Abstract
Purpose
Filamin A (FLNa) cross-links actin filaments into dynamic orthogonal networks and interacts with binding proteins of diverse cellular functions that are implicated in cell growth and motility regulation. Here, we tested the hypothesis that FLNa plays a role in cancer proliferation and metastasis via the regulation of epidermal growth factor receptor (EGFR) function.
Methods
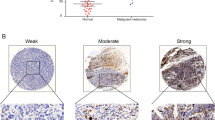
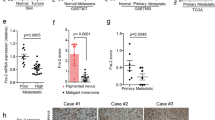
Ectopic expression and knockdown of FLNa in human melanoma cell lines was performed to investigate changes in cellular proliferation, migration and invasion in vitro and tumor growth in a xenograft model in the mouse. The role of FLNa in EGFR expression and signaling was evaluated by Western blot. Immunohistochemistry was performed on histological sections of human melanoma tumors to determine whether an association existed between FLNa and overall survival.
Results
The depletion of FLNa significantly reduced the proliferation, migration and invasion of two melanoma cell lines in vitro and was associated with smaller tumors in a xenograft model in vivo. EGF-induced phosphorylation of EGFR and activation of the Raf-MEK-ERK cascade was negatively affected by the silencing of FLNa both in vitro and in vivo. Cancer patients with low melanoma tumor FLNa expression have improved survival benefit.
Conclusion
These data indicate that enhanced tumorigenesis occurs through increase in EGF-induced EGFR activation in FLNa-expressing melanoma cells and that high FLNa levels are predictors of negative outcome for patients with melanoma tumors.





Similar content being viewed by others
Abbreviations
- FLNa:
-
Filamin A
- EGFR:
-
Epidermal growth factor receptor
- ERK:
-
Ras-extracellular signal-regulated kinase
- JNK:
-
c-Jun NH2-terminal kinase
- MEM:
-
Minimal essential medium
- FBS:
-
Fetal bovine serum
- SFM:
-
Serum-free medium
- EGF:
-
Epidermal growth factor
- ECL:
-
Enhanced chemiluminescence
- pY-EGFR:
-
Phosphorylated form of EGFR
- pERK1/2:
-
Phosphorylated ERK1/2
- IHC:
-
Immunohistochemical
- MMPs:
-
Matrix metalloproteinases
- ECM:
-
Extracellular matrix
References
Alper O, Stetler-Stevenson WG, Harris LN, Leitner WW, Ozdemirli M, Hartmann D, Raffeld M, Abu-Asab M, Byers S, Zhuang Z, Oldfield EH, Tong Y, Bergmann-Leitner E, Criss WE, Nagasaki K, Mok SC, Cramer DW, Karaveli FS, Goldbach-Mansky R, Leo P, Stromberg K, Weil RJ (2009) Novel anti-filamin-A antibody detects a secreted variant of filamin-A in plasma from patients with breast carcinoma and high-grade astrocytoma. Cancer Sci 100(9):1748–1756. doi:10.1111/j.1349-7006.2009.01244.x
Avraham R, Yarden Y (2011) Feedback regulation of EGFR signaling: decision making by early and delayed loops. Nat Rev Mol Cell Biol 12(2):104–117. doi:10.1038/nrm3048
Barriere H, Nemes C, Du K, Lukacs GL (2007) Plasticity of polyubiquitin recognition as lysosomal targeting signals by the endosomal sorting machinery. Mol Biol Cell 18(10):3952–3965. doi:10.1091/mbc.E07-07-0678
Baselga J, Albanell J (2002) Epithelial growth factor receptor interacting agents. Hematol/Oncol Clin N Am 16(5):1041–1063
Bedolla RG, Wang Y, Asuncion A, Chamie K, Siddiqui S, Mudryj MM, Prihoda TJ, Siddiqui J, Chinnaiyan AM, Mehra R, de Vere White RW, Ghosh PM (2009) Nuclear versus cytoplasmic localization of filamin A in prostate cancer: immunohistochemical correlation with metastases. Clin Cancer Res 15(3):788–796. doi:10.1158/1078-0432.CCR-08-1402
