Abstract
Purpose
NF-κB transcription factor has been associated with cancer development and chemoresistance. We studied the signaling pathway activated by doxorubicin (DOX) leading to NF-κB activation in breast cancer cells.
Methods
NF-κB activity was evaluated by electrophoretic mobility shift in T47D, ZR75.30 and primary culture (MBCDF) from a ductal infiltrating carcinoma. Cell viability was measured by crystal violet. Western blotting was performed to check the expression and phosphorylation of IκBα Ser-32/36. c-Abl was inhibited with Imatinib or by overexpressing a dominant negative form of c-Abl (K290R).
Results
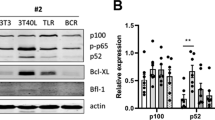
We found a correlation between sensitivity to DOX and amplitude of NF-κB activation. In cells least sensitive to DOX, NF-κB remained activated for longer time (T47D and MBCDF). The opposite effect was observed in cells sensitive to DOX (ZR75.30). DOX did not induce IκBα degradation or Ser-32/36 phosphorylation. Instead, there were modifications in the levels of IκBα tyrosine phosphorylation, suggesting an atypical NF-κB activation. In DOX-resistant cells, Imatinib treatment reduced IκBα tyrosine phosphorylation and NF-κB activity. The Imatinib–DOX combination significantly enhanced cell death of T47D and MBCDF breast cancer cells. Overexpression of c-Abl K290R in T47D and MBCDF cells reduced basal and DOX-induced NF-κB activation as well as IκBα tyrosine phosphorylation. In c-Abl K290R cells, DOX treatment did not mimic the combination Imatinib–DOX-induced cell death.
Conclusions
Inhibition of c-Abl inactivated IκBα/NF-κB pathway is associated with IκBα tyrosine phosphorylation in breast cancer cells. These results also raise the potential use of a combined therapy with Imatinib and DOX for breast cancer patients.






Similar content being viewed by others
Abbreviations
- NF-κB:
-
Nuclear factor kappa-light-chain-enhancer of activated B cells
- IκBα:
-
Nuclear factor of kappa light polypeptide gene enhancer in B cell inhibitor, alpha
- DOX:
-
Doxorubicin
- EMSA:
-
Electrophoretic mobility shift assay
References
Baldwin AS (2001) Control of oncogenesis and cancer therapy resistance by the transcription factor NF-κB. J Clin Invest 107(3):241–246
Bargou RC, Emmerich F, Krappmann D, Bommert K, Mapara MY, Arnold W, Roger HD, Grinstein E, Scheidereit C (1997) Constitutive nuclear factor-κB-RelA activation is required for proliferation and survival of Hodgkin’s disease tumor cells. J Clin Invest 100:2961–2969
Basseres DS, Baldwin AS (2006) Nuclear factor-κB and inhibitor of κB kinase pathways in oncogenic initiation and progression. Oncogene 25(51):6817–6830
Bednarski BK, Ding XY, Coombe K, Baldwin AS, Kim HJ (2008) Active roles for inhibitory kappa B kinases alpha and beta in nuclear factor-kappa B-mediated chemoresistance to doxorubicin. Mol Cancer Ther 7(7):1827–1835
Biswas DK, Shi Q, Baily S, Strickland I, Ghosh S, Pardee AB, Iglehart JD (2004) NF-κB activation in human breast cancer specimens and its role in cell proliferation and apoptosis. Proc Natl Acad Sci USA 101(27):10137–10142
Cogswell PC, Guttridge DC, Funkhouser WK, Baldwin AS Jr (2000) Selective activation of NF-κB subunits in human breast cancer: potential roles for NF-κB2/p52 and for Bcl-3. Oncogene 19(9):1123–1131
