Abstract
Purpose
As one form of tumor invasion, cancer cells can invade the extracellular matrix (ECM) through tracks that have been physically remodeled by cancer-associated fibroblasts (CAFs). However, CAFs are a heterogeneous population with diverse matrix-remodeling capacities. The purpose of this study was to investigate how CAFs with various matrix-remodeling capacities influence cancer cell invasion.
Methods
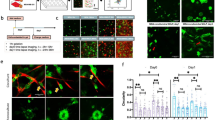
We established single-cell-derived clones from three primary cultures of CAFs from lung adenocarcinoma patients (Case 1, 5 clones; Case 2, 5 clones; and Case 3, 7 clones). Using a co-culture model, we evaluated the correlations between the number of invaded cancer cells and the remodeling areas generated by CAF clones in each case.
Results
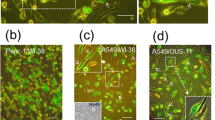
When A549 lung adenocarcinoma cells and CAF clones were co-cultured, both the numbers of invaded cancer cells and the remodeling areas generated by the CAF clones varied greatly. The number of invaded cancer cells was moderately and strongly correlated with the remodeling areas generated by each CAF clone originating from Cases 1 and 2 (R 2 value = 0.53 and 0.68, respectively), suggesting that the remodeling areas in the ECM may determine the number of invaded cancer cells. In contrast, the number of invaded cancer cells was not correlated with the remodeling areas generated by CAF clones originating from Case 3, suggesting that factors other than the remodeling areas might determine the number of invading cancer cells.
Conclusions
These findings showed two types of fibroblast-dependent cancer cell invasion that are dependent on and independent of the remodeling areas generated by CAFs.





Similar content being viewed by others
References
Allen M, Louise Jones J (2011) Jekyll and Hyde: the role of the microenvironment on the progression of cancer. J Pathol 223:162–176. doi:10.1002/path.2803
Allinen M et al (2004) Molecular characterization of the tumor microenvironment in breast cancer. Cancer Cell 6:17–32. doi:10.1016/j.ccr.2004.06.010
Bauer M, Su G, Casper C, He R, Rehrauer W, Friedl A (2010) Heterogeneity of gene expression in stromal fibroblasts of human breast carcinomas and normal breast. Oncogene 29:1732–1740. doi:10.1038/onc.2009.463
Bissell MJ, Radisky D (2001) Putting tumours in context. Nat Rev Cancer 1:46–54. doi:10.1038/35094059
Bonaventure J, Domingues MJ, Larue L (2013) Cellular and molecular mechanisms controlling the migration of melanocytes and melanoma cells. Pigment Cell Melanoma Res 26:316–325. doi:10.1111/Pcmr.12080
Bremnes RM et al (2011) The role of tumor stroma in cancer progression and prognosis: emphasis on carcinoma-associated fibroblasts and non-small cell lung cancer. J Thorac Oncol 6:209–217. doi:10.1097/JTO.0b013e3181f8a1bd
Cameron MD et al (2000) Temporal progression of metastasis in lung: cell survival, dormancy, and location dependence of metastatic inefficiency. Cancer Res 60:2541–2546
Counter CM et al (1998) Dissociation among in vitro telomerase activity, telomere maintenance, and cellular immortalization. Proc Natl Acad Sci USA 95:14723–14728
De Wever O, Demetter P, Mareel M, Bracke M (2008) Stromal myofibroblasts are drivers of invasive cancer growth. Int J Cancer 123:2229–2238. doi:10.1002/ijc.23925
Friedl P, Locker J, Sahai E, Segall JE (2012) Classifying collective cancer cell invasion. Nat Cell Biol 14:777–783. doi:10.1038/ncb2548
Gaggioli C, Hooper S, Hidalgo-Carcedo C, Grosse R, Marshall JF, Harrington K, Sahai E (2007) Fibroblast-led collective invasion of carcinoma cells with differing roles for RhoGTPases in leading and following cells. Nat Cell Biol 9:1392–1400. doi:10.1038/ncb1658
Guttery DS, Shaw JA, Lloyd K, Pringle JH, Walker RA (2010) Expression of tenascin-C and its isoforms in the breast. Cancer Metastasis Rev 29:595–606. doi:10.1007/s10555-010-9249-9
Hoshino A, Chiba H, Nagai K, Ishii G, Ochiai A (2008) Human vascular adventitial fibroblasts contain mesenchymal stem/progenitor cells. Biochem Biophys Res Commun 368:305–310. doi:10.1016/j.bbrc.2008.01.090
Ishii G et al (2010) Fibroblasts associated with cancer cells keep enhanced migration activity after separation from cancer cells: a novel character of tumor educated fibroblasts. Int J Oncol 37:317–325