Bullock MD, Sayan AE, Packham GK, Mirnezami AH (2012) MicroRNAs: critical regulators of epithelial to mesenchymal (EMT) and mesenchymal to epithelial transition (MET) in cancer progression. Biol Cell 104(1):3–12. doi:10.1111/boc.201100115
Cano CE, Motoo Y, Iovanna JL (2010) Epithelial-to-mesenchymal transition in pancreatic adenocarcinoma. Sci World J 10:1947–1957. doi:10.1100/tsw.2010.183
Citri A, Yarden Y (2006) EGF–ERBB signaling: towards the systems level. Nat Rev Mol Cell Biol 7(7):505–516. doi:10.1038/nrm1962
Cock-Rada A, Weitzman JB (2013) The methylation landscape of tumour metastasis. Biol Cell 105(2):73–90. doi:10.1111/boc.201200029
Djinovic-Carugo K, Carugo O (2010) Structural portrait of filamin interaction mechanisms. Cur Protein Pept Sci 11(7):639–650
Eggermont AM, Spatz A, Robert C (2014) Cutaneous melanoma. Lancet 383(9919):816–827. doi:10.1016/S0140-6736(13)60802-8
Fernandez-Flores A (2012) Prognostic factors for melanoma progression and metastasis: from Hematoxylin–Eosin to genetics. Rom J Morphol Embryol 53(3):449–459
Fiori JL, Zhu TN, O’Connell MP, Hoek KS, Indig FE, Frank BP, Morris C, Kole S, Hasskamp J, Elias G, Weeraratna AT, Bernier M (2009) Filamin A modulates kinase activation and intracellular trafficking of epidermal growth factor receptors in human melanoma cells. Endocrinology 150(6):2551–2560. doi:10.1210/en.2008-1344
Flanagan MB, Dabbs DJ, Brufsky AM, Beriwal S, Bhargava R (2008) Histopathologic variables predict Oncotype DX recurrence score. Mod Pathol 21(10):1255–1261. doi:10.1038/modpathol.2008.54
Flevaris P, Stojanovic A, Gong H, Chishti A, Welch E, Du X (2007) A molecular switch that controls cell spreading and retraction. J Cell Biol 179(3):553–565. doi:10.1083/jcb.200703185
Grandal MV, Madshus IH (2008) Epidermal growth factor receptor and cancer: control of oncogenic signaling by endocytosis. J Cell Mol Med 12(5A):1527–1534. doi:10.1111/j.1582-4934.2008.00298.x
Grovdal LM, Stang E, Sorkin A, Madshus IH (2004) Direct interaction of Cbl with pTyr 1045 of the EGF receptor (EGFR) is required to sort the EGFR to lysosomes for degradation. Exp Cell Res 300(2):388–395. doi:10.1016/j.yexcr.2004.07.003
Grunewald TG, Herbst SM, Heinze J, Burdach S (2011) Understanding tumor heterogeneity as functional compartments—superorganisms revisited. J Trans Med 9:79. doi:10.1186/1479-5876-9-79
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674. doi:10.1016/j.cell.2011.02.013
He HJ, Zhu TN, Xie Y, Fan J, Kole S, Saxena S, Bernier M (2006) Pyrrolidine dithiocarbamate inhibits interleukin-6 signaling through impaired STAT3 activation and association with transcriptional coactivators in hepatocytes. J Biol Chem 281(42):31369–31379. doi:10.1074/jbc.M603762200
Hodis E, Watson IR, Kryukov GV, Arold ST, Imielinski M, Theurillat JP, Nickerson E, Auclair D, Li L, Place C, Dicara D, Ramos AH, Lawrence MS, Cibulskis K, Sivachenko A, Voet D, Saksena G, Stransky N, Onofrio RC, Winckler W, Ardlie K, Wagle N, Wargo J, Chong K, Morton DL, Stemke-Hale K, Chen G, Noble M, Meyerson M, Ladbury JE, Davies MA, Gershenwald JE, Wagner SN, Hoon DS, Schadendorf D, Lander ES, Gabriel SB, Getz G, Garraway LA, Chin L (2012) A landscape of driver mutations in melanoma. Cell 150(2):251–263. doi:10.1016/j.cell.2012.06.024