Cusack JC Jr, Liu R, Baldwin AS Jr (2000) Inducible chemoresistance to 7-ethyl-10-[4-(1-piperidino)-1-piperidino]- carbonyloxycamptothe cin (CPT-11) in colorectal cancer cells and a xenograft model is overcome by inhibition of nuclear factor-κB activation. Cancer Res 60(9):2323–2330
Cusack JC Jr, Liu R, Houston M, Abendroth K, Elliott PJ, Adams J, Baldwin AS Jr (2001) Enhanced chemosensitivity to CPT-11 with proteasome inhibitor PS-341: implications for systemic nuclear factor-kappaB inhibition. Cancer Res 61(9):3535–3540
DeSantis C, Jemal A, Ward E, Thun MJ (2008) Temporal trends in breast cancer mortality by state and race. Cancer Causes Control 19(5):537–545
DiDonato JA, Mercurio F, Karin M (2012) NF-κB and the link between inflammation and cancer. Immunol Rev 246(1):379–400. doi:10.1111/j.1600-065X.2012.01099.x
Garg A, Aggarwal BB (2002) Nuclear transcription factor-kappaB as a target for cancer drug development. Leukemia 16(6):1053–1068
Hayden MS, Ghosh S (2004) Signaling to NF-κB. Genes Dev 18(18):2195–2224
Ibarra-Sánchez M, Wagner J, Ong M-T, Lampron C, Tremblay M (2001) Murine embryonic fibroblast lacking TC-PTP display delayed G1 phase through a defective NF-κB activation. Oncogene 20:4728–4739
Imbert V, Rupec RA, Livolsi A, Pahl HL, Traenckner EB, Mueller-Dieckmann C, Farahifar D, Rossi B, Auberger P, Baeuerle PA, Peyron JF (1996) Tyrosine phosphorylation of IκB-α activates NF-κB without proteolytic degradation of IκB-α. Cell 86(5):787–798
Janssens S, Tinel A, Lippens S, Tschopp J (2005) PIDD mediates NF-κB activation in response to DNA damage. Cell 123(6):1079–1092
Jemal A, Siegel R, Ward E, Hao YP, Xu JQ, Murray T, Thun MJ (2008) Cancer statistics, 2008. Ca-a Cancer J Clin 58(2):71–96
Karin M (2006) Nuclear factor-κB in cancer development and progression. Nature 441(7092):431–436
Karin M, Greten FR (2005) NF-κB: linking inflammation and immunity to cancer development and progression. Nat Rev Immunol 5(10):749–759
Karin M, Lin A (2002) NF-κB at the crossroads of life and death. Nat Immunol 3(3):221–227
Karin M, Cao Y, Greten FR, Li ZW (2002) NF-κB in cancer: from innocent bystander to major culprit. Nat Rev Cancer 2(4):301–310
Kawai H, Nie L, Yuan ZM (2002) Inactivation of NF-κB-dependent cell survival, a novel mechanism for the proapoptotic function of c-Abl. Mol Cell Biol 22(17):6079–6088
Kim HJ, Hawke N, Baldwin AS (2006) NF-κB and IKK as therapeutic targets in cancer. Cell Death Differ 13(5):738–747
Mabb AM, Wuerzberger-Davis SM, Miyamoto S (2006) PIASy mediates NEMO sumoylation and NF-κB activation in response to genotoxic stress. Nat Cell Biol 8(9):986–993
Miura K, Karasawa H, Sasaki I (2009) cIAP2 as a therapeutic target in colorectal cancer and other malignancies. Expert Opin Ther Targets 13(11):1333–1345. doi:10.1517/14728220903277256
Moynagh PN (2005) The NF-κB pathway. J Cell Sci 118(Pt 20):4589–4592
Mukhopadhyay A, Manna SK, Aggarwal BB (2000) Pervanadate-induced nuclear factor-κB activation requires tyrosine phosphorylation and degradation of IκBα. Comparison with tumor necrosis factor-alpha. J Biol Chem 275(12):8549–8555
Nakshatri H, BhatNakshatri P, Martin DA, Goulet RJ, Sledge GW (1997) Constitutive activation of NF-κB during progression of breast cancer to hormone-independent growth. Mol Cell Biol 17(7):3629–3639