Ito S et al (2012) Tumor promoting effect of podoplanin-positive fibroblasts is mediated by enhanced RhoA activity. Biochem Biophys Res Commun 422:194–199. doi:10.1016/j.bbrc.2012.04.158
Jodele S, Blavier L, Yoon JM, DeClerck YA (2006) Modifying the soil to affect the seed: role of stromal-derived matrix metalloproteinases in cancer progression. Cancer Metastasis Rev 25:35–43. doi:10.1007/s10555-006-7887-8
Kalluri R, Zeisberg M (2006) Fibroblasts in cancer. Nat Rev Cancer 6:392–401. doi:10.1038/nrc1877
Kharaishvili G, Simkova D, Bouchalova K, Gachechiladze M, Narsia N, Bouchal J (2014) The role of cancer-associated fibroblasts, solid stress and other microenvironmental factors in tumor progression and therapy resistance. Cancer Cell Int 14:41. doi:10.1186/1475-2867-14-41
Masutomi K et al (2003) Telomerase maintains telomere structure in normal human cells. Cell 114:241–253
Neri S, Ishii G, Hashimoto H, Kuwata T, Nagai K, Date H, Ochiai A (2015) Podoplanin-expressing cancer-associated fibroblasts lead and enhance the local invasion of cancer cells in lung adenocarcinoma. Int J Cancer. doi:10.1002/ijc.29464
Ohlund D, Elyada E, Tuveson D (2014) Fibroblast heterogeneity in the cancer wound. J Exp Med 211:1503–1523. doi:10.1084/jem.20140692
Orimo A et al (2005) Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121:335–348. doi:10.1016/j.cell.2005.02.034
Oskarsson T et al (2011) Breast cancer cells produce tenascin C as a metastatic niche component to colonize the lungs. Nat Med 17:867–874. doi:10.1038/nm.2379
Paulsson J, Micke P (2014) Prognostic relevance of cancer-associated fibroblasts in human cancer. Semin Cancer Biol 25:61–68. doi:10.1016/j.semcancer.2014.02.006
Pietras K, Pahler J, Bergers G, Hanahan D (2008) Functions of paracrine PDGF signaling in the proangiogenic tumor stroma revealed by pharmacological targeting. PLoS Med 5:e19. doi:10.1371/journal.pmed.0050019
Satoyoshi R, Kuriyama S, Aiba N, Yashiro M, Tanaka M (2015) Asporin activates coordinated invasion of scirrhous gastric cancer and cancer-associated fibroblasts. Oncogene 34:650–660. doi:10.1038/onc.2013.584
Taddei ML, Giannoni E, Comito G, Chiarugi P (2013) Microenvironment and tumor cell plasticity: an easy way out. Cancer Lett 341:80–96. doi:10.1016/j.canlet.2013.01.042
Tlsty TD, Coussens LM (2006) Tumor stroma and regulation of cancer development. Annu Rev Pathol 1:119–150. doi:10.1146/annurev.pathol.1.110304.100224
Acknowledgments
S. Neri is a recipient of the Research Resident Fellowship from the Research for Promotion of Cancer Control Programmes (Japan).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to disclose.
Funding
This work was supported by the National Cancer Center Research and Development Fund (23-A-12 and 23-K-18), the Foundation for the Promotion of Cancer Research, the Third-Term Comprehensive 10-Year Strategy for Cancer Control, the Advanced Research for Medical Products Mining Programme of the National Institute of Biomedical Innovation (NIBIO), and JSPS KAKENHI (24659185).
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
432_2015_2046_MOESM1_ESM.pptx
Supplementary Fig. 1. Examples of image analysis using CL-Quant software. Detection of the invasion region using the first frame of the phase-contrast images (upper panel). Detection of the remodeling areas generated by the invaded CAFs in the invasion region using phase-contrast images (middle panel, pink region). Detection of the number of cancer cells in the invasion region using fluorescence images (lower panel, green region). (PPTX 624 kb)
432_2015_2046_MOESM2_ESM.pptx
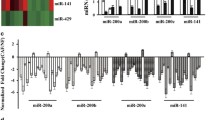
Supplementary Fig. 2. Relationship between the relative mRNA expressions (/GAPDH) of the CAF clones and the invaded cancer cells in Case 3. R2 = coefficient of determination. (PPTX 107 kb)
432_2015_2046_MOESM3_ESM.wmv
Supplementary Movie 1. Time-lapse imaging of single-cell cloning of CAFs. Representative time-lapse images for 1 week of the single-cell cloning of CAFs expressing Venus in their cytoplasm (green). (WMV 6466 kb)
Rights and permissions
About this article
Cite this article
Neri, S., Hashimoto, H., Kii, H. et al. Cancer cell invasion driven by extracellular matrix remodeling is dependent on the properties of cancer-associated fibroblasts. J Cancer Res Clin Oncol 142, 437–446 (2016). https://doi.org/10.1007/s00432-015-2046-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-015-2046-7