Hynes NE, MacDonald G (2009) ErbB receptors and signaling pathways in cancer. Curr Opin Cell Biol 21(2):177–184. doi:10.1016/j.ceb.2008.12.010
Jiang X, Yue J, Lu H, Campbell N, Yang Q, Lan S, Haffty BG, Yuan C, Shen Z (2013) Inhibition of filamin-A reduces cancer metastatic potential. Int J Biol Sci 9(1):67–77. doi:10.7150/ijbs.5577
Kim EK, Choi EJ (2010) Pathological roles of MAPK signaling pathways in human diseases. Biochim Biophys Acta 1802(4):396–405. doi:10.1016/j.bbadis.2009.12.009
Kim H, McCulloch CA (2011) Filamin A mediates interactions between cytoskeletal proteins that control cell adhesion. FEBS Lett 585(1):18–22. doi:10.1016/j.febslet.2010.11.033
Kim H, Nakamura F, Lee W, Hong C, Perez-Sala D, McCulloch CA (2010) Regulation of cell adhesion to collagen via beta1 integrins is dependent on interactions of filamin A with vimentin and protein kinase C epsilon. Exp Cell Res 316(11):1829–1844. doi:10.1016/j.yexcr.2010.02.007
Kim Y, Stolarska MA, Othmer HG (2011) The role of the microenvironment in tumor growth and invasion. Prog Biophys Mol Biol 106(2):353–379. doi:10.1016/j.pbiomolbio.2011.06.006
Kunz M (2014) Oncogenes in melanoma: an update. Eur J Cell Biol 93(1–2):1–10. doi:10.1016/j.ejcb.2013.12.002
Kyndt F, Gueffet JP, Probst V, Jaafar P, Legendre A, Le Bouffant F, Toquet C, Roy E, McGregor L, Lynch SA, Newbury-Ecob R, Tran V, Young I, Trochu JN, Le Marec H, Schott JJ (2007) Mutations in the gene encoding filamin A as a cause for familial cardiac valvular dystrophy. Circulation 115(1):40–49. doi:10.1161/CIRCULATIONAHA.106.622621
Lemmon MA, Schlessinger J (2010) Cell signaling by receptor tyrosine kinases. Cell 141(7):1117–1134. doi:10.1016/j.cell.2010.06.011
Leung R, Wang Y, Cuddy K, Sun C, Magalhaes J, Grynpas M, Glogauer M (2010) Filamin A regulates monocyte migration through Rho small GTPases during osteoclastogenesis. J Bone Min Res 25(5):1077–1091. doi:10.1359/jbmr.091114
Loy CJ, Sim KS, Yong EL (2003) Filamin-A fragment localizes to the nucleus to regulate androgen receptor and coactivator functions. Proc Natl Acad Sci USA 100(8):4562–4567. doi:10.1073/pnas.0736237100
Maurer G, Tarkowski B, Baccarini M (2011) Raf kinases in cancer-roles and therapeutic opportunities. Oncogene 30(32):3477–3488. doi:10.1038/onc.2011.160
McCubrey JA, Steelman LS, Chappell WH, Abrams SL, Wong EW, Chang F, Lehmann B, Terrian DM, Milella M, Tafuri A, Stivala F, Libra M, Basecke J, Evangelisti C, Martelli AM, Franklin RA (2007) Roles of the Raf/MEK/ERK pathway in cell growth, malignant transformation and drug resistance. Biochim Biophys Acta 1773(8):1263–1284. doi:10.1016/j.bbamcr.2006.10.001
Mendelsohn J, Baselga J (2003) Status of epidermal growth factor receptor antagonists in the biology and treatment of cancer. J Clin Oncol 21(14):2787–2799. doi:10.1200/JCO.2003.01.504
Nakamura F, Heikkinen O, Pentikainen OT, Osborn TM, Kasza KE, Weitz DA, Kupiainen O, Permi P, Kilpelainen I, Ylanne J, Hartwig JH, Stossel TP (2009) Molecular basis of filamin A-FilGAP interaction and its impairment in congenital disorders associated with filamin A mutations. PLoS ONE 4(3):e4928. doi:10.1371/journal.pone.0004928
Nakamura F, Stossel TP, Hartwig JH (2011) The filamins: organizers of cell structure and function. Cell Adh Migr 5(2):160–169
Nallapalli RK, Ibrahim MX, Zhou AX, Bandaru S, Sunkara SN, Redfors B, Pazooki D, Zhang Y, Boren J, Cao Y, Bergo MO, Akyurek LM (2012) Targeting filamin A reduces K-RAS-induced lung adenocarcinomas and endothelial response to tumor growth in mice. Mol Cancer 11:50. doi:10.1186/1476-4598-11-50