Pahl HL (1999) Activators and target genes of Rel/NF-κB transcription factors. Oncogene 18(49):6853–6866
Perkins ND (2006) Post-translational modifications regulating the activity and function of the nuclear factor kappa B pathway. Oncogene 25(51):6717–6730. doi:10.1038/sj.onc.1209937
Schoonbroodt S, Ferreira V, Best-Belpomme M, Boelaert JR, Legrand-Poels S, Korner M, Piette J (2000) Crucial role of the amino-terminal tyrosine residue 42 and the carboxyl-terminal PEST domain of IκBα in NF-κB activation by an oxidative stress. J Immunol 164(8):4292–4300
Singh S, Darnay BG, Aggarwal BB (1996) Site-specific tyrosine phosphorylation of IκBα negatively regulates its inducible phosphorylation and degradation. J Biol Chem 271(49):31049–31054
Srinivasan D, Plattner R (2006) Activation of Abl tyrosine kinases promotes invasion of aggressive breast cancer cells. Cancer Res 66(11):5648–5655
Srinivasan D, Sims JT, Plattner R (2008) Aggressive breast cancer cells are dependent on activated Abl kinases for proliferation, anchorage-independent growth and survival. Oncogene 27(8):1095–1105
Tergaonkar V, Bottero V, Ikawa M, Li Q, Verma IM (2003) IκB kinase-independent IκBα degradation pathway: functional NF-κB activity and implications for cancer therapy. Mol Cell Biol 23(22):8070–8083
Wang CY, Cusack JC Jr, Liu R, Baldwin AS Jr (1999) Control of inducible chemoresistance: enhanced anti-tumor therapy through increased apoptosis by inhibition of NF-κB. Nat Med 5(4):412–417
Weigel MT, Dahmke L, Schem C, Bauerschlag DO, Weber K, Niehoff P, Bauer M, Strauss A, Jonat W, Maass N, Mundhenke C (2010) In vitro effects of imatinib mesylate on radiosensitivity and chemosensitivity of breast cancer cells. BMC Cancer 10:412. doi:10.1186/1471-2407-10-412
Wu JT, Kral JG (2005) The NF-κB/IκB signaling system: a molecular target in breast cancer therapy. J Surg Res 123(1):158–169
Yang WS, Chang JW, Han NJ, Lee SK, Park SK (2012) Spleen tyrosine kinase mediates high glucose-induced transforming growth factor-β1 up-regulation in proximal tubular epithelial cells. Exp Cell Res 318(15):1867–1876. doi:10.1016/j.yexcr.2012.05.016
Zhang J, Xin X, Chen Q, Xie Z, Gui M, Chen Y, Lin L, Feng J, Li Q, Ding J, Geng M (2012) Oligomannurarate sulfate sensitizes cancer cells to doxorubicin by inhibiting atypical activation of NF-κB via targeting of Mre11. Int J Cancer 130(2):467–477. doi:10.1002/ijc.26021
Acknowledgments
We acknowledge the technical work of Karla Alejandra López Zelada, Julio César Garibay Díaz and Carmita Pérez Guitar. We are grateful to Dr. Adolfo García-Sainz and Dr. Alberto Huberman for their critical review of the manuscript. MJIS and AZD are members of The National System for Researchers (Sistema Nacional de Investigadores, SNI). This work was funded by operating grants to MJIS from CONACyT [102825-3] and AZD from Dirección General del Personal Académico, UNAM (DGAPA) [IN229607-2].
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Esparza-López, J., Medina-Franco, H., Escobar-Arriaga, E. et al. Doxorubicin induces atypical NF-κB activation through c-Abl kinase activity in breast cancer cells. J Cancer Res Clin Oncol 139, 1625–1635 (2013). https://doi.org/10.1007/s00432-013-1476-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-013-1476-3