lung cancer staging: a concise update. Eur Respir J 17:1800190. https://doi.org/10.1183/13993003.00190-2018
Rusch VW, Crowley J, Giroux DJ, Goldstraw P, Im JG, Tsuboi M et al (2007) The IASLC Lung Cancer Staging Project: proposals for the revision of the N descriptors in the forthcoming seventh edition of the TNM classification for lung cancer. J Thorac Oncol 2:603–612. https://doi.org/10.1097/JTO.0b013e31807ec803
Rusch VW, Asamura H, Watanabe H, Giroux DJ, Rami-Porta R, Goldstraw P et al (2009) The IASLC Lung Cancer Staging Project: a proposal for a new international lymph node map in the forthcoming seventh edition of the TNM classification for lung cancer. J Thorac Oncol 4:568–577. https://doi.org/10.1097/JTO.0b013e3181a0d82e
Saji H, Tsuboi M, Yoshida K, Kato Y, Nomura M, Matsubayashi J et al (2011) Prognostic impact of number of resected and involved lymph nodes at complete resection on survival in non-small cell lung cancer. J Thorac Oncol 6:1865–1871. https://doi.org/10.1097/JTO.0b013e31822a35c3
Schmid K, Oehl N, Wrba F, Pirker R, Pirker C, Filipits M (2009) EGFR/KRAS/BRAF mutations in primary lung adenocarcinomas and corresponding locoregional lymph node metastases. Clin Cancer Res 15:4554–4560. https://doi.org/10.1158/1078-0432.CCR-09-0089
Shimada Y, Saji H, Yoshida K, Kakihana M, Honda H, Nomura M et al (2013) Prognostic factors and the significance of treatment after recurrence in completely resected stage I non-small cell lung cancer. Chest 143:1626–1634. https://doi.org/10.1378/chest.12-1717
Soria JC, Ohe Y, Vansteenkiste J, Reungwetwattana T, Chewaskulyong B, Lee KH et al (2018) Osimertinib in untreated EGFR-mutated advanced non-small-cell lung cancer. N Engl J Med 378:113–125. https://doi.org/10.1056/NEJMoa1713137
Takenaka T, Takenoyama M, Yamaguchi M, Toyozawa R, Inamasu E, Kojo M et al (2015) Impact of the epidermal growth factor receptor mutation status on the post-recurrence survival of patients with surgically resected non-small-cell lung cancer. Eur J Cardiothorac Surg 47:550–555. https://doi.org/10.1093/ejcts/ezu227
Travis WD, Brambilla E, Noguchi M, Nicholson AG, Geisinger KR, Yatabe Y et al (2011) International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol 6:244–285. https://doi.org/10.1097/JTO.0b013e318206a221
Travis WD, Brambilla E, Nicholson A et al (2015) The 2015 World Health Organization classification of lung tumors: impact of genetic, clinical and radiologic advances since the 2004 classification. J Thorac Oncol 10(9):1243–1260
Vignot S, Frampton GM, Soria JC, Yelensky R, Commo F, Brambilla C et al (2013) Next-generation sequencing reveals high concordance of recurrent somatic alterations between primary tumor and metastases from patients with non-small-cell lung cancer. J Clin Oncol 31:2167–2172. https://doi.org/10.1200/JCO.2012.47.7737
Watanabe S, Ladas G, Goldstraw P (2002) Inter-observer variability in systematic nodal dissection: comparison of European and Japanese nodal designation. Ann Thorac Surg 73:245–248 (discussion 248–249)
Wei S, Asamura H, Kawachi R, Sakurai H, Watanabe S (2011) Which is the better prognostic factor for resected non-small cell lung cancer: the number of metastatic lymph nodes or the currently used nodal stage classification? J Thorac Oncol 6:310–318. https://doi.org/10.1097/JTO.0b013e3181ff9b45
Wisnivesky JP, Arciniega J, Mhango G, Mandeli J, Halm EA (2011) Lymph node ratio as a prognostic factor in elderly patients with pathological N1 non-small cell lung cancer. Thorax 66:287–293. https://doi.org/10.1136/thx.2010.148601
Xie F, Zhang Y, Mao X, Zheng X, Han-Zhang H, Ye J et al (2018) Comparison of genetic profiles among primary lung tumor, metastatic lymph nodes and circulating tumor DNA in treatment-naïve advanced non-squamous non-small cell lung cancer patients. Lung Cancer 121:54–60. https://doi.org/10.1016/j.lungcan.2018.05.002
Yatabe Y, Matsuo K, Mitsudomi T (2011) Heterogeneous distribution of EGFR mutations is extremely rare in lung adenocarcinoma. J Clin Oncol 29:2972–2977. https://doi.org/10.1200/JCO.2010.33.3906
Acknowledgements
This work was partially conducted at the Analysis Center of Life Science, Natural Science Center for Basic Research and Development, Hiroshima University (Hiroshima, Japan). The authors would like to thank Editage (http://www.editage.jp) for English language editing.
Funding
This work was supported by the Japan Society for the Promotion of Science (JSPS KAKENHI Grant number 15K19938) and ESTS-ACECP travelling fellowship 2013.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ito, M., Miyata, Y., Hirano, S. et al. Synchronicity of genetic variants between primary sites and metastatic lymph nodes, and prognostic impact in nodal metastatic lung adenocarcinoma. J Cancer Res Clin Oncol 145, 2325–2333 (2019). https://doi.org/10.1007/s00432-019-02978-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-019-02978-0