Neuzillet C, Tijeras-Raballand A, de Mestier L, Cros J, Faivre S, Raymond E (2014) MEK in cancer and cancer therapy. Pharmacol Ther 141(2):160–171. doi:10.1016/j.pharmthera.2013.10.001
Ravid D, Chuderland D, Landsman L, Lavie Y, Reich R, Liscovitch M (2008) Filamin A is a novel caveolin-1-dependent target in IGF-I-stimulated cancer cell migration. Exp Cell Res 314(15):2762–2773. doi:10.1016/j.yexcr.2008.06.004
Razinia Z, Makela T, Ylanne J, Calderwood DA (2012) Filamins in mechanosensing and signaling. Ann Rev Biophys 41:227–246. doi:10.1146/annurev-biophys-050511-102252
Roepstorff K, Grovdal L, Grandal M, Lerdrup M, van Deurs B (2008) Endocytic downregulation of ErbB receptors: mechanisms and relevance in cancer. Histochem Cell Biol 129(5):563–578. doi:10.1007/s00418-008-0401-3
Savoy RM, Ghosh PM (2013) The dual role of filamin A in cancer: can’t live with (too much of) it, can’t live without it. Endocr-Related Cancer 20(6):R341–R356. doi:10.1530/ERC-13-0364
Wang Y, Kreisberg JI, Bedolla RG, Mikhailova M, de Vere White RW, Ghosh PM (2007) A 90 kDa fragment of filamin A promotes Casodex-induced growth inhibition in Casodex-resistant androgen receptor positive C4-2 prostate cancer cells. Oncogene 26(41):6061–6070. doi:10.1038/sj.onc.1210435
Wickstead B, Gull K (2011) The evolution of the cytoskeleton. J Cell Biol 194(4):513–525. doi:10.1083/jcb.201102065
Yarden Y, Pines G (2012) The ERBB network: at last, cancer therapy meets systems biology. Nat Rev Cancer 12(8):553–563. doi:10.1038/nrc3309
Yewale C, Baradia D, Vhora I, Patil S, Misra A (2013) Epidermal growth factor receptor targeting in cancer: a review of trends and strategies. Biomaterials 34(34):8690–8707. doi:10.1016/j.biomaterials.2013.07.100
Zhou AX, Hartwig JH, Akyurek LM (2010) Filamins in cell signaling, transcription and organ development. Trends Cell Biol 20(2):113–123. doi:10.1016/j.tcb.2009.12.001
Zhu TN, He HJ, Kole S, D’Souza T, Agarwal R, Morin PJ, Bernier M (2007) Filamin A-mediated down-regulation of the exchange factor Ras-GRF1 correlates with decreased matrix metalloproteinase-9 expression in human melanoma cells. J Biol Chem 282(20):14816–14826. doi:10.1074/jbc.M611430200
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 81071846, to T.N.Z); Natural Science Foundation of Hebei Province of China (No. H2013505059, to T.N.Z); Department of Science and Technology of Hebei Province of China (No. 12396107D, to R.J.Z); Wu Jieping Foundation (No. 320.6750.12604, to T.N.Z) and, in part, by the Intramural Research Program of National Institutes of Health, National Institute on Aging.
Conflict of interest
We, all authors declare that there is no any financial arrangements that could be related to the manuscript and confirm that we do not have any disclosure to make at submission. None of the authors has any potential financial conflict of interest related to this manuscript.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhang, K., Zhu, T., Gao, D. et al. Filamin A expression correlates with proliferation and invasive properties of human metastatic melanoma tumors: implications for survival in patients. J Cancer Res Clin Oncol 140, 1913–1926 (2014). https://doi.org/10.1007/s00432-014-1722-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-014-1722-3