Abstract
Purpose
To detect new predictive markers from the prostate cancer tissue, to study the expression by cultured cancer-associated fibroblasts (CAFs) of stromal factors implicated in prostate carcinogenesis, and to compare their expressions in localized, metastatic, castration-sensitive (CSCP), castration-resistant prostate tumors (CRCP) as well as in fibroblasts from benign prostatic hyperplasia (BPH).
Materials and methods
The genomic expression of 20 stroma-derived factors, including the androgen receptor (AR), growth factors (FGF2, FGF7, FGF10, HGF, TGFβ, PDGFB), protein implicated in invasion (MMP-2, MMP-9 and MMP-11), inflammation (IL-6, IL-17, STAT-3 and NFκB), stroma/epithelium interaction (CDH11, FAP, CXCL12 and CXCL14) and chaperones (HPA1A and HSF1), was evaluated in cultured fibroblasts both from BHP and prostate carcinomas (PCa). After isolation and culture of fibroblasts by biopsy specimens, RNA was isolated and genomic studies performed.
Results
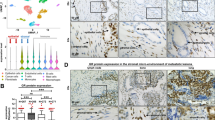
Finally, 5 BPH and 37 PCa specimens were selected: clinically localized (19), metastatic (5), CSCP (7) and CRPC (6). Interleukin-17 receptor (IL-17RB) was highly expressed in CAFs compared with fibroblasts from BPH. However, metalloproteinase-2 and chemokine ligand 14 (CXCL14) were expressed at higher levels by fibroblasts from BPH. The fibroblastic growth factor-7 was highly expressed by CAFs from localized tumors, but metalloproteinase-11 in metastatic tumors. MMP-11, androgen receptor (AR) and heat–shock-70kda-protein-1A (HSPA1A) expressions were significantly higher in CAFs from CRPC.
Conclusions
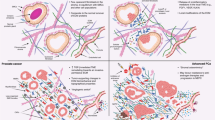
These results demonstrate a CAFs heterogeneity among prostate carcinomas with regard to some molecular profile expressions that may be relevant in tumor development (IL-17RB), progression (MMP-11) and castration resistance (AR, MMP-11 and HSPA1A).





Similar content being viewed by others
References
Adisetiyo H, Liang M, Liao CP, Jeong JH, Cohen MB, Roy-Burman P, Frenkel B (2014) Dependence of castration-resistant prostate cancer (CRPC) stem cells on CRPC-associated fibroblasts. J Cell Physiol 229:1170–1176. doi:10.1002/jcp.24546
Buhler P, Wolf P, Katzenwadel A, Schultze-Seemann W, Wetterauer U, Freudenberg N, Elsasser-Beile U (2010) Primary prostate cancer cultures are models for androgen-independent transit amplifying cells. Oncol Rep 23:465–470
Casanova-Salas I et al (2014) Identification of miR-187 and miR-182 as biomarkers of early diagnosis and prognosis in patients with prostate cancer treated with radical prostatectomy. J Urol 192:252–259. doi:10.1016/j.juro.2014.01.107
Chiarugi P, Paoli P, Cirri P (2014) Tumor microenvironment and metabolism in prostate cancer. Semin Oncol 41:267–280. doi:10.1053/j.seminoncol.2014.03.004
De Marzo AM et al (2007) Inflamm Prostate Carcinog Nat Rev Cancer 7:256–269. doi:10.1038/nrc2090
Eiro N, Fernandez-Garcia B, Gonzalez LO, Vizoso FJ (2013) Cytokines related to MMP-11 expression by inflammatory cells and breast cancer metastasis. Oncoimmunology 2:e24010. doi:10.4161/onci.24010
Eiro N, Fernandez-Garcia B, Vazquez J, Del Casar JM, Gonzalez LO, Vizoso FJ (2015) A phenotype from tumor stroma based on the expression of metalloproteases and their inhibitors, associated with prognosis in breast cancer. Oncoimmunology 4:e992222. doi:10.4161/2162402X.2014.992222
Erez N, Truitt M, Olson P, Arron ST, Hanahan D (2010) Cancer-associated fibroblasts are activated in incipient neoplasia to orchestrate tumor-promoting inflammation in an NF-kappaB-dependent manner. Cancer Cell 17:135–147. doi:10.1016/j.ccr.2009.12.041
Fernandez-Gomez J et al (2011) Relationship between metalloprotease expression in tumour and stromal cells and aggressive behaviour in prostate carcinoma: simultaneous high-throughput study of multiple metalloproteases and their inhibitors using tissue array analysis of radical prostatectomy samples. Scand J Urol Nephrol 45:171–176. doi:10.3109/00365599.2010.545074
Fujita K et al (2008) Cytokine profiling of prostatic fluid from cancerous prostate glands identifies cytokines associated with extent of tumor and inflammation. Prostate 68:872–882. doi:10.1002/pros.20755
Gandellini P et al (2014) miR-205 hinders the malignant interplay between prostate cancer cells and associated fibroblasts. Antioxid Redox Signal 20:1045–1059. doi:10.1089/ars.2013.5292
Garrido C, Brunet M, Didelot C, Zermati Y, Schmitt E, Kroemer G (2006) Heat shock proteins 27 and 70: anti-apoptotic proteins with tumorigenic properties. Cell Cycle 5:2592–2601
Gerber PA, Hippe A, Buhren BA, Muller A, Homey B (2009) Chemokines in tumor-associated angiogenesis. Biol Chem 390:1213–1223. doi:10.1515/BC.2009.144
Gonzalez L, Eiro N, Fernandez-Garcia B, Gonzalez LO, Dominguez F, Vizoso FJ (2015) Gene expression profile of normal and cancer-associated fibroblasts according to intratumoral inflammatory cells phenotype from breast cancer tissue. Mol Carcinog. doi:10.1002/mc.22403
Herranz-Amo F, Molina-Escudero R, Ogaya-Pinies G, Ramirez-Martin D, Verdu-Tartajo F, Hernandez-Fernandez C (2015) Prediction of biochemical recurrence after radical prostatectomy. New tool for selecting candidates for adjuvant radiation therapy. Actas Urol Esp. doi:10.1016/j.acuro.2015.07.006
Karkoulis PK, Stravopodis DJ, Margaritis LH, Voutsinas GE (2010) 17-Allylamino-17-demethoxygeldanamycin induces downregulation of critical Hsp90 protein clients and results in cell cycle arrest and apoptosis of human urinary bladder cancer cells. BMC Cancer 10:481. doi:10.1186/1471-2407-10-481
Kessenbrock K, Plaks V, Werb Z (2010) Matrix metalloproteinases: regulators of the tumor microenvironment. Cell 141:52–67. doi:10.1016/j.cell.2010.03.015
Kogan-Sakin I et al (2009) Prostate stromal cells produce CXCL-1, CXCL-2, CXCL-3 and IL-8 in response to epithelia-secreted IL-1. Carcinogenesis 30:698–705. doi:10.1093/carcin/bgp043
Koontongkaew S (2013) The tumor microenvironment contribution to development, growth, invasion and metastasis of head and neck squamous cell carcinomas. J Cancer 4:66–83. doi:10.7150/jca.5112
Lee YJ, Park JE, Jeon BR, Lee SM, Kim SY, Lee YK (2013) Is prostate-specific antigen effective for population screening of prostate cancer? Syst Rev Ann Lab Med 33:233–241. doi:10.3343/alm.2013.33.4.233
Marsh T, Pietras K, McAllister SS (2013) Fibroblasts as architects of cancer pathogenesis. Biochim Biophys Acta 1832:1070–1078. doi:10.1016/j.bbadis.2012.10.013
Niu Y et al (2008) Androgen receptor is a tumor suppressor and proliferator in prostate cancer. Proc Natl Acad Sci USA 105:12182–12187. doi:10.1073/pnas.0804700105
Quante M et al (2011) Bone marrow-derived myofibroblasts contribute to the mesenchymal stem cell niche and promote tumor growth. Cancer Cell 19:257–272. doi:10.1016/j.ccr.2011.01.020
Sartori DA, Chan DW (2014) Biomarkers in prostate cancer: what’s new? Curr Opin Oncol 26:259–264. doi:10.1097/CCO.0000000000000065
Tan J, Buache E, Alpy F, Daguenet E, Tomasetto CL, Ren GS, Rio MC (2014) Stromal matrix metalloproteinase-11 is involved in the mammary gland postnatal development. Oncogene 33:4050–4059. doi:10.1038/onc.2013.434
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65:87–108. doi:10.3322/caac.21262
Vizoso FJ et al (2007) Study of matrix metalloproteinases and their inhibitors in breast cancer. Br J Cancer 96:903–911. doi:10.1038/sj.bjc.6603666
Ward JF, Blute ML, Slezak J, Bergstralh EJ, Zincke H (2003) The long-term clinical impact of biochemical recurrence of prostate cancer 5 or more years after radical prostatectomy. J Urol 170:1872–1876. doi:10.1097/01.ju.0000091876.13656.2e
Williams KA et al (2014) A systems genetics approach identifies CXCL14, ITGAX, and LPCAT2 as novel aggressive prostate cancer susceptibility genes. Plos Genet 10:e1004809. doi:10.1371/journal.pgen.1004809
Yu S, Xia S, Yang D, Wang K, Yeh S, Gao Z, Chang C (2013) Androgen receptor in human prostate cancer-associated fibroblasts promotes prostate cancer epithelial cell growth and invasion. Med Oncol 30:674. doi:10.1007/s12032-013-0674-9
Acknowledgements
This work was supported by a grant from Abbvie Inc, Chicago (Study No: ACA-SPAI-10-24). The expert technical assistance of Sandra Cid was gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Noemi Eiro and Jesus Fernandez-Gomez have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Eiro, N., Fernandez-Gomez, J., Sacristán, R. et al. Stromal factors involved in human prostate cancer development, progression and castration resistance. J Cancer Res Clin Oncol 143, 351–359 (2017). https://doi.org/10.1007/s00432-016-2284-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-016-2284-3